- The process of creating a drug - May 23, 2023
- How do scientists model mental disorders in animals? - May 23, 2023
Decanting
When there is a need to separate a solid-liquid mixture, it can be done by a pouring off the liquid while separating the solid away. This process is known decanting, and it is the simplest separation method. Decanting is commonly used to remove hydrated sodium sulfate (Na2SO4) from an organic solution. Na2SO4 Often clings to the glassware (Fig.1 a), letting the liquid to be poured off (Fig.1 b). If liquid is to be poured into a small container, a funnel could be used or liquid poured down a glass stirring rod to head the flow (Fig.1 c). Unfortunately, there are numerous mixtures which do not decant well.

a) Na2SO4 sticking to the beaker, b) Decanting a solid-liquid mixture, c) Using a glass stirring rod while decanting.
Decantation is a procedure which separates mixture components based on differences in density. You can use decantation in everyday life with wine or spirits, but it’s also a useful technique in chemistry for a solid and liquid separating or two immiscible liquids isolating.
Decantation is easy, but there is a disadvantage, which doesn’t allow to carry out perfect separation of mixture components. A small amount of one mixture component gets lost during collecting the other component, or else the collection is carried out too far and the collection gets contaminated with the second component.
How decantation works
Decantation assumes two steps:
• Precipitation: precipitation uses gravity or a centrifuge to separate mixture components based on their density.
• Decantation: decantation is pouring or siphoning off the top component of a mixture or draining the bottom component.
A solid component is called “sediment” (or “pellet” when centrifugation is used). The liquid component which is collected is called “decant.”
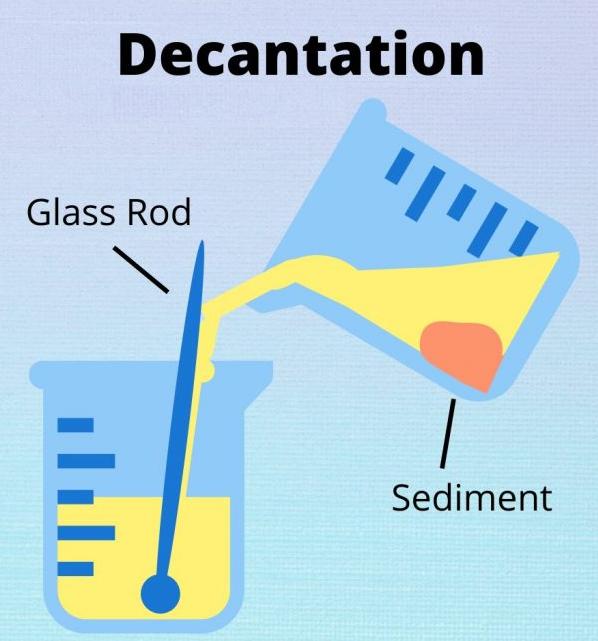
The basic decantation principle is that heavier (denser) substances sink, whereas lighter (less dense) substances rise. In its simplest form, decantation uses gravity to separate a solid and liquid or two immiscible liquids. The lighter compound is poured or siphoned off the top of the mixture. As another option, a separatory funnel drains the heavier component from the bottom layer.
Small volumes are decanted with help of test tubes inclined at 45° in a test tube rack. This angle lets heavier particles glide down the tube, while lighter particles rise to the top. The angle also makes pouring the lighter compound convenient. Pouring off the liquid is easier in case it’s poured along a stirring rod. The decantation process is slower on condition that the test tubes are kept vertical because the heavier component can form a plug and block lighter particles from floating.
Centrifugation speeds up decantation by using centrifugal and centripetal force. Inherently, the artificial gravity separates mixture components faster. Centrifugation thickens solid components into a pellet. Pouring the liquid away from the pellet get in less loss than in simple decantation. A separatory funnel decants mixtures with immiscible liquid components. One component floats on top of the other one. The funnel drains the bottom layer component.
Filtering methods
There are several methods applied to separate a mixture containing a solid and liquid phases. If the solid settles well, the liquid can sometimes be poured off (decanted). If the solid has very small sized particles or forms a cloudy mixture, the mixture can be centrifuged or passed through a filter pipette (on the microscale, < 5 ml) in special cases.
The most common methods of solid-liquid separation in the organic laboratory are gravity and suction filtration. Gravity filtration belongs to pouring a solid-liquid mixture through a funnel with a filter paper, allowing the liquid to seep through while trapping the solid on the filter (Fig.1 a). Suction filtration is a similar process with the difference being the use of a vacuum under the funnel in order to pull liquid through the filter paper by suction (Fig.1 b).

a) Gravity filtration, b) Suction filtration.
Gravity and suction filtration have advantages and disadvantages. Whether the solid or filtrate have to be retained, it which has passed through a filter paper (as indicated in Fig.1 a). Gravity filtration is typically used when the filtrate is kept, whereas suction filtration is used when the solid is kept. Gravity filtration is preferred when the filtrate is retained as suction has the potential of pulling small solid particles through the filter paper pores, potentially producing a filtrate polluted with the solid substance. Suction filtration is preferred when the solid is retained as gravity filtration is much less efficient at purging residual liquid from the solid on the filter paper.Gravity filtration
When there is a goal to divide a solid-liquid mixture, it is usual that the particles are so fine that they swirl and disperse when the flask is tilted. These mixtures cannot be decanted, and an another method is gravity filtration. Gravity filtration is mainly used when the filtrate (liquid which has passed through the filter paper) will be retained, whereas the solid on the filter paper will be discarded.
A ordinary use for gravity filtration is for separating anhydrous magnesium sulfate (MgSO4) from an organic solution that it has dried (Fig. b). Anhydrous magnesium sulfate is powdery, and with swirling in an organic solvent creates a fine dispersal of particles similar to a snow ball.

a) An organic solution dried with anhydrous MgSO4, b) Gravity filtration of this solution.
To gravity filter a mixture, the mixture is poured through a quadrant-folded filter paper (Fig. 4) or fluted filter paper in a funnel and the liquid is allowed to filter using only the force of gravity (Fig. 3 c). It is best to pour as if attempting to decant, meaning to keep the solid settled in the flask for as long as possible. When solid begins to pour onto the filter paper, it has the possibility of clogging the filter paper pores or slowing filtration. After finished pouring, wash the solid on the filter paper (and in the flask) with a few portions of fresh solvent to remove residual compound adhering to the solid.
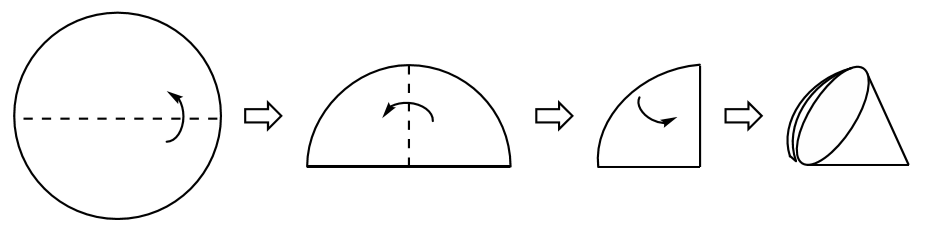
Making a quadrant-folded filter paper. The dotted lines show locations to crease and fold the filter paper. The arrows head the direction of folding.
Transferring liquids
Pouring Liquids
If transferring liquids with volumes bigger than 5 ml, they can be poured directly into vessels. Graduated cylinders and beakers have an deepening in their edge, so they can be poured controllably as long as the two pieces of glass touch one another (Fig.5 a). If pouring from an Erlenmeyer flask, or transferring a liquid into a vessel containing a narrow neck (like Erlenmeyer flask), a funnel have to be used. Funnels can be securely held with a ring clamp (Fig.5 b), or held with one hand while pouring with the other (Fig.5 c).

a) Pouring liquid, b) Pouring into a funnel held with ring clamp, c) Pouring into a funnel held by hand.
Comments about measurements
In order to define a relevant yield of a chemical reaction, it is significant to have precise measurements on the limited reactant. It is less important to be precise when manipulating a reagent that is in excess, especially if the reagent is in several times excess.
A liquid portion measured by a graduated cylinder always sticks to the glassware after pouring, meaning that the true volume dispensed is never equivalent to the markings on the cylinder. Hence, graduated cylinders can be applied for dispensing solvents or liquids that are in excess, whereas more precise methods (e.g. mass, calibrated pipettes or syringes) should be used when dispensing or measuring the limited reactant. A graduated cylinder can be used to measure out a limiting reactant if its mass will be determined to find the precise quantity actually dispensed.

a) Cork ring on an analytical balance, b) Beaker on a pan balance.
During the mass determination of a vessel on a scale, it’s worth to not include the mass of a cork ring (Fig.6 a) or other support (e.g. the beaker in Fig.6 b). A cork ring may get wet, have reagents poured on it, or have pieces of cork drop out, heading to changes in mass which cannot be counted for. Beakers, which is used to support flasks, can get mixed up, every beaker doesn’t have the same mass. It is also best to transport vessels containing chemicals to the scale in sealed flasks, so that to minimize evaporation and prevent a possible spilling during moving.
Using Pasteur pipettes
Pasteur pipettes (or pipets) are the most ordinarily used tool for transferring small liquids volumes (< 5 ml) from one vessel to another on.
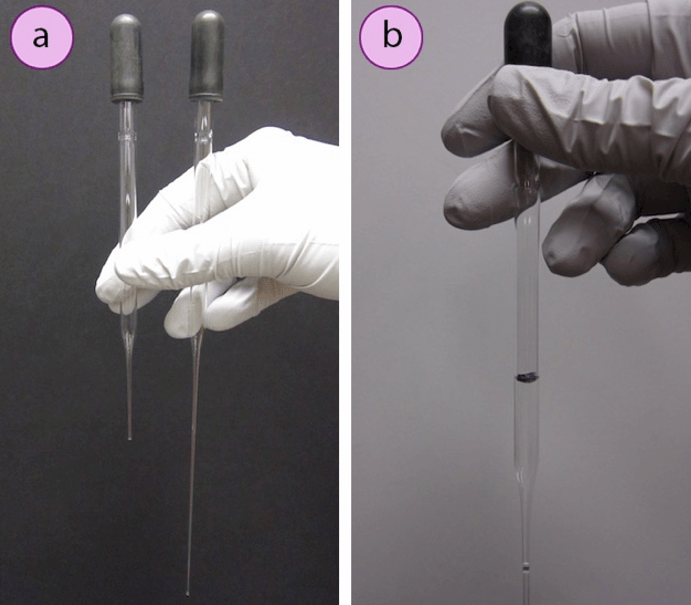
a) Short and long pipettes; b) 1ml marked on a pipette with a permanent marker.
There are Pasteur pipettes in two sizes (Fig.7 a): short (5.75″) and long (9″). Each pipette can take about 1.5 ml of liquid, although the volume delivered is dependent on the size of the dropper bulb. The general guideline that “1 mL is equivalent to 20 drops” does not always hold for Pasteur pipettes, and may be incompatible between different pipettes. The drop ratio for a certain pipette and solution can be defined by counting drops until 1 mL is accrued in a graduated cylinder. As another option, a pipette can be roughly calibrated by withdrawing 1 mL of liquid from a graduated cylinder and marking the volume line with a marker (Fig.7 b).

a and b) Creating suction with a Pasteur pipette, c) Delivering liquid from a Pasteur pipette, d) Incorrect delivering of reagent (liquid should not touch the sides of the glass).
To utilize a pipette, attach a dropper bulb and place the pipette tip into a liquid. Squeeze then release the bulb to generate suction, which will cause liquid to withdraw into the pipette ( Fig.8 a and b). Holding the pipette vertical, bring it to the flask where it is to be transferred, and place the pipette tip below the flask neck but not touching the sides before squizing the bulb to deliver the liquid into the flask (Fig.7 c). The bulb can be squeezed a couple times afterward to “blow out” residual liquid from the pipette.
In case the receiving flask has a ground glass joint, the pipette nozzle should be below the joint while delivering so that liquid doesn’t splash onto the joint, which can cause pieces to freeze together when connected. If the pipette is to be reused (for example is the designated pipette for a reagent bottle), the pipette have to be held so it does not touch the glassware, where it can become contaminated by other reagents in the flask (Fig.8 d).
Calibrated pipettes usage
Calibrated plastic pipettes
When an accuracy is needed in dispensing small liquid volumes (1-2 ml), a graduated cylinder is not suitable because the pouring action results in a significant loss of material. Calibrated plastic pipettes have markings at 0.25 ml increments for a 1 ml pipette, and are economical ways to dispense relatively accurate volumes.

a) 1 ml Calibrated plastic pipette, b) Suction of liquid, c) Depressing bulb to required volume (the arrow points to the 1 ml mark), d and e) Transfer of liquid.
To utilize a calibrated plastic pipette, withdraw some of the liquid to be transferred into the bulb (Fig. 9 b). Then squeeze the bulb just enough so that the liquid pours to the desired volume (Fig.9 c), and maintain your position. While keeping the bulb squeezed so the liquid still reads to the desired volume, rapidly move the pipette to the transfer flask (Fig.9 d), and push the bulb farther to pour liquid into the flask (Fig.9 e).
Calibrated Glass Pipettes
When a high level of accuracy is required while dispensing liquids, calibrated glass pipettes (volumetric or graduated) can be utilized. Volumetric pipettes have a glass bulb at the top of their neck, and are able of dispensing only one certain volume (for example, the top pipette in Fig. 10 is a 10.00 ml pipette). Graduated pipettes (Mohr pipettes) have marks that let them to deliver many volumes. Both pipettes have to be connected to a pipette bulb to provide suction.

The volume marks on graduated pipettes show the transferred volume, which may seem a bit “backward” at first. For instance, when a graduated pipette is held vertically, the highest marking is 0.0 ml, which indicates that no volume has been transferred when the pipette is still full. As liquid is poured out into a vessel, the volume marks increase on the pipette, with the lowest mark often being the total capacity of the pipette (e.g. 1.0 ml for a 1.0 ml pipette).
Graduated pipettes can transfer any volume of liquid made possible by differences in the volume marks. For instance, a 1.0 ml pipette can be used to deliver 0.4 ml of liquid by: a) Withdrawing liquid to the 0.0 ml mark, then pouring out and delivering liquid to the 0.4 ml mark, or b) Withdrawing liquid to the 0.2 ml mark and pouring out and delivering liquid to the 0.6 ml mark (or any combination where the difference in volumes is 0.4 ml).
It is important to look attentively at the marks on a graduated pipette. Three different 1 ml pipettes are shown in Fig.11 a. The left-most pipette has markings every 0.1 ml, but no intermediary marks so that is less precise than the other two pipettes in Fig.11 a. The other two pipettes differ in the markings on the bottom. The lowest mark on the middle pipette is 1 ml, while the lowest mark on the right-most pipette is 0.9 ml. To transfer 1.00 ml with the middle pipette, the liquid must be poured out from the 0.00 ml to the 1.00 ml mark, and the final inch of liquid should be retained. To transfer 1.00 ml with the right-most pipette, liquid must be poured out from the 0.00 ml mark completely out the tip, with the intent to deliver its total capacity.
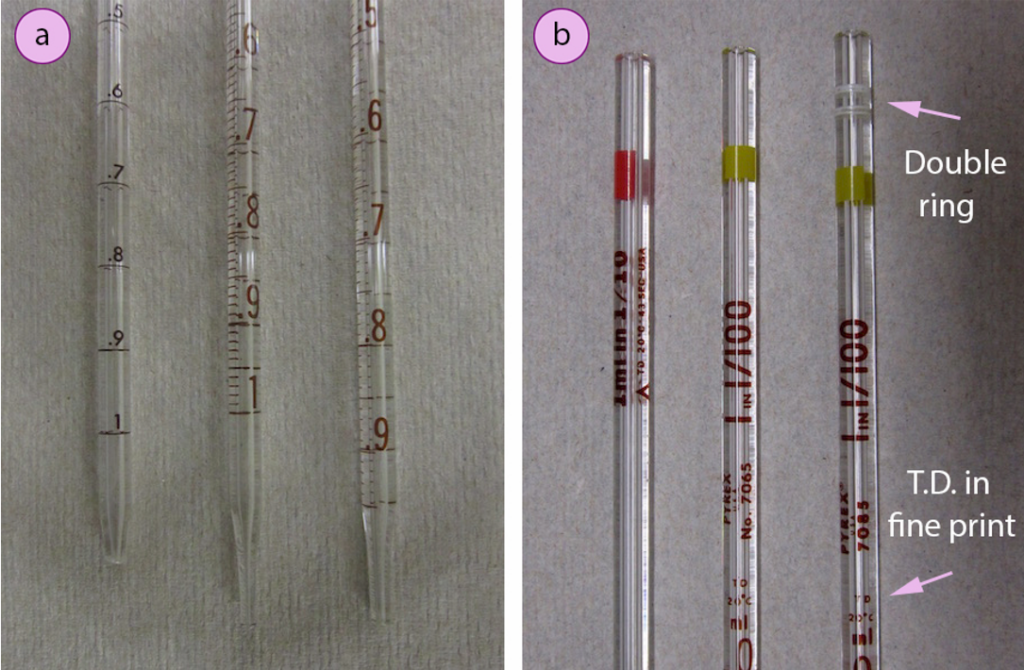
a) Bottom of pipettes, b) Top of pipettes
Pipettes are calibrated “to-deliver” (TD) or “to-contain” (TC) the marked volume. Pipettes are marked with T.C. or T.D. to differentiate between these two kinds, and to-deliver pipettes are also marked with a double ring near the top (Fig.12 b). After pouring out a “to-deliver” pipette, the tip should be touched to the side of the flask to withdraw any clinging drops, and a small amount of residual liquid will remain in the tip. A “to-deliver” pipette is calibrated to deliver only the liquid that freely drains from the tip. Nevertheless, after draining a “to-contain” pipette, the residual liquid in the tip should be “blown out” with overpressure from a pipette bulb. “To-contain” pipettes may be useful for dispensing viscous liquids, when a solvent can be used to wash out the entire content.
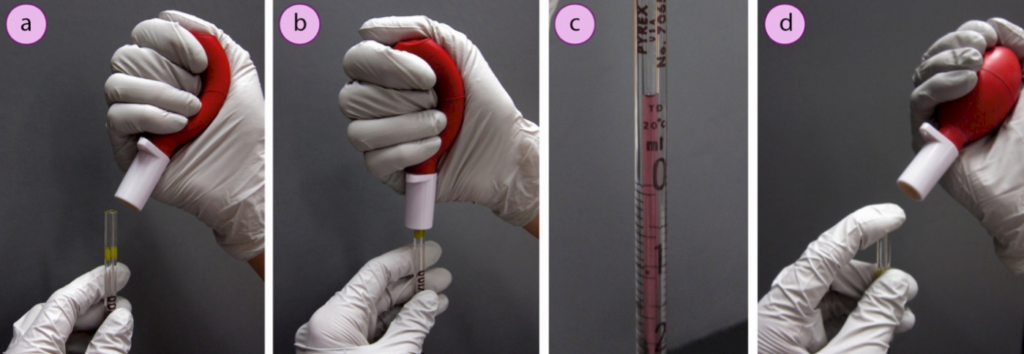
a and b) Applying suction to the pipette, c) Liquid withdrawn above the desired volume, d) The bulb is released and tip of the pipette sealed with a finger to maintain the liquid’s position.
Methods on how to use a calibrated glass pipette are described in this section. These methods are for use with a clean and dry pipette. If residual liquid is in the pipette tip from water or from previous use with a different solution, a fresh pipette have to be used.
As an alternative, if the reagent is not particularly expensive or reactive, the pipette can be “washed” with the reagent to remove residual liquid. To a pipette washing, rinse the pipette twice with a full volume of the reagent and collect the rinsing in a waste container. After two rinses, any residual liquid in the pipette will have been replaced by the reagent. When the reagent is then withdrawn into the pipette it will not be diluted or changed in any way.
To use a calibrated glass pipette:
- Place the pipette tip in the reagent, squeeze the bulb and connect it to the top of the pipette (Fig.12 a and b).
- Partially release pressure on the bulb to make suction, but do not fully release your hand or you may create too big a vacuum, causing liquid to be violently withdrawn into the pipette bulb. Suction should be applied until the liquid rises to just past the desired mark (Fig.12 c).
- Break the seal and remove the pipette bulb, then quickly put your finger atop the pipette to prevent the liquid from draining (Fig.12 d).
- With a slight rocking motion or slight release of pressure from your finger, allow tiny amounts of air to be let into the top of the pipette in order to slowly and controllably pour out the liquid until the meniscus is at the desired volume (Fig.13 a shows a volume of 0.00 ml).
- Holding the top of the pipette tightly with your finger, bring the pipette to the flask where the liquid is to be delivered and again allow tiny amounts of air into the top of the pipette in order to slowly drain the liquid to the desired mark (Fig.13 b and c indicate the delivered volume to be lightly below 0.20 ml).
- Touch the pipette tip to the side of the vessel to remove any hanging drops and remove the pipette.
- If liquid was pored out to the bottom of the pipette with a T.C. pipette, use pressure from a pipette bulb to blow out the residual liquid. Do not blow out the residual drop when using a T.D. pipette!
- If a volumetric pipette is used, the liquid have to be withdrawn with suction to the mark line above the glass bulb (shown in Fig.13 d). The liquid can be poured out into the new vessel with your finger fully released from the top. When the liquid draining end, the tip have to be touched to the side of the glass vessel to withdraw any clinging drops, but the residual drop should not be forced out (similar to a T.D. pipette).
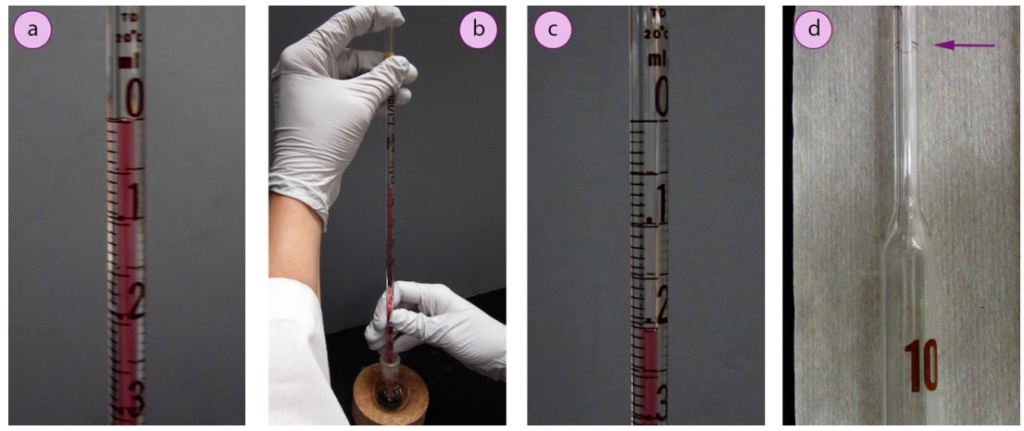
a) Red liquid to the 0 ml mark, b) Delivering substance, c) Final volume, d) Volumetric pipette (arrow show the fill mark).
Dispensing Highly Volatile Liquids
During dispense highly volatile liquids attempting (e.g. diethyl ether or acetone) via pipette, it is very usual that liquid drips out of the pipette even without pressure from the dropper bulb. This happens as the liquid evaporates into the pipette’s headspace, and the additional vapor causes the headspace pressure to exceed the atmospheric pressure. In order to avoid a pipette from dripping, withdraw and pour out the liquid into the pipette several times. As soon as the headspace is saturated with solvent vapors, the liquid will no more drip.
Pouring Hot Liquids
It can be hard to manipulate a glassware of hot liquid with your unprotected hands. If draining a hot liquid from a beaker, a silicone hot hand protector can be used (Fig.14 a) or beaker tongs (Fig.14 b and c).

a) Hot hand protector, b and c) Beaker tongs, d) Paper towel holder.
Hot hand protectors can also be used when pouring a hot liquid from an Erlenmeyer flask, but they do not hold the inconvenient shape of the flask very safely. Pouring from hot Erlenmeyer flasks can be completed with help of a improvised “paper towel holder”. A long part of paper towel is folded several times in one direction to the thickness of approximately 3-4 cm (and fixed with lab tape, Fig.15 a). This folded paper towel can be wrapped around the top of a beaker or Erlenmeyer flask and clamped to hold the vessel (Fig.14 d and Fig. 15 b).
While pouring hot liquid from an Erlenmeyer flask, the paper towel holder have to be narrow enough that the towel does not reach the top of the flask. If it happens, liquid will wick toward the paper as it is poured, thus weakening the holder and also removing possibly valuable solution (Fig. 15 c). When the paper towel is a distance away from the top of the flask, liquid can be poured from the flask without absorbing the liquid (Fig. 15 d).

a) Paper towel holder, b) Holding an Erlenmeyer flask with a paper towel holder, c) A too-wide holder, causing liquid to wick onto the paper as it is poured, d) A narrower holder, which pours without wicking.
