- The process of creating a drug - May 23, 2023
- How do scientists model mental disorders in animals? - May 23, 2023
Acid-base extraction is a chemical process used to separate compounds based on their acid-base properties. The technique is commonly used in organic chemistry to isolate and purify organic compounds from mixtures.
The process involves adding a mixture of compounds to an organic solvent, such as ether or dichloromethane, and then adding an aqueous solution of an acid or a base. The acid or base reacts with the compounds in the mixture, changing their solubility and allowing them to be selectively extracted into either the organic or aqueous layer.
For example, if a mixture contains a weakly acidic compound and a weakly basic compound, adding an acid to the mixture will protonate the basic compound, making it more soluble in the organic layer, while the acidic compound will remain in the aqueous layer. Conversely, adding a base to the mixture will deprotonate the acidic compound, making it more soluble in the organic layer, while the basic compound will remain in the aqueous layer.
The layers can then be separated and the desired compound can be isolated and purified by further processes such as drying, filtration or evaporation. Acid-base extraction is a versatile technique that can be used for a wide range of organic compounds and is widely used in research, analytical chemistry, and industrial processes.
A potential alteration of the extractions already talked about in this chapter is to do a chemical reaction in the separatory funnel to alter the polarity and consequently the separation of a compound in the aqueous and organic layers. A popular technique is to do an acid-base reaction, which can convert some compounds from neutral to ionic forms (or the other way around).
Example
For instance, if a blend of benzoic acid and cyclohexane is blended in a natural solvent, such as ethyl acetate, in a separatory funnel. To divide the components, a water wash may be attempted to take out benzoic acid, but benzoic acid is not notably soluble in water due to its nonpolar aromatic ring, and only minor quantities would be taken out into the aqueous layer.
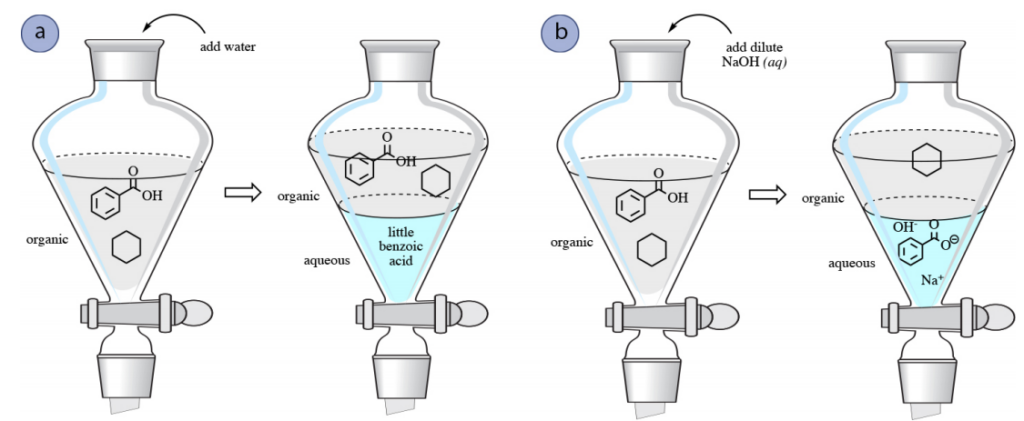
The partition of a blend of benzoic acid and cyclohexane may be accomplished by a rinse with a base like NaOH. Because of its acidic nature, benzoic acid can go through a response with NaOH like so, leading to the carboxylate sodium benzoate.
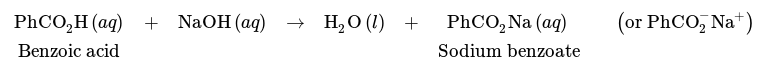
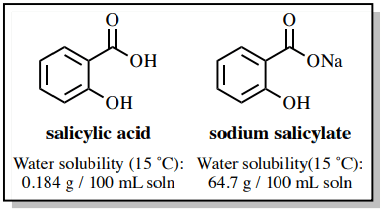
The solubility properties of carboxylic acids are significantly different from their comparable carboxylate salts. Sodium salicylate is approximately 350 times more soluble in water than salicylic acid because of its ionic character (Fig.2), and it is relatively insoluble in organic solvents such as diethyl ether.
Consequently, a wash with sodium hydroxide would transform benzoic acid into its ionic carboxylate state, which would then be more soluble in the aqueous layer, permitting the sodium benzoate to be extracted into the aqueous layer. Cyclohexane would stay in the organic layer as it has no attraction to the aqueous phase, nor can it react with NaOH in any fashion. In this way, a combination of benzoic acid and cyclohexane can be separated (Fig.1 b). The aqueous layer could then be acidified with hydrochloric acid (HCl(aq)) if desired to convert the benzoic acid back to its impartial form.
Sodium Bicarbonate Washes
An acid-base extraction can be employed to extract carboxylic acids from the organic layer into the aqueous layer. As mentioned in the preceding section, NaOH can be utilized to transform a carboxylic acid into its more water-soluble ionic carboxylate form. But if the mixture contains a wanted substance that can react with NaOH, a milder base such as sodium bicarbonate should be applied. A comparable reaction occurs:
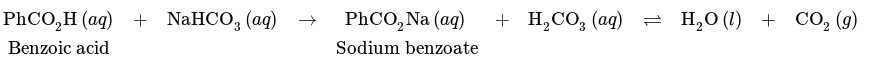
Substituting the base NaHCO3 for NaOH yields carbonic acid (H2CO3) as a byproduct. When shaking an acidic solution with sodium bicarbonate in a separating funnel, one should be careful to mix calmly and frequently release pressure from the gas.
One example of a process that routinely utilizes sodium bicarbonate wash in the post-reaction is Fischer Esterification. Illustrating this, benzoic acid refluxed in ethanol with concentrated sulfuric acid to give ethyl benzoate (Fig.3 a and b). A TLC plate of the reaction mixture at 1 hour of reflux showed remaining unreacted carboxylic acid (Fig.3 c), which is not uncommon due to the energetics of the reaction.
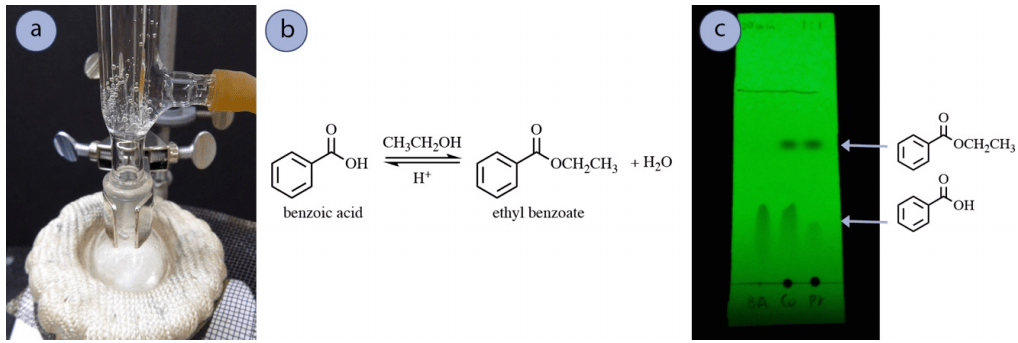
The leftover carboxylic acid can be extracted from the desired ester product using an acid-base separation in a separatory funnel. A cleaning with sodium bicarbonate converts benzoic acid into its more water-soluble sodium benzoate structure, taking it into the aqueous layer (Fig. 4). Furthermore, the sodium bicarbonate cancels out the catalytic acid in this reaction.
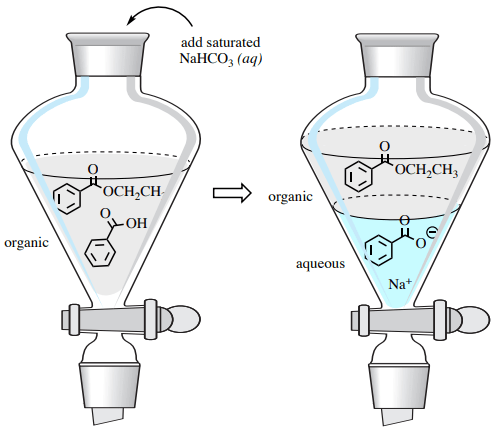
Sodium bicarbonate is preferred over NaOH in this process, as it is a much weaker base and using NaOH could lead to the hydrolysis of the ester product.
Mixtures of Acids and Bases
The acid-base properties of compounds can be taken advantage of to segregate certain constituents from mixtures. This practice can be extended to other scenarios.
Extracting Bases
Basics, such as amines, can be taken out of organic solutions by agitating them with acidic solutions to transform them into more water-soluble salts, allowing them to be extracted from the organic layer into an aqueous layer.
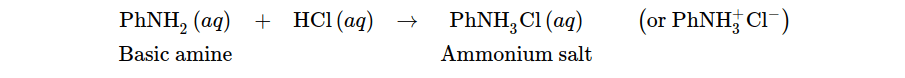
Extracting Carboxylic Acids vs. Phenols
As previously discussed, carboxylic acids can be transferred from an organic layer to an aqueous layer by shaking them with alkaline solutions, which transforms them into their more water-soluble salts.
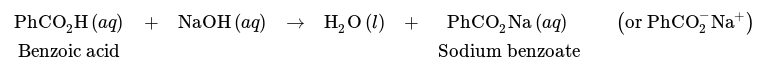
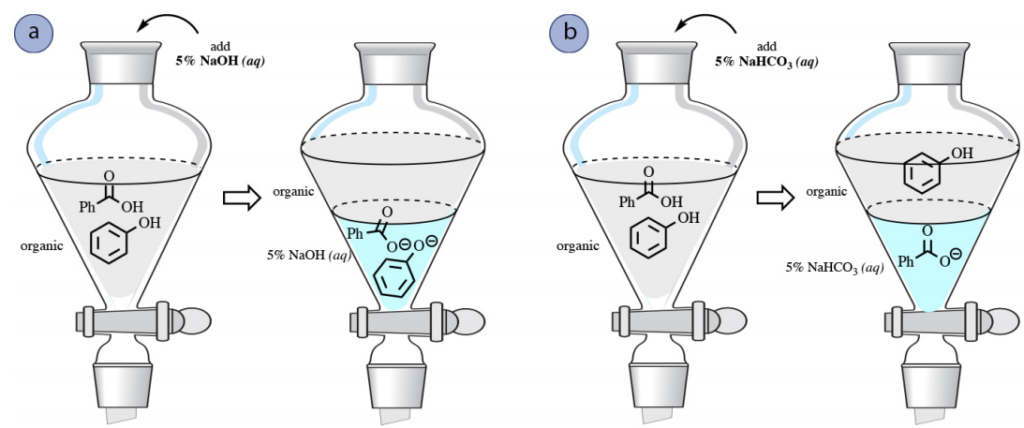
A comparable reaction takes place with phenols (PhOH), and they can be moved into an aqueous NaOH layer (Fig.5 a).
Nevertheless, phenols are notably less acidic than carboxylic acids, and are not acidic enough to interact totally with NaHCO3, a weaker base. As a result, a solution of bicarbonate can be employed to separate mixtures of phenols and carboxylic acids (Fig.5 b).
Extracting Acid, Base, and Neutral Compounds
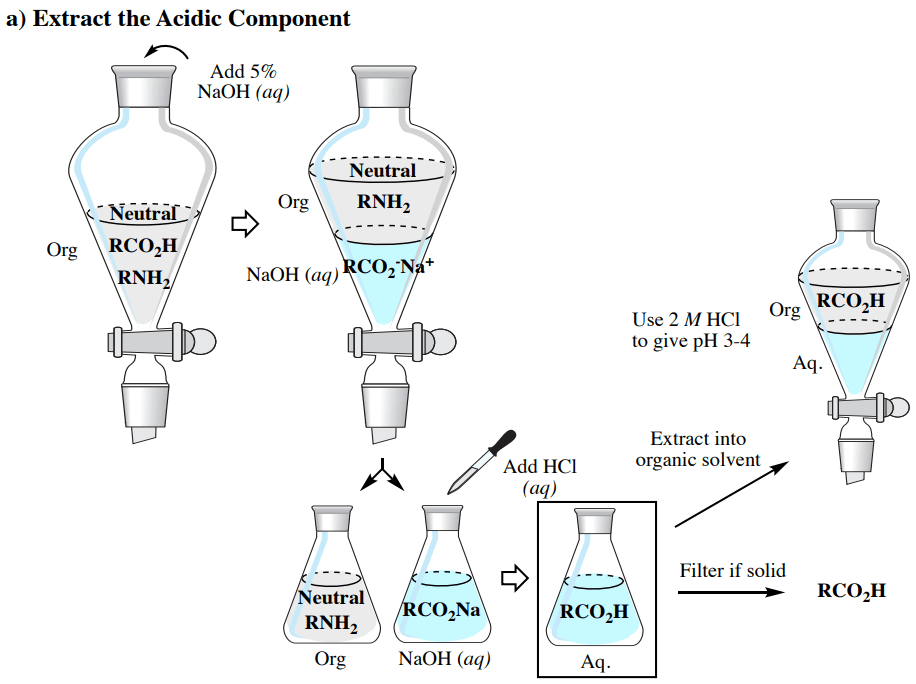
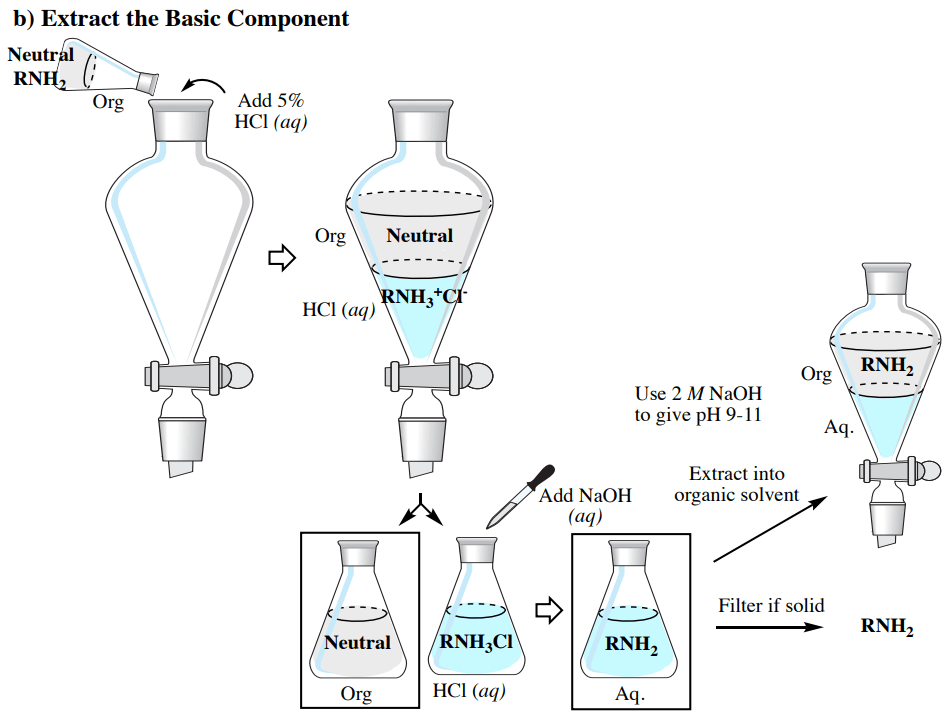
The acid-base characteristics that were talked about before enable a blend including acidic (e.g. RCO2H), basic (e.g. RNH2), and neutral elements to be purified through a progression of extractions, as summarized in Fig.6 (which utilizes an organic solvent less heavy than water).
It is assumed that scholars making this kind of experiment have previous experience with single and multiple extractions. In this segment are described contrasts between normal extraction systems and the process as summarized in Fig. 6.
1. Separating the Acidic component:
a) When the acidic component is in the aqueous layer in an Erlenmeyer flask, it can be converted back to the neutral component through the addition of 2M HCl(aq) until the mixture gives a pH of 3-4 (as measured by pH paper). If huge amounts of acid are present such that acidification would need an excessive amount of 2M HCl(aq), concentrated HCl(aq) may be instead added drop by drop. Lower concentrations of Hcl(aq) are less dangerous, but increasing the capacity of the aqueous layer by a large sum would influence the effectiveness of succeeding extractions and filtering steps.
b) Subsequent to acidification, two paths may be taken, depending on whether the acidic component is solid or liquid.
•If a solid forms upon acidification of the ionic salt, it can be gathered through suction filtration. This approach should just be utilized if large amounts of large-sized crystals are seen. If fine crystals form (which are normal), they will obstruct the filter paper and interfere with satisfactory drainage. If only a tiny amount of solid is seen related to the hypothetical amount, it is probable the compound is very water-soluble, and filtration would lead to low recovery.
•If no solid forms upon acidification (or if fine crystals or small quantity of solid forms), extract the acidic component back into an organic solvent (×3). As a general principle, use one-third as much solvent for the extractions as the original layer (e.g. if utilizing 100ml aqueous solution, extract with 33ml organic solvent each time). Be sure to first cool the aqueous solution in an ice bath before extraction if the acidification caused recognizable heat. Follow up with a brine wash (×1) if utilizing diethyl ether or ethyl acetate, dry with a drying agent, and remove the solvent via rotary evaporator to leave the pure acidic component.
2. Isolating the Basic component:
Employ a similar process as the extraction of the acidic component, apart from basify the solution using 2M NaOH(aq) until it gives a pH of 9-10 as determined by pH paper.
3. Extracting the Neutral component:
The neutral component will be the “leftover” compound in the organic layer. To isolate, wash with brine (×1) if utilizing diethyl ether or ethyl acetate, dry with a drying agent, and remove the solvent via rotary evaporator to leave the pure neutral component.
Conclusion
In conclusion, acid-base extraction is a powerful technique used in organic chemistry to separate and isolate compounds based on their acid-base properties. By adding an acid or a base to a mixture of compounds in an organic solvent, the solubility of the compounds can be selectively altered, allowing them to be partitioned into either the organic or aqueous layer. This technique is widely used in research, analytical chemistry, and industrial processes to purify and isolate compounds from complex mixtures. The versatility and effectiveness of acid-base extraction make it an essential tool in organic chemistry and a valuable skill for any chemist to master.
