- The process of creating a drug - May 23, 2023
- How do scientists model mental disorders in animals? - May 23, 2023
In this topic you can read about of boiling point definition method, which can be utilized for liquid reagents analysis. When you need to be sure in a liquid reagent, you can define its boiling point and compare your analysis result with a literature data of checked compound.
The boiling point of any compound is the temperature where the liquid-gas phase change happens. In more technical words, it is when a liquid’s vapor pressure equals its applied pressure (usually the atmospheric pressure). Boiling points are very sensitive to changes in applied pressure, so all boiling points should be reported with the measured pressure. A compound’s “normal boiling point” refers to its boiling point at a pressure of 760 mm Hg (1 atm).
A compound’s boiling point is a physical constant just like melting point, and hence can be utilized to help the identification of a substance. Nonetheless, boiling points are not generally used as a gauge of purity unlike melting points. Impure liquids boil over a range of temperatures (similar to how melting points have breadth), but the temperature interval does not correlate linearly with purity. Consequently, a compound’s boiling point measurement is mainly utilized to help its identification.
An experimental boiling point is generally compared to the literature boiling point, which is typically recorded under a pressure of 1 atmosphere. If a boiling point is identified at a pressure different from 1 atm, the pressure should be adjusted. A basic rule is that for pressures within 10% of one atmosphere, a 10 mm Hg decrease in pressure will cause a decrease of 0.3-0.5 °C in the boiling point. Another rule is that for every halving of pressure, the boiling point will drop by about 10 °C. There are various ways to determine a sample’s boiling point, such as distillation, reflux, and using a Thiele tube. The simplest technique utilizes a Thiele tube and has the benefit of using less than 0.5mL of material.
Distillation Method
Rather than a distillation, there are other more straightforward ways to measure a compound’s boiling point, and it is suggested to investigate those alternatives (e.g. Thiele tube) if that is the only aim. But if there is limited material, or a purification is to be carried out anyway, a distillation can be used to determine a compound’s boiling point.
In the majority of cases, a straightforward distillation should do the trick (Fig. 1), and at least 5 mL of sample should be put in the distilling flask along with a few boiling stones or a stir bar. As the major part of the material distills, the highest temperature noted on the thermometer equals the boiling point. A common source of mistake with this technique is recording a temperature that is too low, before hot vapors completely engulf the thermometer bulb. Be sure to check the thermometer at regular intervals, particularly when the distillation is ongoing. Record the barometric pressure as well as the boiling point.

Reflux Method
A reflux system can also be employed to calculate a substance’s boiling point. Reflux is a procedure wherein a liquid is boiling vigorously and vaporizing, with the evaporated liquid coming back to the original container. It is similar to a distillation system, with the major discrepancy being the upright placement of the condenser.

In the event that materials are accessible, the most ideal reflux arrangement for this application is shown in Fig. 2b and incorporates a microscale condenser and advanced thermometer. The arrangement requires 5 mL of fluid, and a couple of bubbling stones or stir bar. The condenser is appended to the round bottomed container, with the lower water hose associated with the water fixture and the upper water hose depleting to the sink. It is significant to check that the joint associating the container and condenser is safely fixed. The liquid is warmed to a bubble on a sand shower, and the thermometer is put low into the apparatus (Fig. 2c) such that the lower inch is between the bubbling liquid and the base of the condenser. In this situation, the thermometer can precisely gauge the hot mists and the temperature will settle at the compound’s bubbling point.
Record the barometric weight alongside the bubbling point.
Despite the fact that it may appear to be prudent to submerge the thermometer straightforwardly into the bubbling liquid, it is conceivable the liquid may be superheated, or hotter than its bubbling point. After deciding the bubbling point, the container ought to be raised from the sand shower (Fig. 2d) to chill off, and condenser kept running until the container is just warm to the touch. At this point the arrangement can be dismantled.
In the event that a microscale condenser isn’t accessible, an elective reflux technique can likewise be utilized as shown in Fig. 3. Around 5 mL of example is put in a medium test tube (18 x 150 mm) with thermometer attached inside so it doesn’t contact the sides of the glass. The apparatus is cautiously warmed on a sand bath such that reflux happens controllably and mists don’t get away from the tube. The temperature during reflux will in the end stabilize (this takes some time), and the most elevated temperature noted corresponds to the compound’s bubbling point. The bubbling focuses estimated with this technique may have huge blunder if the bubbling point is exceptionally low or high (<70 °C or >150 °C) as low boiling compounds bubble away excessively effectively and high boiling compounds have a tendency to cool excessively effectively.

Thiele Tube Method
Thiele Tube Theory
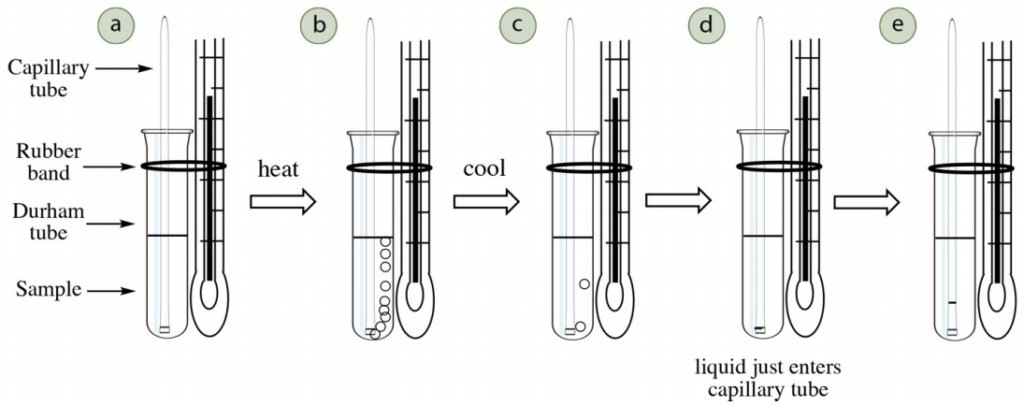
The Thiele tube approach is one of the easiest ways to find out a substance’s boiling point, and it has the bonus of only requiring a tiny amount of material (less than 0.5 mL of sample). The sample is placed in a minuscule tube with an inverted capillary tube. The system is linked to a thermometer (Fig. 5) and heated in a Thiele tube (Fig. 4) to a temperature slightly higher than the compound’s boiling point (which can be seen by an ongoing flow of bubbles emanating from the capillary tube). The tube is then allowed to cool, and the temperature when liquid is drawn into the capillary tube is the compound’s boiling point.

This technique makes use of the concept of boiling point: the temperature at which the vapor pressure of the compound is equal to the atmospheric pressure. The inverted capillary tube acts as a repository to contain the substance’s fumes. As the device is heated, the air that was originally confined in the capillary tube will expand and cause bubbles to come out of the tube (Fig. 5 b). As the heat is further increased, the compound’s vapors will eventually displace all of the contained air, thus necessitating the application of heat until a constant stream of bubbles is observed.
When the equipment is cooled, at some point the pressure inside the capillary tube (caused only by the compound’s vapors) will equal the atmospheric pressure, which is when the bubbles will start to slow down and liquid will be drawn into the tube. The temperature at which this begins is the boiling point of the compound (Fig. 5 d).
Thiele tube procedure

• Secure a Thiele tube to a laboratory stand inside the fume hood (as shown in Fig. 6a). Generally, this tube is filled with transparent mineral oil, however it may be darkened due to oxidation or contamination. If the oil is heavily discoloured, it should be changed. The oil should be poured to a minimum of 1cm above the highest arm (Fig. 6a indicates the ideal level). If the oil level is lower, it won’t be able to move as it should (Fig. 7c).
• Put a thermometer into a one-holed stopper with a slit along one side. Affix a small glass container (Durham tube or 6 x 50mm culture tube) to the thermometer with a small rubber band (Fig. 6b). The bottom of the container should be even with the bottom of the thermometer.
• Fill the vial around halfway with the sample, which needs between 0.25-0.5 mL of sample (Fig. 6c).
• Insert a capillary tube into the sample (the same type as those used for melting points), open end facing downwards and sealed end facing up (Fig. 6d).

Fig. 7: a) Placing the combination into the Thiele tube, b) The elastic band is on top of the oil, c) Warming, d) Active effervescence of the specimen.
• Insert the rubber stopper and thermometer arrangement into the Thiele tube, changing the level so that the sample is situated in the middle (if possible) within the tube (Fig. 7a). The rubber band should be positioned above the top of the mineral oil (Fig. 7b), remembering that the oil could possibly swell somewhat while being heated. The thermometer should not make contact with the edges of the glass, and if it does it should be attached in such a manner that it no longer touches.
• Gently heat the oil on the side arm of the Thiele tube with a microburner if obtainable, or Bunsen burner utilizing a back and forth motion (Fig. 7c). As the oil warms up and becomes less dense, it will ascend and flow up the triangular area of the tube. The cooler, heavier oil will fall, thereby forming a flow as displayed in Fig. 7c). This technique is a great way to indirectly and slowly heat the sample.
• Although bubbles should not be visible in the Thiele tube while it is warming, they usually are seen if the tube was used previously for boiling point determinations. In this technique, the rubber band occasionally snaps causing the sample to fall into the oil and contaminate it. If the oil is not later changed, the sample may boil when heated in the tube. It is permissible to keep heating a Thiele tube if bubbles are visible.
• Studies of this approach have found that it is best to heat the oil gently and in a continuous manner, as ceasing and starting have caused the results to be inadequate.
• Carry on heating until a lively stream of bubbles erupts from the tip of the capillary tube (Fig. 7d), such that separate bubbles can barely be identified. The purpose of this step is to eliminate the air initially present in the capillary tube and substitute it with the sample’s vapor. Do not heat so energetically that the entire sample vaporizes. when bubbles are energetically bursting from the capillary tube, the vapor pressure inside the tube is greater than the atmospheric pressure (the oil is at a higher temperature than the boiling point).
• Switch off the burner and permit the apparatus to cool down. The bubbles will slow and ultimately stop. At some point the vapor pressure inside the capillary tube will be equivalent to the atmospheric pressure and liquid will be drawn into the tube. The boiling point should be documented as the temperature when liquid just begins to enter the capillary tube (Fig. 8b).

• Make a note of the atmospheric pressure together with the boiling point.

