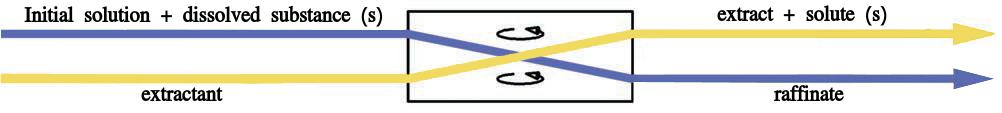
“Extraction” is the act of transferring compounds from either a solid or liquid into a different solvent or phase. For example, when a tea bag is put in hot water, the elements that give the tea its flavor and color are drawn out of the grounds and into the water. Decaffeinated coffee is made by using solvents or supercritical carbon dioxide to remove the caffeine from the beans. In the chemistry lab, liquid-liquid extraction is the most prevalent process. This occurs in a separatory funnel and involves the placement of a solution that contains the components that need to be removed. An immiscible solvent is then added, creating two layers which are shook together. Normally, one layer is aqueous and the other is an organic solvent. The components are extracted from one layer to the other. The shape of the separatory funnel is conducive to the efficient drainage and division of the two layers.
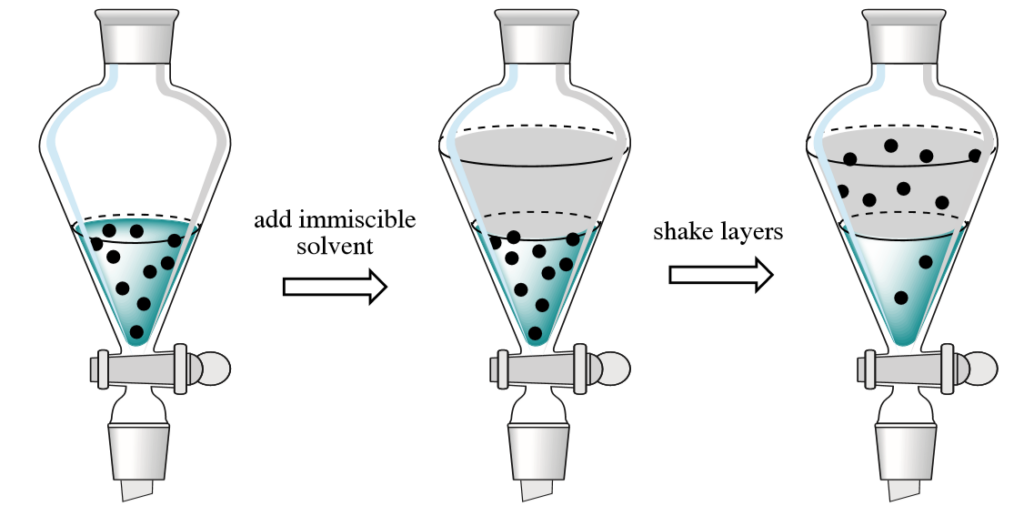
Substances migrate between liquids according to their comparative solubility in each liquid. A simple rule of thumb regarding solubility is the “like dissolves like” principle, which postulates that non-polar compounds will easily dissolve in non-polar solvents (and likewise for polar compounds). As the compounds that give tea its taste and color are polar, they are easily extracted into hot water. Given the chance to reach equilibrium between two liquids in a separatory funnel, most of a compound will generally end up in the layer in which it is more soluble.
Step-by-Step Extractions Procedures
Single Extraction
This section displays an illustration of extracting methyl red (hued element, Fig. 3) from an aqueous solution (lower level) into 25 ml of ethyl acetate (upper level). The aqueous solution was initially a pink tinge, since methyl red exhibits red in acidic solution (the aqueous solution was formed from 50 ml water, 5 drops of 0.1M HCl and 5 drops of 1% methyl red indicator solution). The methyl red has an extensive partition coefficient and is drawn out from the aqueous layer into the ethyl acetate in this undertaking.

Prepare the Setup (for single extraction)
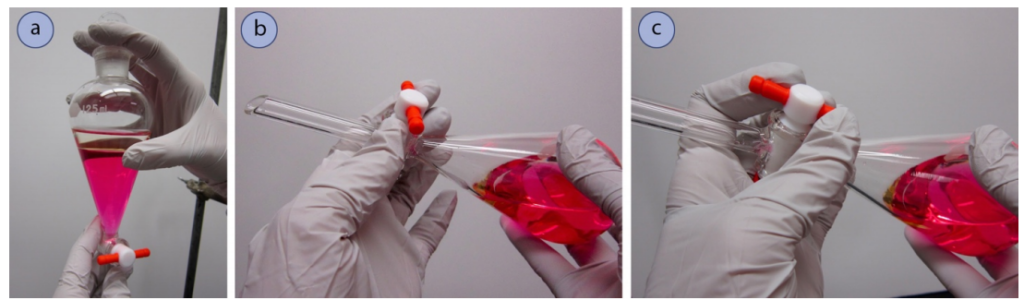
1. Get a separatory funnel (Fig.4 a).
a) If the separatory funnel has a Teflon stopcock, reassemble the stopcock if it was taken apart for drying, putting the elements into the correct arrangement (Fig.4 b). Make sure the Teflon stopcock is properly tightened up so that it can still be rotated with ease, but not too loose that liquid can trickle around the joint.
b) If you are utilizing a glass stopcock (Fig.4 c), it may not require any further preparation. There should be a very thin film of grease used to secure the stopcock and inhibit freezing. In the event that both glass and Teflon stopcocks can be obtained, Teflon is a more suitable choice, given that there is always a probability that solvent can dissolve the grease utilized with glass stopcocks and sully the sample.
c) Furthermore, get a stopper (Teflon or ground glass) that fits snugly into the top joint of the funnel (Fig.4 a).
2. Secure the separatory funnel to a ring stand or frame using a ring clamp. The funnels are fragile, so protect it in the metal clamp by inserting sections of cut rubber or plastic tubing (Fig. 4 d).

Add the Solutions (for single extraction)
3. Before introducing anything into a separatory funnel, guarantee that the stopcock is in the “closed” position, with the stopcock horizontal (Fig.5 a). To be sure, always put an Erlenmeyer flask underneath the separatory funnel before introducing (Fig.5 b). This can catch any liquid if the stopcock is by chance left open, or if the stopcock is loose and liquid seeps out inadvertently.
4. Utilize a funnel to pour the fluid to be isolated into the separatory funnel (Fig.5 b). A separatory funnel should never be used with a hot or warm liquid. The ground glass joint on top of a separatory funnel is more likely to stick to the stopper if liquid was in the joint at some point. Introducing liquid into the separatory funnel using a short-stemmed funnel avoids getting the joint wet, thus making it unlikely to freeze during mixing.

Add a volume of the extracting fluid to the separatory funnel, as indicated in the method (Fig.5 c). It is not necessary to utilize precise amounts of solvent for extractions, and the amounts can be calculated in a graduated cylinder. If a process requires 20mL of solvent, it is okay if between 20-25ml is employed each time.
Mix the Solutions (for single extraction)
6. Securely affix the plug to the spout and hold the spout in such a way that one hand firmly covers the stopper, while the other hand grasps the base of the funnel (Fig.6 a).
7. Carefully turn the funnel upside-down (Fig.6 b), and gently swirl the combination a bit. While it is not rare for some liquid to seep into the ground glass joint after being flipped, it should be minimal. In case liquid drips on to your fingers or gloves when you flip the funnel, it is very likely that the stopper is too large.
8. Pressure can accumulate in the separatory funnel when mixtures are blended, so after swirling, and with the funnel still inverted, “release” the funnel quickly by briefly opening the stopcock to permit a decrease in pressure (Fig. 6 c). Pressure increases in the funnel as solvent evaporates into the headspace and provides additional vapor to the starting ∼1 atmosphere of air pressure in the funnel. With highly volatile solvents (such as diethyl ether), a clear “swoosh” can be heard when releasing, and small amounts of liquid may even sputter out the stopcock. If liquid spurts out the stopcock, try to let it to drain back into the funnel. The noise associated with releasing usually stops after the second or third inversions, as the headspace becomes saturated with solvent vapors and the pressures inside and outside the funnel are equalized.
Safety note: Never point the stopcock towards someone as you release, as it is possible some liquid may splash on him.
9. Shut the stopcock and blend the mixtures a bit more vigorously, occasionally halting to vent the system. There are differences of opinion on how intensely mixtures should be combined in separatory funnels, and for how long. As a general guide, a mild mixing for 10-20 seconds should be enough. With some solutions (e.g. dichloromethane), attention should be taken to not shake too energetically, as these solutions often form emulsions (where the boundary between the solutions does not clarify). With solutions prone to emulsions, a funnel should be softly rocked for one minute.
10. Put the separatory funnel upright in the ring clamp to enable the layers to completely separate. The interface between the layers should settle rather speedily, often within 10 seconds or so. If the boundary is foggy or not clearly defined (an emulsion has shaped), see the troubleshooting section for tips.
Separate the Layers (for single extraction)
11. If the stopper is kept in place, liquid will not be able to come out of the separatory funnel efficiently as no air will be able to come in to fill the space left by the liquid. Should the liquid leave without being replaced by an equivalent amount of air, a vacuum effect would be created. So, prior to draining the liquid from the separatory funnel, the stopper should be taken out (Fig.7 a).
12. The majority of the bottom layer should be poured into a pristine Erlenmeyer flask, with the ring clamp fixed in such a way that the end of the separatory funnel is placed inside the Erlenmeyer flask to avoid splashing (Fig.7 b). Stop pouring when the interface is close to 1 cm from the lower part of the stopcock.
13. Carefully agitate the funnel to remove any droplets stuck to the sides of the glass (Fig.7 c). If necessary, a glass stirring rod can be employed to remove any droplets that are hard to remove.
14. Then, drain the bottom layer further, ceasing when the interface just enters the opening of the stopcock chamber (Fig.7 d). Label the Erlenmeyer flask (e.g. “bottom layer”).

15. Carefully transfer the top layer from the top of the separatory funnel into a different clean Erlenmeyer flask (Fig.8 a), remembering to label the flask (Fig.8 b). To ensure minimal mixing of the liquids, it is best practice to release the bottom layer through the stopcock, and to pour out the top layer from the top of the funnel.

16. Never discard any liquids from a separation prior to being completely sure that the sought-after element is present. Undesired elements can be disposed of once the desired one is in possession (e.g. after the rotary evaporator has eliminated the solvent). Blunders made during separations (e.g. continuing with the wrong layer), can be remedied provided the liquids have not been placed in the trash can! The layers should also be preserved until after evaporation due to the fact that the desired compound may not be very soluble in the solvent utilized. If the substance failed to be extracted with one solvent, a diverse solvent could be tried later on, once more only if the layers had not been discarded.
Cleaning (for single separation)
To clean a separatory funnel, start by rinsing it with acetone into a waste container. Then clean the funnel with soap and water at your work area. Take apart the Teflon stopcock (if employed). After rinsing with distilled water, enable the parts to dry apart in your locker (Fig.8 c).
Multiple Extractions
In this part, step-by-step directions are provided for separating an aqueous solution with an organic solvent which has a lower density than water (resulting in the organic layer on top). As an example, the instructions are written for extracting an aqueous solution three times with 25 ml of diethyl ether each time (3×25 ml diethyl ether). An outline of the initial two extractions is shown in Fig. 9 two extractions when the organic layer is on the top.
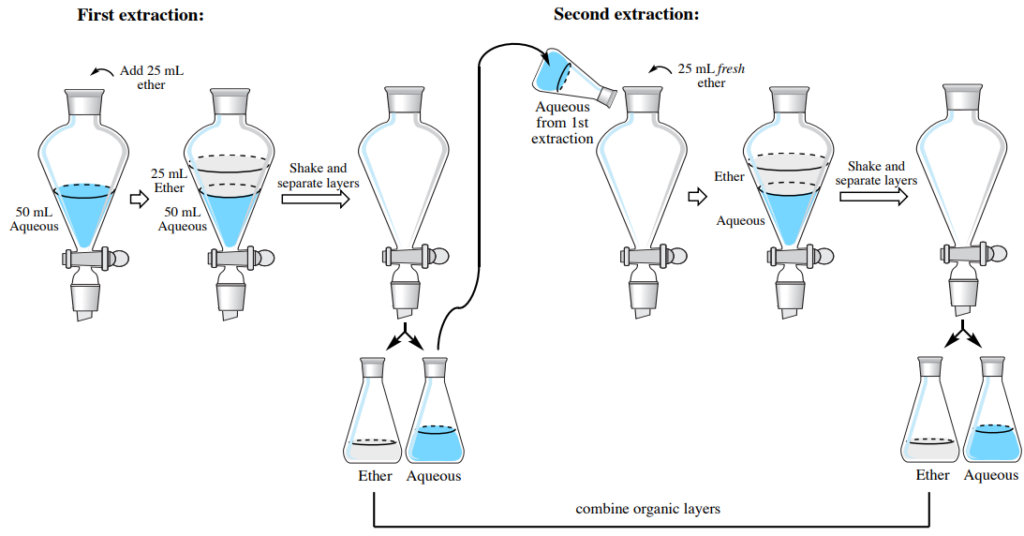
Fig.9 Two extractions when the organic layer is top
Extraction #1
1. Carry out one separation utilizing close to 25 ml of diethyl ether (an exact amount is not mandatory), as demonstrated before, making sure to label each layer properly (e.g. “upper organic layer” and “lower aqueous layer”).
Extraction #2
2. Return the liquid from the separatory funnel to its original flask. There is no need to wash the funnel between extractions.
3. Introduce 25 ml of fresh diethyl ether to the separatory funnel. Seal the funnel, invert and shake with venting, then let the layers to divide.
At this point, the separatory funnel should contain two layers. If this is not the situation, it is likely that the wrong layer was included in the funnel in step 2 (a regular error). To determine if this was the mistake, a little water from a spray bottle can be added. If the layer returned to the funnel is the organic layer (incorrect), the water from the spray bottle will not blend with the solution, but will rather drop as droplets to the bottom. If the organic layer (incorrect) was mistakenly returned to the separatory funnel, nothing is wrong, as the organic layer was simply diluted. Pour the liquid back to the flask assigned for the organic layer, and instead add the aqueous solution to the funnel.
4. Pour the aqueous layer from the bottom of the separatory funnel into an Erlenmeyer flask: it is acceptable to use the same flask that was used for the aqueous layer in the initial extraction (that may have been labeled “bottom aqueous layer”).
5. Since it is the most popular to combine the organic layers in multiple extractions, the top organic layer can be emptied from the separatory funnel into the same flask that was used for the organic layer in the primary extraction (that may have been labeled “top organic layer”). This flask should contain approximately 50 ml of diethyl ether from the two extractions.
Extraction #3
6. For a third time, put the aqueous layer from the second extraction into the separatory funnel, then add another 25 ml of fresh diethyl ether. Put the stopper on the funnel, turn it upside down, shake it with venting, then let the layers divide.
7. Pour the aqueous layer into the right flask, and pour the upper layer into the organic layer flask, where there should be approximately 75 ml of diethyl ether from the three extractions.
Troubleshooting
This section outlines typical difficulties and solutions when performing extractions.
A Common Cause of Only One Layer
The most probable cause of having a single layer in a separatory funnel instead of two (the expected outcome when the procedure directs to “separate the layers”), is an error. It is likely that the incorrect layer was put into the separatory funnel – for example, the organic layer was inadvertently added in place of the aqueous layer. When organic solvent is added to an organic layer in the separatory funnel, the result is a single layer. If the right layer is added to the funnel, it will go as planned. To avoid this mistake in the future, make sure to label the Erlenmeyer flasks. Also, be sure to never discard a layer until you are certain that you have done everything correctly.
An occasional reason that only one layer appears in a separatory funnel is if there are significant amounts of compounds present that are soluble in both solvents, for example if large amounts of ethanol are present, which dissolve well in both aqueous and organic solvents. In this case, the best approach is to separate the troublesome compound (i.e. the ethanol) on a rotary evaporator prior to extraction.
Three Layers Occurring
The most widespread source of three layers in a separatory funnel is inadequate mixing (Fig.10 a). If the funnel is shook more energetically, it will likely form two layers (Fig.10 b). It is also feasible that a third intermediate layer is an emulsion, in which the two layers are not completely separated.

There is Material that cannot be Dissolved at the Boundary
It is not unusual for a thin, insoluble layer to form between two layers during an extraction. Polymeric substances tend to accumulate at the interface due to the decreased solvent interactions. This is nothing to fret about, as a little residue in the organic layer can usually be taken away through a drying and filtration process.
The Interface is Invisible
Occasionally, the materials in a separating funnel are so murky that they make it hard to identify the boundary between the two layers. If that is the case, there are various tactics that may come in handy. You can hold the collecting vessel up to a source of light or use a torch to beam some light on the glass (Fig.11 b). This additional luminosity may make it easier to detect the interface. Another option is to vigilantly watch the layers as you slightly tilt the beaker from side to side (Fig.11 c). Your eye may be able to spot minor differences in the way the liquids move. The third strategy is to put a bit more liquid into the vessel to weaken one of the layers, or to add a different solvent to modify the refractive index.

The Layers Are Poorly Divided (An Emulsion Was Created)
Emulsions are when tiny droplets of one layer are suspended in the other layer, resulting in no distinct interface between the two layers (Fig.12). Often an emulsion looks like a bubbly mess near the interface, and can even appear to be an odd-looking third layer.

Emulsions can be caused by a number of factors:
1. The density of both layers may be so comparable that there is no impetus for them to separate.
2. Soap-like compounds or other emulsifying agents may be present that enable the liquids to mix.
Avoiding emulsions is best done by shaking solutions that are likely to create them (such as dichloromethane with highly basic or dense solutions) gently in a separatory funnel. However, if an emulsion is formed, there are several methods to clear it:
a) Gently agitate the layers and use a glass stirring rod to attempt to break down any suspended droplets.
b) Let the solution sit, if possible, for a period of time. Sometimes, with enough time, the emulsion will separate by itself.
c) For smaller volumes, use a centrifuge if available. The centrifuge will speed up the process of allowing the emulsion to settle. Make sure the centrifuge is balanced, as an unbalanced one may slip off the bench top. Put tubes of equal size in the centrifuge on opposite sides.
d) If the two layers have comparable densities, change their densities to make the separation easier. To remove the emulsion, try to reduce the density of the top layer or increase the density of the bottom layer. For instance, if the emulsion occurs with ethyl acetate (top layer) and an aqueous solution (bottom layer), add some NaCl, which will dissolve in the aqueous layer and raise the density of the aqueous solution. Alternatively, add more ethyl acetate, which will dilute the organic layer and reduce its density. As a last resort, add some pentane, which will mix with the upper organic layer and lower its density (pentane is one of the least dense organic solvents). The addition of pentane should be used as a last resort because it will limit the organic layer’s ability to extract some polar compounds.
If the emulsion is formed with an aqueous solution (top layer) and dichloromethane (bottom layer), add some water from a squirt bottle to thin out the top layer and lower its density. This method worked well to clarify the emulsion in Fig 13 c, as seen in Fig 13 d.
e) Reduce the solubility of one component in the other. One method is to add NaCl or NH4Cl to the separatory funnel, which will dissolve in the aqueous layer and decrease the capacity of organic compounds to dissolve in water (“salting out”).

