- The process of creating a drug - May 23, 2023
- How do scientists model mental disorders in animals? - May 23, 2023
Distillation is the process of evaporating a liquid and then cooling and condensing the vapor. It is primarily used to separate and refine components of multi-component substances, but also for other processes involving phase transformation and mass transfer, such as sublimation, crystallization, and liquid extraction. Distillation can be further divided into distillation with condensation of steam into a liquid, which results in a distillate with an average composition due to mixing, and distillation with condensation of steam into a solid, which results in a condensate with a distribution of component concentrations. The outcome of distillation is either a distillate or a residue, or both, depending on the substance and the purpose of the process. The main parts of a distillation device are a heated container of the liquid to be distilled, a cooled condenser, and a heated steam line connecting them.
Distillation setup
Partial vaporization of a liquid blend by ongoing extraction and condensation of the ensuing vapors in a cooling system is referred to as simple distillation. The resulting condensed liquid is known as a distillate and the unevaporated liquid is a vat residue.
Fractional distillation (or fractional distillation) is a process of segregating multicomponent liquid mixtures into distinct parts, fractions, by collecting parts with different volatility, beginning with the first, enriched with low boiling components. The residue of the liquid is augmented with a high-boiling element. To improve fraction separation, a deflagmator is used.
Rectification is a method of distillation whereby a part of the liquid condensate (phlegm) is constantly recycled back into the vat, towards the steam in the tower. As a consequence, the impurities contained in the steam are partly passed into the phlegm and sent back to the vat, thus increasing the purity of the steam (and condensate).
Applications
The use of distillation can generally be divided into four categories: laboratory scale, industrial distillation, distillation of herbs for perfumery and medicinals (herbal distillate), and food processing. The main difference between laboratory scale distillation and industrial distillation is that laboratory scale distillation is usually performed on a batch basis, while industrial distillation usually occurs in a continuous manner. In batch distillation, the composition of the source material, the vapors of the distilling compounds, and the distillate vary during the distillation.
In batch distillation, a still is loaded (provided) with a batch of feed mixture, which is then separated into its constituent fractions, which are collected sequentially from most volatile to less volatile, with the bottoms – the residual least or non-volatile fraction – removed at the end. The still can then be replenished and the process repeated.
Description
Laboratory display of distillation: 1: A heat source 2: Round bottomed flask 3: Still head (Würz flask ) 4: Thermometer 5: Condenser 6: Cooling water in 7: Cooling water out 8: Distillate/receiving flask 9: Vacuum/gas inlet 10: Still receiver 11: Heat control 12: Stirrer speed control 13: Stirrer/heat plate 14: Heating (Oil/sand) bath 15: Stirring mechanism (not shown) e.g. boiling chips or mechanical stirrer 16: Cooling bath.
Assembling
Chemical coolers can be employed either in reverse or downward (differ in the placement and manner of attachment when installing the apparatus).
The top of the condenser 5 is linked to a Würz flask 3, a Würz head or a tube stemming from the flask containing the initial combination 2. The bottom is connected to the allonge 10, through which the product of synthesis or distillation moves into the receiver 8. The cooling agent (water) is provided exclusively from the bottom 6 up 7. If the condenser 5 is fed from the top downward, the condenser jacket will not fill up completely, making the cooling ineffective. In addition, with such a supply, the condenser may break (crack) due to localized overheating of the shirt. It is essential to always monitor that the circulation of water through the condenser jacket does not stop, as deactivation of the condenser can lead to fires and explosions.
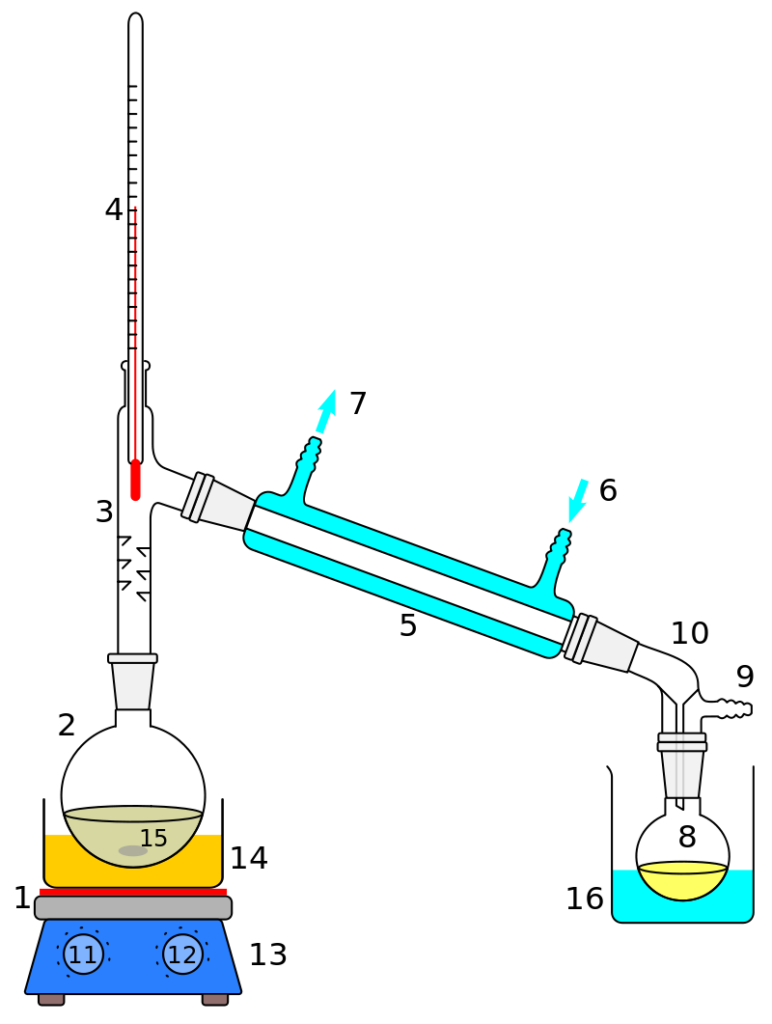
Condensers are an essential element of distillation. Types of condensers:
Straight cooler (downward) – utilized to condense vapors and extract the ensuing condensate from the reaction system. The collection of condensate is completed in a receiving flask;
Reflux condenser – used to condense vapors and return condensate to the reaction mass. These refrigerators are usually installed vertically.
Reflux condensers are used when engaging in the reaction at the boiling point of the reaction mixture, but without distilling off the liquid; they provide for the condensation of vapors and the condensate seeping back into the reactor along the walls of the refrigerator.
Dephlegmator – a condenser for partial condensation of a light part of steam, reflux.
The most uncomplicated type of laboratory condenser is an air condenser, which is generally just a glass tube that is cooled by ambient air. It is used solely in dealing with high-boiling liquids (preferably with a boiling point of at least 300 °C), which, when working with a water cooler, due to the large temperature difference, could split in the glass of the refrigerator.
Liebig condenser
It is mainly utilized as a downward flow to about 160 °C. The cooling agent for substances with a boiling point of less than 120 °C is running water, and in the range of 120-160 °C it is not moving. The Liebig condenser consists of two glass tubes sealed one into the other. Liquid vapors move along the inner tube, and a cooling agent (cold water) moves along the outer (jacket). As a reverse, such a condenser is ineffective, since it has a small cooling surface and a laminar vapor flow; for this purpose it is employed for high-boiling compounds (boiling point over 100 °C) compounds. Atmospheric moisture condenses on the outer surface of the condenser through capillary cracks. It is also recommended to put on a cuff made of dry filter paper on the refrigerator above the section. Higher boiling liquids at the junction can cause internal stress, resulting in the glass to break. Therefore, Liebig condenser cannot be made of non-heat-resistant glass.

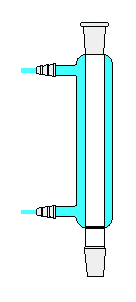
Ball (Allihn) condenser
Exclusively employed for backward flow. Since this condenser contains globular expansions, the vapour flux turns out to be chaotic in it; the cooling impact of this kind of refrigerator is significantly higher than that of a Liebig condenser. Nevertheless, atmospheric humidity also condenses on its external surface and the joint is likewise hazardous. The cooling agent is provided from the bottom to the top. It is expedient to insert the stirrer stem through the ball cooler, introduce various substances into the reactor, which are perfectly washed off into the flask by condensation and heated by it. Usually, the number of balls in this sort of condenser ranges from 3 to 8. To prevent inundation, when condensation does not have time to drain back into the flask with boiling liquid, the reflux condenser is established in an inclined position, yet the slope should not be too pronounced so that condensation does not accumulate in the balls. The accumulation of condensate results in a reduction in the efficient cooling surface of the refrigerator.
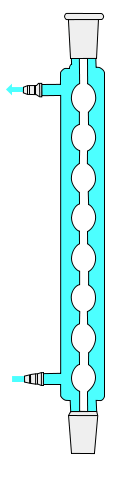
Grahams condenser
A Graham or Grahams condenser has a spiral coil with a cooling jacket along its length serving as the vapor–condensate route. This should not be mistaken with the coil condenser. The coiled condenser tubes inside will offer more area for refrigeration, making it the more favorable option; however, the downside of this condenser is that as the vapors condense, they tend to ascend the tube, which can lead to flooding of the solution combination. It may also be referred to as Inland Revenue condenser since it was designed for this purpose.


Dimroth’s condenser
An exceedingly productive reflux condenser. It is also employed as a downstream provided the relatively extensive wastage of distillate in the coil can be overlooked. The convergence of the jacketed coil A is beyond the area with a hefty temperature contrast, thus utilizing this chiller when working with liquids boiling over 160° C, one need not be apprehensive of complications. Since the external jacket of the chiller is air at room temperature, atmospheric moisture does not collect on its surface. Low-boiling substances can “creep” along the inner side of the jacket and thereby “pull” the cooling region. The Dimroth condenser is thus unsuitable as a reverse for relatively low boiling substances such as ether. At the upper open end of the condenser, atmospheric moisture readily gathers on the water supply hoses, so it is provided with a calcium chloride tube.
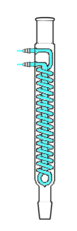


An alonge and a Spider alonge. The Spider alonge is a tool for gathering various components of distillate without needing to take apart a distillation system.


Instruction

For the segregation of fluids that vaporize at temperatures underneath 150 °C at 1 atm from non-volatile impurities and another liquid evaporating 25 °C higher than the initial liquid, and that are able to dissolve in each other, methods of heating should be taken into consideration.
Heat Source
If one of the constituents boils at temperatures below 70 °C, it is suggested to employ a steam bath or a heating mantle since the use of a Bunsen burner could cause an outbreak. Moreover, one has to be knowledgeable of the utilization of boiling stones and clamps.
Würz Flask
When assembling the apparatus, it is vital to ensure that the joints are properly aligned, as the pressure of one connection tends to loosen the other. It is necessary that all junctions are firmly in place.
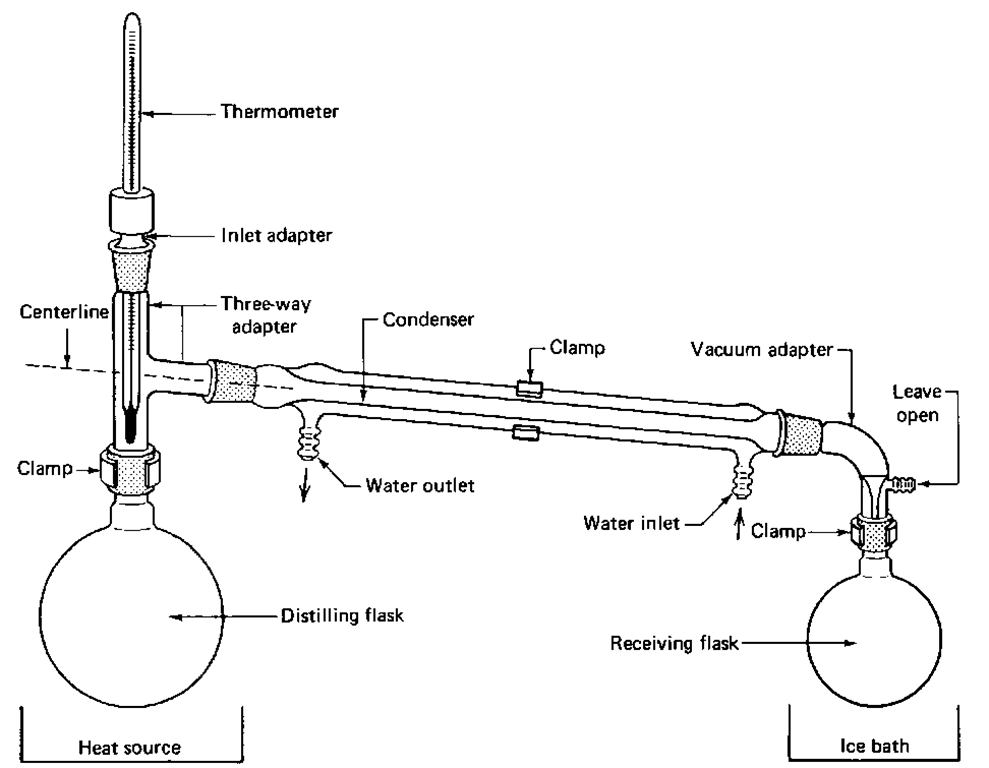
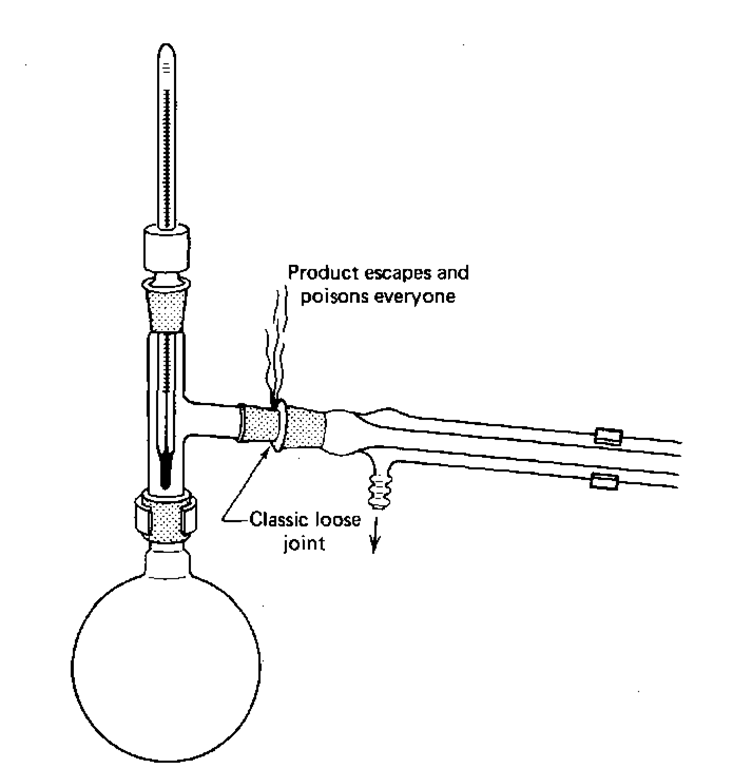
The Distilling Flask
Pour the liquid you wish to distill into the distilling flask. Take out the thermometer and its adapter, fill the flask utilizing a funnel, then reinsert the thermometer and its adapter. Don’t exceed half the capacity of the flask. Place a boiling stone if you have not already. These spongy little stones support bubbling and stop the liquid from overheating and leaping out of the flask. This leaping around is identified as bumping. NEVER throw a boiling stone into hot liquid or you may be showered by the hot liquid as it splashes out at you.
Make sure all the ties in your setup are firm. Begin the heat gradually until a gentle boiling starts and liquid begins to trickle into the receiving flask at the rate of around 10 drops every minute. This is essential. If nothing is obtained, you’re not distilling, just wasting time. You may have to raise the heat to keep material flowing over.
The Thermometer
Ensure that the full extent of the thermometer is beneath the ‘T’ joint of the 3-way connector. If droplets of liquid are not crystallizing on the thermometer, the reading you take is incorrect. Make a note of the temperature of the liquid that is being distilled. It serves as a measure of its quality. If the liquid is gathered over a 2°C gap, it is likely to be pure.
The Condenser
Always keep a flow of icy liquid running through the condenser, so much so that at least the lower half is frigid to the touch. Take into account that the water should enter at the bottom and exit at the top. Note that the water pressure in the lab may alter occasionally and usually rises during the night, since it is not used as much then. Subsequently, if you plan to have the condenser cooling water running overnight, secure the tubing at the condenser and the faucet with wire or something alike. Moreover, to avoid flooding the lab, make sure the output hose will not slip out of the sink.
The Vacuum Adapter
It is of utmost importance that the tubing connector is exposed to the atmosphere or else the entire system will be destroyed.
WARNING: Do not merely attach the vacuum adapter to the end of the condenser and suppose that it will not detach and break.
The Receiving Flask
The collection vessel must be of a size sufficient to collect whatever is desired. It may be necessary to substitute multiple ones over the course of the distillation, but the general rule is to have one receptacle on hand for discarding the output and others to retain the desired substances.
The Ice Bath (if applicable)
The practice of cramming a bucket with ice and unsuccessfully attempting to plunge a flask into this chaos has always mystified me. What temperature reduction can you realistically expect from merely having a few small areas of the flask in contact with the ice? Obtain a suitable container – a sizeable beaker, enameled tray, or any other. It should not be prone to leaking. Position it beneath the flask and fill it with water. Add the ice and stir.


