- The process of creating a drug - May 23, 2023
- How do scientists model mental disorders in animals? - May 23, 2023
Introduction
Ethylamine (ethanamine), an organic compound with the formula CH3CH2NH2, is an important object in the field of chemistry. The combination of an ethyl group with an amine functional group makes it an important member of amines, a class of organic compounds widely used in the chemical industry and science. Ethylamine has unique physicochemical properties that determine its behavior under various conditions.
Ethylamine is a natural product found in fruits and vegetables, grains, coffee, cheeses, and seafood. It is also a component of tobacco smoke.
In terms of its applications, ethylamine is used in the chemistry laboratory, to create drugs, produce polymers, develop fertilizers and herbicides in agriculture, and corrosion inhibitors. It is also used to synthesize phenylethylamines.
This article describes the physicochemical properties of ethylamine, its methods of preparation, chemical reactions, applications in various fields, health effects and safety precautions.
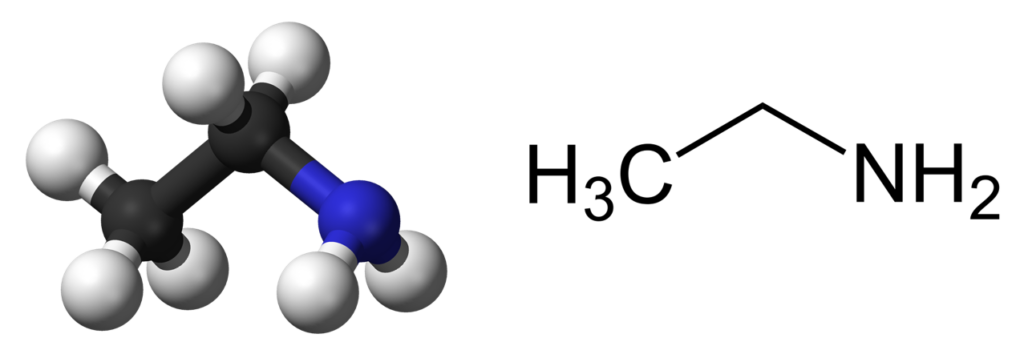
Physical and Chemical Properties of Ethylamine
Ethylamine (ethanamine) is an organic compound with the chemical formula CH3CH2NH2 and a molecular weight 45.085 g/mol. This is colorless gas with a strong ammonia-like odor. Its density is 0.688 g/cm3 (at 15°C). It is soluble in water, diethyl ether, and ethanol. Ethylamine is flammable, with flash point -37°C, autoignition temperature 383°C, and a relatively low boiling point of about 17°C.
As a derivative of ammonia, ethylamine exhibits its basic properties due to the lone pair of electrons on the nitrogen atom. The amine functional group in ethylamine (–NH2) imparts nucleophilic properties.
Synthesis of Ethylamine
The synthesis of ethylamine involves various methods, the most common method being the reaction between ammonia and alcohol. The following are examples of methods for synthesizing ethylamine:
Ethylamine can be synthesized by reacting ammonia with alcohol. The reaction is carried out in the presence of Cu or Ni catalyst supported on Al2O3. Typically, the reaction is carried out at low temperatures, from -20°C to 50°C. In this reaction, ethylamine is coproduced together with diethylamine and triethylamine. This process typically results in the formation of ethylamine and is represented by the following chemical equation:

Another method involves reductive amination, where aldehydes or ketones react with ammonia in the presence of reducing agents. Here, acetaldehyde reacts with ammonia in the presence of hydrogen gas to produce ethylamine and water. The reaction can be represented as follows:

There is also a method of synthesis ethylamine is the hydrogenation from acetamide, using a catalyst. This reaction can be carried out using lithium aluminum hydride catalyst in the presence of hydrogen.

One of the methods for producing ethylamine is the hydrogenation of nitroethane. This reaction can be carried out using lithium aluminum hydride or on a nickel catalyst in the presence of hydrogen.

These examples show some of the varied methods for synthesizing ethylamine. The choice of synthesis depends on the availability of reagents and equipment.

Chemical Reactions of Ethylamine
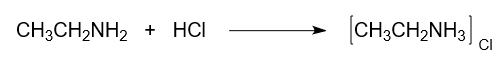
- Reaction with Acid:
Ethylamine, being a basic compound, reacts with hydrochloric acid to form ethyl ammonium chloride. This reaction shows the basicity of ethylamine as it accepts a hydrochloric acid proton.
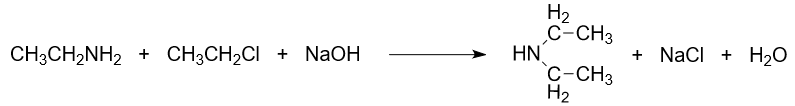
- Alkylation Reaction:
Ethylamine can participate in alkylation reactions with alkyl halides. The reaction is carried out in two stages. The intermediate product is a salt that can be treated with alkali to release the amine. In this reaction, an alkyl group is transferred to the nitrogen atom of ethylamine. For example, the reaction of ethylamine with ethyl chloride produces diethylamine, sodium chloride and water.
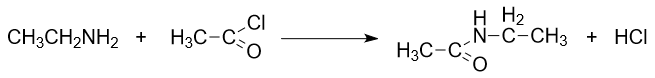
- Reaction with Acyl Chlorides:
Ethylamine reacts with acyl chlorides (acid chlorides) to form amides. This reaction demonstrates the ability of ethylamine to function as a nucleophile. The reaction of ethylamine with acetyl chloride is presented as follows:
- Reactions with Ketones:
Ethylamine reacts with ketones. For example, synthesis of 3,4-Methylenedioxy-N-ethylamphetamine (MDE, MDEA) using ethylamine. For this synthesis usually used 70% ethylamine with cold isopropyl alcohol and methanol (75%, 25%) with aluminium amalgam catalysator. Magnesium sulfate can be added to the solution to remove water.
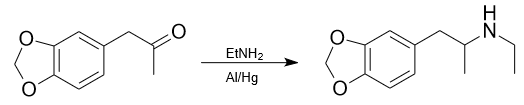
These examples show the ability of ethylamine to participate in various reactions making it a valuable material in the synthesis of various organic compounds.

Applications of Ethylamine
- Organic Synthesis:
Ethylamine is used as an intermediate in the synthesis of various chemical compounds. Its reactions with acyl chlorides, aldehydes, and ketones lead to the formation of amides and amines, making it a valuable intermediate in the preparation of a wide range of chemicals. - Pharmaceutical Industry:
Ethylamine finds application in the pharmaceutical industry for the synthesis of a variety of drugs. A prime example is the antihypertensive drug guanethidine. Ethylamine is a main intermediate in the synthesis of guanethidine. - Polymerization Reagent:
In the field of polymer chemistry, ethylamine is used as a reagent in the polymerization of certain materials. It can be used in the production of polyamides, which are important polymers with applications in the manufacture of fibers, plastics, and resins. - Agricultural Applications:
Ethylamine is used in agriculture as a component in the synthesis of some pesticides. For example, the herbicide atrazine is produced using ethylamine as one of the starting materials. Atrazine plays a critical role in weed control in various crops. - Corrosion Inhibitors:
Ethylamine derivatives are used in corrosion inhibitors. These inhibitors help protect metal surfaces from corrosion, and the use of ethylamine-based compounds facilitates the development of effective anti-corrosion coatings.
Health Effects of Ethylamine
- Inhalation Exposure:
Inhalation of ethylamine vapors may cause irritation to the respiratory system, leading to symptoms such as coughing, shortness of breath, and nasal irritation. Prolonged exposure to high concentrations may result in more severe respiratory effects. - Skin Contact:
Ethylamine is a corrosive substance that can cause irritation and burns upon contact with the skin. Prolonged or repeated exposure may lead to dermatitis and contact with concentrated solutions should be avoided. - Eye Exposure:
Contact with ethylamine can cause severe irritation to the eyes, leading to redness, tearing, and potential corneal injury. - Ingestion:
Ingestion of ethylamine is not a common route of exposure, but if it occurs, it may cause irritation and damage to the gastrointestinal tract. Avoid ingestion and in case of accidental ingestion, seek medical attention. - Toxicological Effects:
Ethylamine is considered to have low acute toxicity, but chronic exposure or exposure to high concentrations may lead to adverse effects on the central nervous system, liver, and kidneys.
Image: Tanker for transportation ethylamine
Safety Precautions
When working with ethylamine, it is important to take precautions to minimize the risk of exposure and ensure a safe work environment. Here are the basic safety precautions:
- Use appropriate personal protective equipment (PPE), including chemical resistant gloves, goggles, respirators, and protective clothing, when handling ethylamine.
- In case of skin contact, immediately rinse the affected area with water and seek medical attention if irritation persists.
- In case of eye contact, rinse the eyes with water and seek immediate medical attention.
- Work in a well-ventilated area or under the fume hood to minimize inhalation exposure.
- Store ethylamine in a cold, well-ventilated area away from incompatible materials such as acids, oxidizing agents, and strong reducing agents.
Conclusion
In conclusion, ethylamine, with its unique chemical properties, plays an important role in various industrial applications and organic synthesis. Ethylamine participates in various reactions such as acid-base reactions, alkylation, and acylation, showing its versatility in organic synthesis. Its reactivity makes it a valuable intermediate block in the manufacture of pharmaceuticals, agrochemicals, and polymers. However, it is important to recognize the health risks associated with ethylamine and take safety precautions.
Bibliography
- Electrochemical reduction of acetonitrile to ethylamine Rong Xia, Dong Tian, Shyam Kattel, Bjorn Hasa, Haeun Shin, Xinbin Ma Nature Communications volume 12, Article number: 1949 (2021) https://www.nature.com/articles/s41467-021-22291-0
- Rotation/Inversion Study of the Amino Group in Ethylamine Daniel Zeroka, James O. Jensen, and Alan C. Samuels J. Phys. Chem. A 1998, 102, 32, 6571–6579 Publication Date: July 17, 1998 https://doi.org/10.1021/jp982031a
- Pyrolysis of ethylamine. I. Microwave spectrum and molecular constants of vinylamine Frank J. Lovas; Frank O. Clark; Eberhard Tiemann J. Chem. Phys. 62, 1925–1931 (1975) https://doi.org/10.1063/1.430679
- Pyrolysis of ethylamine. II. Synthesis and microwave spectrum of ethylidenimine (CH3CH=NH) Frank J. Lovas; Richard D. Suenram; Donald R. Johnson; Frank O. Clark; Eberhard Tiemann J. Chem. Phys. 72, 4964–4972 (1980) https://doi.org/10.1063/1.439783
- On the role of strength and location of Brønsted acid sites for ethylamine synthesis on mordenite catalysts Victor A Veefkind, Martin L Smidt, Johannes A Lercher Applied Catalysis A: General
Volumes 194–195, 13 March 2000, Pages 319-332 https://doi.org/10.1016/S0926-860X(99)00378-6

