- The process of creating a drug - May 23, 2023
- How do scientists model mental disorders in animals? - May 23, 2023
Introduction
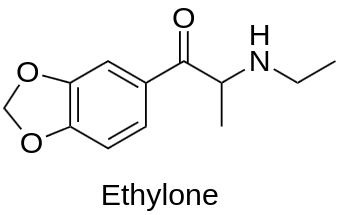
Ethylone is a synthetic compound that falls under the category of substituted cathinones, commonly known as “bath salts” or “research chemicals.” It is a member of the phenethylamine, amphetamine, and cathinone chemical classes and shares structural similarities with other stimulants such as MDMA (ecstasy) and methylone. Ethylone gained popularity in the early 2000s as a recreational drug due to its stimulating effects and empathogenic properties. However, its use has raised concerns regarding safety and potential health risks. In this article, we will explore the general information, physico-chemical properties, synthesis, pharmacology, effects, applications, and legal status of Ethylone.

General Information About Ethylone
As a designer drug, ethylone is commonly sold on the street along with other cathinones like butylone or 3-MMC as a substitute or counterfeit for MDMA and/or methylone due to methylone’s declining availability on the research chemicals market. However, in spite of behavioral and pharmacological similarities between ethylone, MDMA and methylone, it should be noted that the observed subjective effects of both drugs are not completely identical.
Ethylone has only a short history of human use and is reported to be less potent than its relative methylone as well as possessing more classical stimulant-type as opposed to entactogenic effects.
As an illicit synthetic substance, ethylone is frequently peddled on the streets, often alongside other cathinones like butylone or 3-MMC. This is primarily done as a substitution or imitation for MDMA and/or methylone, given the decreasing availability of methylone in the research chemicals market. Despite sharing behavioral and pharmacological similarities with MDMA and methylone, it is essential to highlight that the subjective effects of these drugs are not entirely identical.
Ethylone has a relatively brief history of human use and is reported to be less potent than its counterpart, methylone. Additionally, it is said to exhibit more typical stimulant-like effects rather than entactogenic effects.
- IUPAC Name of Ethylone: (RS)-1-(1,3-benzodioxol-5-yl)-2-(ethylamino)propan-1-one;
- Other synonyms names of Ethylone are: 1-Propanone, 1-(1,3-benzodioxol-5-yl)-2-(ethylamino)-; 1-(1,3-Benzodioxol-5-yl)-2-(ethylamino)-1-propanone; 2-Ethylamino-1-(3,4-methylenedioxyphenyl)propan-1-one; 1-(Benzo[d][1,3]dioxol-5-yl)-2-(ethylamino)propan-1-one; 3,4-methylenedioxy-N-ethylcathinone; MDEC; βk-MDEA.
- CAS №: 1112937-64-0 (base); 1454266-19-3 (hydrochloride salt)
Physico-chemical Properties of Ethylone
Ethylone hydrochloride is a white to off-white crystalline solid at room temperature and ambient pressure, often obtained in its hydrochloride salt form. It has a molar mass of approximately 221.25 g/mol, with a chemical formula of C12H15NO3. The structure of Ethylone incorporates a phenethylamine core, featuring a phenyl ring bound to an amino (NH2) group through an ethyl chain. Attached to the phenyl ring is a methylenedioxy group, which forms a 1,3-benzodioxole ring when it closes across the 5 and 6 positions on the benzene ring. The substance also incorporates an N-ethylcarboxamide group that confers it the cathinone properties.

In terms of solubility, Ethylone is soluble in organic solvents such as dichloromethane, ethanol, and dimethylformamide. Its solubility in water is low, which is a common feature among synthetic cathinones due to their lipophilic properties. Its hydrochloride salt form melting point ranges from 230 to 240 degrees Celsius, depending on the purity of the compound and the specific conditions under which it’s measured. The compound’s pKa and partition coefficient (LogP) values are not well-studied, although they would be key in understanding its bioavailability and pharmacokinetics.
As a solid substance, Ethylone is stable under normal temperatures and pressures. However, it may be sensitive to light, heat, and moisture, which could potentially cause degradation or changes in its structure. Therefore, it should be stored in a cool, dry, and dark place to maintain its stability. The compound’s reactivity with other substances is not extensively documented but it is known to react with strong oxidizing agents.
Ethylone Synthesis
The synthesis of Ethylone involves complex procedures that require a fair degree of chemical expertise, as well as access to precursor substances that are often controlled due to their potential misuse in illicit drug manufacturing. The process typically starts with the bromination of 3,4-methylenedioxypropiophenone (MDP2P), a phenylpropanoid used in the synthesis of various psychoactive substances.
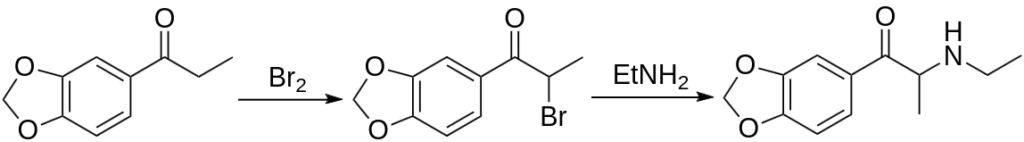
The bromination is achieved by reacting MDP2P with bromine in an organic solvent, such as dichloromethane, under cooling. This reaction yields 2-bromo-3,4-methylenedioxypropiophenone as the product. This intermediate compound, a bromoketone, is then reacted with ethylamine in a reductive amination process.
The overall process needs to be conducted under controlled conditions to avoid side reactions and byproducts. Also, it’s important to emphasize that many of the substances involved in Ethylone synthesis are strictly regulated, and unlicensed possession or use of these substances can carry severe legal penalties. Therefore, the synthesis of Ethylone is generally only carried out in licensed laboratories for the purpose of legitimate research.
Pharmacology of Ethylone
Ethylone operates as a compound that both inhibits the reuptake and stimulates the release of serotonin, norepinephrine, and dopamine. These neurotransmitters play crucial roles in the feelings of pleasure, reward, drive, and concentration. Ethylone impedes these neurotransmitters’ reuptake and reabsorption after they have completed their neural transmission duties, effectively allowing their accumulation for reuse, which results in physical stimulation and feelings of euphoria.
When compared with methylone, Ethylone shows significantly less, approximately three times lower, affinity for the serotonin transporter (which already has three times lower affinity than MDMA). However, its affinity for the norepinephrine and dopamine transporters remains comparable. The consequence of these pharmacological differences when juxtaposed with methylone is that Ethylone has a lower potency per dose and exhibits a more balanced effect on catecholamines compared to serotonin. Its behavior is more akin to a reuptake inhibitor, similar to methylphenidate, than a releaser like amphetamine. Nevertheless, Ethylone still possesses substantial releasing potential.
Ethylone Effects
Physical effects
- Spontaneous physical sensation – Ethylone’s “body high” can be characterized as a moderately to extremely euphoric sensation of tingling that envelops the entire body. This sensation can become overwhelmingly pleasurable at higher dosages and consistently rises until it reaches its peak.
- Stimulation – Ethylone is widely viewed as highly stimulating and energizing concerning its effects on physical energy levels. It promotes activities such as running, climbing, and dancing, making it a popular choice at music events like festivals and raves. The kind of stimulation offered by ethylone could be termed as ‘forced’. Meaning, at higher dosages, it becomes challenging or even impossible to remain motionless, as effects such as jaw clenching, unintentional body shakes, and vibrations become evident, leading to extreme hand unsteadiness and overall motor control disruption.
- Vibrating vision – At high doses, individuals may experience their eyeballs moving rapidly back and forth spontaneously, resulting in blurred and temporarily out-of-focus vision. This condition is known as nystagmus.
- Dehydration – The universal experience with ethylone involves feelings of a dry mouth and dehydration, a consequence of an increased heart rate and the strong drive to engage in strenuous physical activities. While it’s crucial to avoid dehydration (especially when dancing in a warm setting), over-drinking has led to water intoxication in some users, so it’s recommended to sip water without overconsuming.
- Difficulty urinating – Higher ethylone doses can cause temporary and harmless urination difficulties. This effect is due to ethylone stimulating the release of the anti-diuretic hormone (ADH), which regulates urination. This effect can be mitigated by relaxing, or more significantly alleviated by applying a warm cloth to the genital area to stimulate blood flow.
- Temperature regulation suppression
- Tactile enhancement
- Increased heart rate
- Increased perspiration
- Increased blood pressure
- Teeth grinding – This component can be considered to be less intense when compared with that of MDMA.
Cognitive effects
The cognitive impacts of ethylone can be categorized into several elements that increase in intensity with the dosage. The typical mental state induced by ethylone is often characterized as intense cerebral stimulation, subtle feelings of affection or empathy, and moderate euphoria. It encompasses a variety of standard psychedelic, entactogenic, and stimulant cognitive effects. The most noticeable of these cognitive effects typically include:
- Cognitive Euphoria – Ethylone is known to induce powerful emotional euphoria and sensations of joy, likely due to the release of serotonin and dopamine. Compared to MDMA, its effects are more closely related to the euphoria experienced with amphetamine and mephedrone.
- Enhancement of Empathy, Love, and Sociability – While distinctive and potent, this specific effect is less noticeable and therapeutic compared to that of MDMA. It can be described as less aggressive and more introspective, leading to feelings of love and empathy that don’t necessarily need to be outwardly expressed.
- Time Distortion – Ethylone often induces strong feelings of time compression, noticeably accelerating the perception of time. The effects experienced during the decline of a stimulant experience usually feel negative and uncomfortable compared to the peak effects. This is often known as a “comedown,” resulting from neurotransmitter depletion.
- Time distortion – Strong feelings of time compression are common within ethylone and speed up the experience of time quite noticeably.
- Thought acceleration
- Analysis enhancement
- Mindfulness
- Increased libido
- Increased music appreciation
After effects
The sensations experienced during the diminishing phase of a stimulant’s impact are typically unpleasant and distressing compared to the sensations felt at its zenith. This is commonly known as a “comedown”, a phenomenon resulting from the depletion of neurotransmitters. Its effects commonly include:
- Anxiety
- Cognitive fatigue
- Depression
- Irritability
- Motivation suppression
- Thought deceleration
- Wakefulness
Applications of Ethylone
Ethylone has been primarily used in scientific research and is of particular interest in the fields of neurology and psychopharmacology. Its impact on neurotransmitters—serotonin, norepinephrine, and dopamine—makes it a valuable compound for studying the neurobiological mechanisms of mood regulation, motivation, pleasure, and reward.
In academic research, Ethylone has been used as a reference compound for the development of analytical methods. With its structural similarity to other phenethylamines and synthetic cathinones, Ethylone serves as a useful benchmark for comparing and identifying these substances in both biological and environmental samples.
Due to its psychoactive effects, Ethylone has also been used—albeit illicitly—in recreational settings for its euphoric and stimulant properties. It has been found in products sold as “bath salts,” “plant food,” or under various other names, often in combination with other synthetic cathinones. It should be noted, however, that such use is often illegal and poses significant health risks, as the purity and safety of these products can rarely be guaranteed.
Furthermore, as an analogue of substances like MDMA and Methylone, Ethylone provides insights into the structure-activity relationships of these compounds. This could potentially aid in the development of new therapeutics with improved safety profiles. However, it’s crucial to emphasize that any therapeutic use of Ethylone is purely hypothetical at this stage, and further research is necessary to evaluate its potential medical applications.
Ethylone Legal Status
- Brazil: Ethylone is illegal to possess, produce, and sell under Portaria SVS/MS nº 344.
- China: As of October 2015, Ethylone is a controlled substance in China.
- Germany: Ethylone is controlled under Anlage I BtMG (Narcotics Act, Schedule I) as of December 13, 2014. It is illegal to manufacture, possess, import, export, buy, sell, procure or dispense it without a license.
- Sweden: Ethylone is a controlled substance since 1992
- Switzerland: Ethylone is a controlled substance specifically named under Verzeichnis D.
- United Kingdom: Ethylone is a Class B drug in the United Kingdom as a result of the cathinone catch-all clause.
- United States: Ethylone is unscheduled in the United States. However it could be considered an analog of methylone or MDMA, thus putting it under the scope of the Federal Analog Act.
Conclusion
In conclusion, Ethylone—a synthetic cathinone and analog to MDMA and Methylone—has important applications in neurobiological research and offers potential for future therapeutic developments. Its interaction with key neurotransmitters offers insights into mood regulation, motivation, and reward systems. Moreover, it serves as a reference compound for the analysis and identification of similar substances.
Bibliography
- Maheux, Chad R., et al. “Identification of polymorphism in ethylone hydrochloride: synthesis and characterization.” Drug testing and analysis 8.8 (2016): 847-857. https://analyticalsciencejournals.onlinelibrary.wiley.com/doi/full/10.1002/dta.1859
- Simmler, L. D., et al. “Pharmacological characterization of designer cathinones in vitro.” British journal of pharmacology 168.2 (2013): 458-470. https://bpspubs.onlinelibrary.wiley.com/doi/abs/10.1111/j.1476-5381.2012.02145.x
- Coppola, Maurizio, and Rafaella Mondola. “Synthetic cathinones: chemistry, pharmacology and toxicology of a new class of designer drugs of abuse marketed as “bath salts” or “plant food”.” Toxicology letters 211.2 (2012): 144-149. https://www.sciencedirect.com/science/article/abs/pii/S0378427412001117
- https://psychonautwiki.org/wiki/Ethylone
- https://en.wikipedia.org/wiki/Ethylone
- https://commonchemistry.cas.org/detail?cas_rn=1112937-64-0
- https://www.swgdrug.org
