- The process of creating a drug - May 23, 2023
- How do scientists model mental disorders in animals? - May 23, 2023
Introduction
Organic compounds make ~ 90% of all compounds on the Earth. That is quite a lot, isn’t it? In order to study such a huge number of compounds, it is necessary to classify them into groups. This topic about the Classification of Organic Compounds as well as the general groups into which organic compounds are divided.
Rules and features of classification
The classification is based on the structure of organic compounds. The structure description basis is the structural formula. Atoms of elements are denoted by Latin symbols, as they are indicated in the periodic table of chemical elements (Mendeleev’s table). Hydrogen and electron-deficient bonds are indicated by a dotted line, ionic bonds are indicated by a designation of particles charges that make up molecules. Since the vast majority of organic molecules include hydrogen, it is usually not indicated when when illustrating a structure. Thus, if an insufficient valence is shown in a structure of one of the atoms, it means that one or more hydrogen atoms are located near this atom. Atoms can form cyclic and aromatic systems.
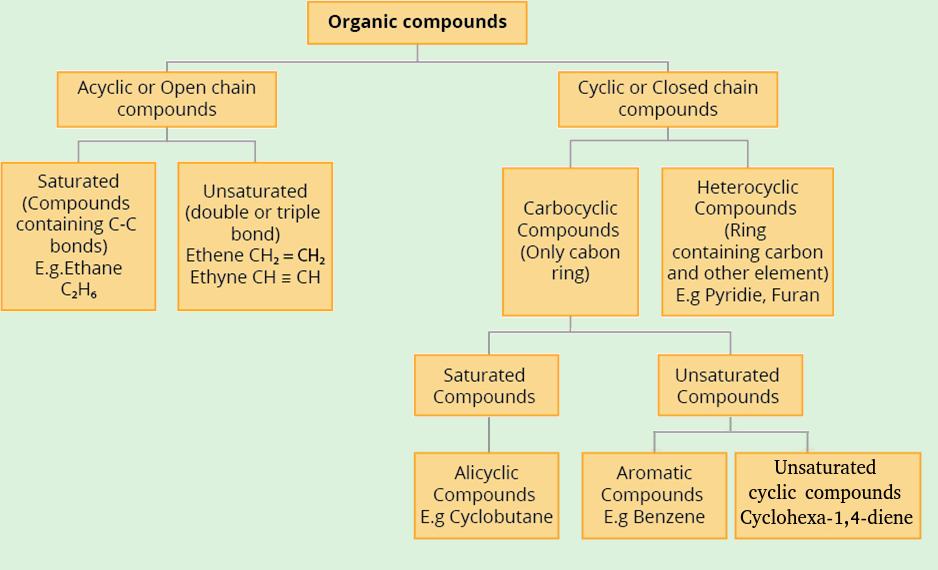
Organic compounds are classified into two types based on the skeleton of the carbon chain:
- Acyclic or Open-chain compounds
- Cyclic or Closed-chain compounds
1. The firs one is Acyclic or Open-chain compounds:
The compounds in which the carbon atoms form a chain-linked in a linear pattern or branched are called as acyclic or open-chain compounds.
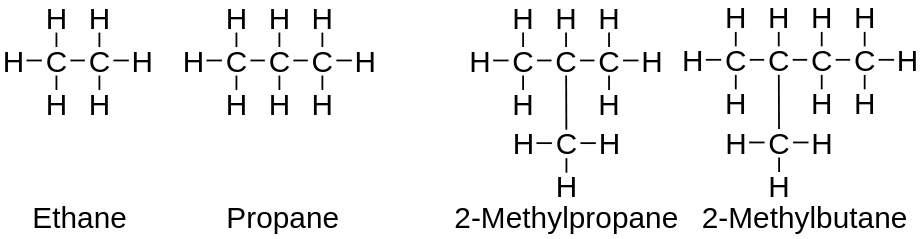
Open-chain and branched chain hydrocarboncompounds
Open-chain compounds are further classified as:
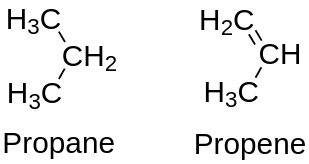
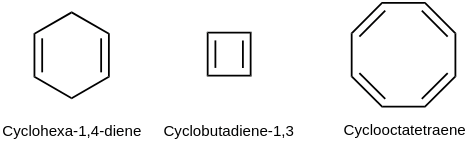
a) Saturated compounds: a compound is saturated if single bonds connect all of the carbon atoms in the chain. In this case, all carbon valencies are satisfied by single bonds.
b) Unsaturated compounds: If there are double or triple bonds between carbon atoms, a compound is unsaturated. In this case, all the valencies of carbon are satisfied by multiple bonds.
2. Cyclic or closed-chain compounds:
Cyclic compounds are organic compounds which are contain closed or cyclic carbon atoms chain.
The chains contains carbon as well as other atoms in heterocyclic compounds such as oxygen, nitrogen, sulphur, phosphorus and so on.
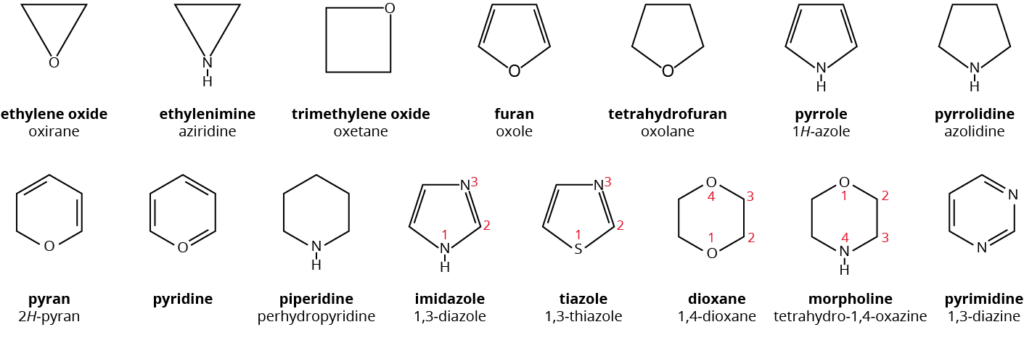
Heterocyclic compounds (saturated and unsaturated)
Carbocyclic (Homocyclic) compounds contain only carbon atoms in a chain.
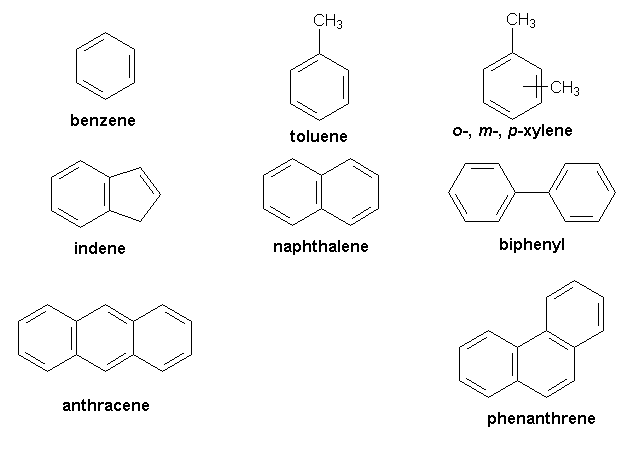
Homocyclic compounds
Carbocyclic compounds are further classified as:
- Saturated ciclic compounds
- a) Alicyclic compounds
- Unsaturated ciclic compound
- b) Aromatic compounds
- c) Non-aromatic compounds
Alicyclic compounds have one or more carbocyclic rings which can be either saturated or unsaturated.
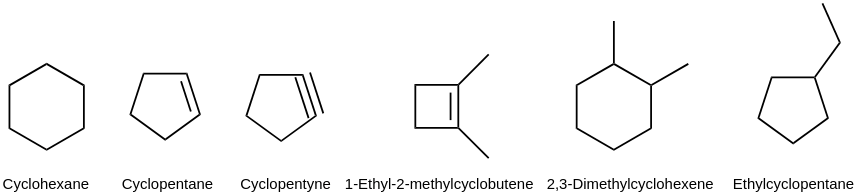
a) Alicyclic saturated and unsaturated compounds
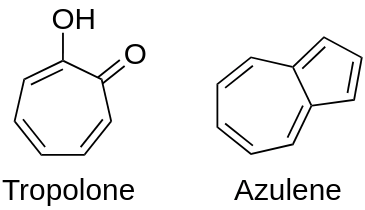
Aromatic compounds have one or more benzene rings. Benzene ring is carbon chain ring containing alternate double bonds between 6 carbon atoms. Usually, these compounds have some fragrant odour and hence, named as aromatic (Greek word aroma meaning sweet smell).
Functional groups
The functional groups conception is pivotal in organic chemistry, both as a means to a structure classification and for properties predicting. A functional group is a molecular block, and the reactivity of any functional group is assumed, within limits, to be the same in a variety of molecules. Functional groups can have a critical influence on the chemical and physical properties of organic compounds. Molecules are classified based on their functional groups.
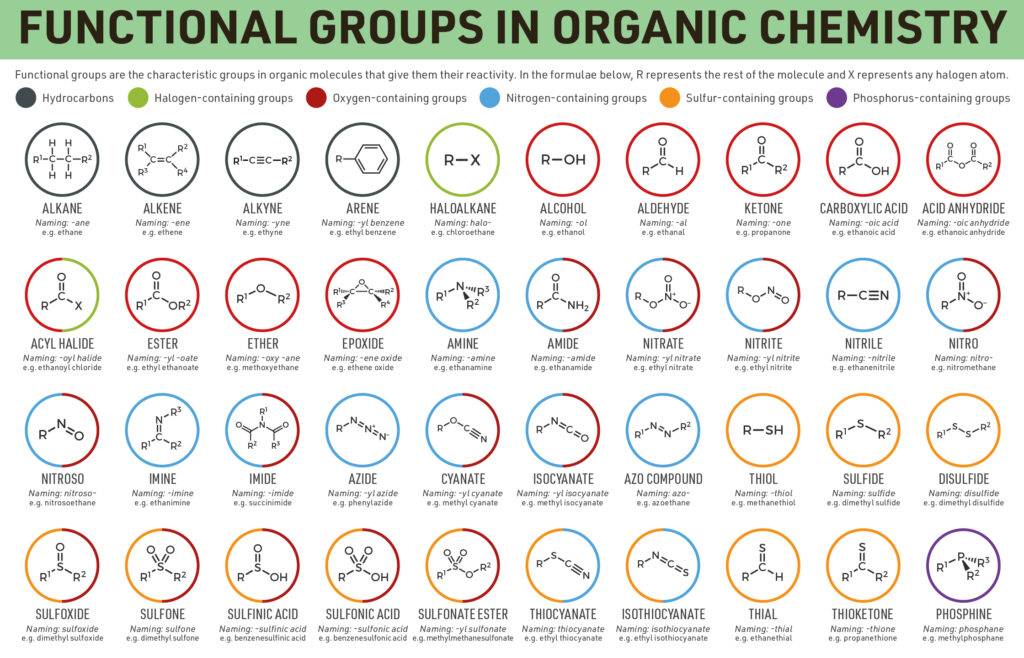
Main classes of organic compounds
1) Hydrocarbons are chemical compounds which consist only carbon and hydrogen atoms. Hydrocarbons are divided into a acyclic and carbocyclic types in accordance with a carbon skeleton structure topology. Hydrocarbons are divided into a saturated (alkanes or saturated), which not containing multiple bonds in their structure, and unsaturated, which containing at least one double and/or triple bond (alkenes, alkynes, dienes), in accordance with a carbon-carbon bonds quantity. Also, cyclic hydrocarbons are divided into alicyclic (with an open chain) and cycloalkanes (limited with a closed chain), aromatic hydrocarbons (unsaturated, containing a cycle).
2) Compounds with heteroatoms in functional groups are compounds, which a carbon radical R is bonded to a functional group. By the nature of the functional groups are divided into:
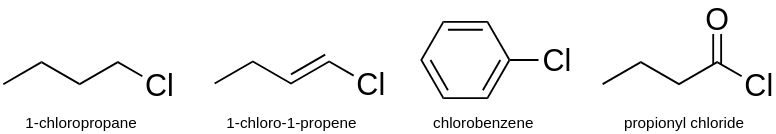
a) Halogenated compounds
b) Alcohols, phenols. Alcohols are organic compounds, which are containe one or more hydroxyl groups (hydroxyl, −OH) directly bounded to a saturated (in sp³ hybridization state) carbon atom. Alcohols can be considered as derivatives of water (H-O-H), which have one replaced hydrogen atom by an organic functional group: R-O-H. In the IUPAC nomenclature for compounds, which have bonded to an unsaturated (sp2 hybridized) carbon atom hydroxyl group, the names “enols” (hydroxyl is bonded to a vinyl C=C bond) and “phenols” (hydroxyl is bonded to benzene or another aromatic ring).
c) Ethers are organic substances which have the formula R-O-R1, where R and R1 are hydrocarbon radicals. It have to be taken into account that such a group can be parts of other functional groups of compounds that are not simple ethers (for example, oxygen-containing organic compounds).
d) Esters are derivatives of oxo acids (both carboxylic and mineral), which are formally products of hydrogen atoms substitution of acid function hydroxyls -OH for a hydrocarbon radical (aliphatic, alkenyl, aromatic or heteroaromatic); they are also considered as acyl derivatives of alcohols. In the IUPAC nomenclature, esters also include acyl derivatives of chalcogenide analogs of alcohols (thiols, selenols, and tellurols).
e) Compounds with a carbonyl group
e-1) Aldehydes is a class of organic compounds which contains a carbonyl group (-C=O) with one alkyl or aryl substituent.
e-2) Ketones are organic compounds which have bonded carbonyl group with two hydrocarbon radicals. General formula of ketones: R1-CO-R2. The presence in ketones of precisely two carbon atoms directly bonded to the carbonyl group distinguishes them from carboxylic acids and their derivatives as well as aldehydes.
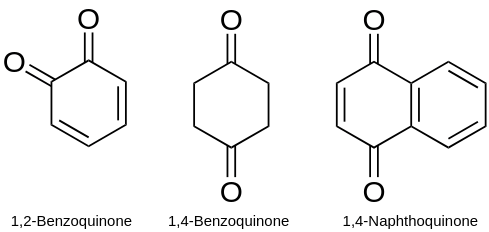
e-3) Quinones are a class of organic compounds that are formally “derived from aromatic compounds [such as benzene or naphthalene] by conversion of an even number of –CH= groups into –C(=O)– groups with any necessary rearrangement of double bonds, resulting in “a fully conjugated cyclic dione structure”. The archetypical member of the class is 1,4-benzoquinone or cyclohexadienedione, often called simply “quinone” (thus the name of the class). Other important examples are 1,2-benzoquinone (ortho-quinone), 1,4-naphthoquinone and 9,10-anthraquinone.
3) Heterocyclic compounds contain heteroatoms in the ring composition. They differ in atoms number in the cycle, type of heteroatom(s), heteroatoms number in the cycle.
4) Organic origin compounds are, as a rule, compounds of a very complex structure, often belong to several classes of organic substances at once, often polymers. In accordance with difficulty of their classification, they are isolated in a separate class of substances.
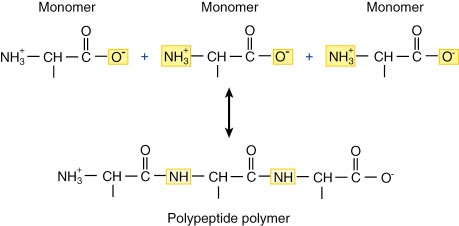
5) Polymers are substances with a very big molecular weight, which consist of periodically repeated fragments (monomer units).