Introduction
Toluene (methylbenzene) is an aromatic hydrocarbon that is widely used in industry as a solvent and also in the production of other chemical compounds. This colorless liquid component has good dissolving properties, which makes it useful for dissolving various substances, including resins, dyes, varnishes and oils.

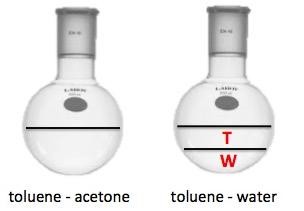
Toluene has the formula C7H8 and a molecular weight of 92.14 g/mol. It has a slight specific odor and evaporates at room temperature, making it easily spreads in the air. Toluene is miscible (soluble in all proportions) with diethyl ether, acetone, ethanol, benzene, chloroform, glacial acetic acid and carbon disulfide, but insoluble in water.
In this article we will look at the chemical properties of toluene, history, production and the dangers associated with its use.
History of Toluene Production
The history of obtaining toluene began in the 19th century, the compound was first isolated in 1837 through a distillation of pine oil by Pierre Joseph Pelletier and Filip Neriusz Walter, who named it retinnaphte. In 1841, Henri Étienne Sainte-Claire Deville isolated a hydrocarbon from balsam of Tolu (an aromatic extract from the tropical Colombian tree Myroxylon balsamum), which Deville recognized as similar to Walter’s retinnaphte and to benzene; hence he called the new hydrocarbon benzoene. In 1843, Jöns Jacob Berzelius recommended the name toluin. In 1850, French chemist Auguste Cahours isolated from a distillate of wood a hydrocarbon which he recognized as similar to Deville’s benzoene and which Cahours named toluene.
During the 20th century, toluene became widely used in industry. Production of toluene became based on synthesis from oil or gas, as opposed to production from benzoic acid. Today toluene is produced on an industrial scale worldwide and is an important raw material for many industrial processes.

Properties of Toluene
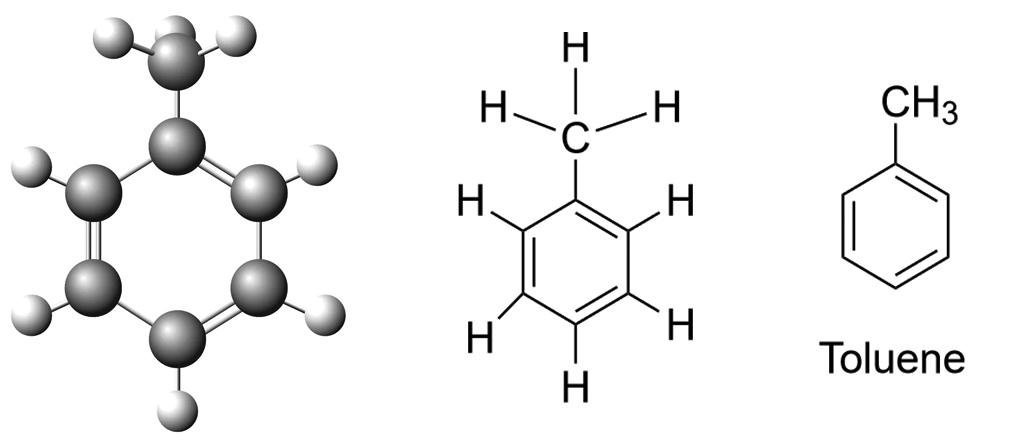
Toluene is an aromatic hydrocarbon, which contains an aromatic ring carbon bonded to a methyl group. This gives toluene several unique chemical properties. Reactions of electrophilic substitution in the aromatic ring and substitution in the methyl group by a radical mechanism. Electrophilic substitution in the aromatic ring occurs mainly in the ortho and para positions relative to the methyl group.
Toluene has a molecular formula C7H8 and the structural formula:
Physical properties
Toluene is a colorless liquid which has good dissolution properties. It has a density 0.87 g/cm³ at room temperature and pressure and a boiling point of 110.6°C. Toluene easily evaporates and forms volatile explosive vapors.
Chemical properties
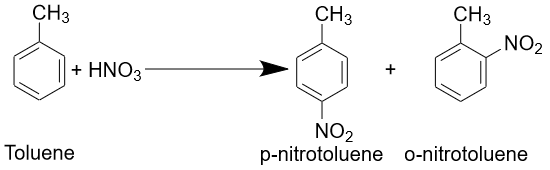
Toluene reacts with many other chemical compounds and can be used in different chemical reactions. It can be oxidized to benzoic acid or oxidized to phenol if a catalyst is presented. Toluene can also be acylated to obtain toluene acids or propionic acids. It can be nitrated to give ortho-, meta- or para-nitrotoluene.
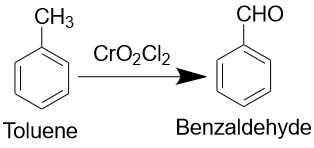
The methyl side chain in toluene is susceptible to oxidation. Toluene reacts with potassium permanganate to yield benzoic acid, and with chromyl chloride to yield benzaldehyde.
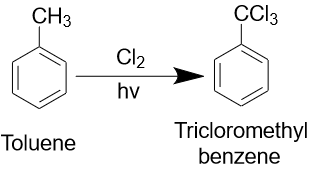
Toluene can react with oxygen at high temperatures to form toluene peroxide. It can also react with chlorine and bromine to form the corresponding toluene derivatives. Toluene can form compounds with amino-, hydroxyl-, acetyl-, formyl- and other functional groups.
Toluene Production
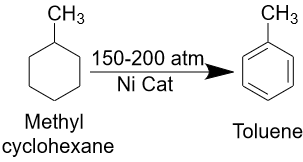
Toluene is produced from petroleum or dehydrogenation of methylcyclohexane. There is a method of producing toluene by isomerization of xylene, which takes place in the presence of catalysts such as aluminum oxide or silica.
Production of toluene from petroleum begins with the distillation of crude oil to produce a petrol fraction that contains toluene. Toluene is then extracted from petrol using extraction and distillation processes.
Another method of producing toluene is the dehydrogenation of methylcyclohexane. This process involves heating methylcyclohexane in the presence of catalysts such as copper oxide or chromium oxide at high temperatures and pressures. In the process, methylcyclohexane is dehydrated to toluene and hydrogen.
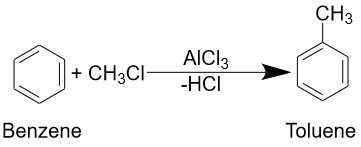
The Friedel-Crafts synthesis is a classic method for the production of toluene. This method involves the reaction of benzene with methyl chloride or dimethyl sulfate in the presence of a Lewis acid catalyst, usually aluminum chloride (AlCl3). The reaction proceeds through the formation of an intermediate carbocation, which is stabilized by the electron-donating effect of the alkyl group. The aluminum chloride catalyst is required to facilitate the formation of the carbocation and to remove the chloride ion that is produced as a byproduct of the reaction.
Toluene also can be obtained from industrial solvents for paints and varnishes in laboratory. When water is added, the mixture will separate into two parts, the components soluble in water and toluene.

Toluene Applications
Toluene has a wide range of applications in various industries. Some of these include:
- Plastics, paints and varnishes manufacturing: Toluene is used as a raw material for the production of plastics, paints and varnishes. These products are used in various industries such as automobile manufacturing, furniture manufacturing, construction and others.
- Solvent for varnishes, paints and adhesives: due to its solvent properties, toluene is used as a solvent for various varnishes, paints and adhesives. This allows these products to be applied more easily to surfaces.
- Benzene production: Toluene can be used as a raw material for benzene production. Benzene is also widely used in industry to make various products including rubbers, plastics, medicines and others.
- Explosives production: Toluene can be used as a raw material for the production of explosives such as TNT.
- Production of azo dyes: Toluene is used to produce azo dyes, which are widely used in the textile industry.
- Production of pharmaceuticals: toluene is used as a solvent for some pharmaceuticals such as aspirin and acetaminophen.
- Other uses: Toluene can also be used as an anti detonation liquid in automotive fuels, in the manufacture of rubber and other products.
The Dangers of Toluene
Toluene can pose a risk to human health if it is used or stored improperly. Here are some of the main hazards associated with toluene:
- Toxicity: Toluene is a toxic substance and can cause serious health problems if inhaled in large quantities. Dizziness, headache, nausea and vomiting occur following inhalation of toxic toluene vapor. Prolonged exposure to toluene can lead to various health problems such as impaired movement, damage to the nervous system and eyesight and the development of cancer.
- Fire hazard: Toluene is highly flammable and can pose a risk in case of fire or explosion. Toluene should therefore be stored in well-ventilated and protected from sources of fire.
- Risk of inhalation dependence: Toluene is sometimes used as a recreational inhalant (“glue sniffing”), likely on account of its euphoric and dissociative effects. Toluene directly stimulates dopamine neurons causing dopamine release. Dopamine is a neurotransmitter and is released by reward centers in the brain causing a feeling of euphoria. The results suggest that the brain likely also interprets sniffing toluene as rewarding. This can lead to the risk of inhalation addiction, which can be a serious health problem. The possession and use of toluene and products containing it are regulated in many jurisdictions, for the supposed reason of preventing minors from obtaining these products for recreational drug purposes.

Toluene is a highly flammable liquid and it can cause mild damage to the skin and the eyes. However, the most-common hazard associated with this chemical is inhalation (opens in a new tab). Products containing toluene can produce dangerous fumes which can cause nausea, headaches, unconsciousness, and even death if inhaled. You can be exposed to toluene when using products that contain it – for instance, when pumping gasoline or painting a room. Prolonged or repeated exposure to toluene vapors can cause severe damage to the central nervous system. Taking the necessary precautions when handling toluene or toluene products is essential.
Conclusion
In conclusion, toluene is an important chemical compound that is widely used in various industries, including solvents, plastics, paints, varnishes, pharmaceuticals and other products. It has unique physical and chemical properties that make it a useful and important component in many manufacturing processes.
Despite the hazards associated with its use, toluene remains an important and essential chemical compound in many industries and scientific research. Its properties and potential continue to be investigated and, in the future, it may become an even more important component in various production processes.
Bibliography
- Production of benzene, toluene, and xylenes from natural gas via methanol: Process synthesis and global optimization Alexander M. Niziolek, Onur Onel, Christodoulos A. Floudas AIChE Journal Volume 62, Issue 5 p. 1531-155623 December 2015
https://doi.org/10.1002/aic.15144 - Standard Reference Data for the Viscosity of Toluene Fernando J. V. Santos; Carlos A. Nieto de Castro; John H. Dymond; Natassa K. Dalaouti; Marc J. Assael; Akira Nagashima Journal of Physical and Chemical Reference Data 35, 1–8 (2006)
https://doi.org/10.1063/1.1928233 - Experimental and modeling study of the oxidation of toluene R. Bounaceur, I. Da Costa, R. Fournet, F. Billaud, F. Battin-Leclerc International Journal of Chemical Kinetics Volume 37, Issue 1 p. 25-49 02 November 2004
https://doi.org/10.1002/kin.20047 - Health effects of toluene: a review. Benignus V.A. Neurotoxicology, 01 Nov 1981, 2(3):567-588 PMID: 7038560
https://europepmc.org/article/med/7038560 - Toluene International Programme on Chemical Safety, World Health Organization & WHO 146p. 1985
https://apps.who.int/iris/handle/10665/41688

