- The process of creating a drug - May 23, 2023
- How do scientists model mental disorders in animals? - May 23, 2023
Introduction. What are Grignard Reagents?
Grignard reagents are a class of organic compounds consisting of alkyl- or aryl-magnesium halogens (e.g. methyl-, ethyl-, butyl-, phenyl-, bromo- and iodide-magnesium reagents) and metal salts (usually halogens) of other elements (e.g. copper, zinc, iron, etc.). Grignard reagents are named after Franz Grignard, the German chemist who first described this type of organic compound in 1900.
Grignard reagents are widely used in organic chemistry, especially in the synthesis of complex organic molecules. They can be used to add alkyl or aryl groups to ketones, aldehydes, carboxylic acids, ether and other compounds. These reactions can be very useful as they allow the synthesis of complex organic molecules from simpler precursors.
Grignard reagents are also used as catalysts, in the synthesis of some polymers and in other industrial research. However, Grignard reagents are extremely reactive and pyrophoric, i.e. they can ignite on contact with air or water, therefore require special conditions for storage and use, as well as safety precautions when handling them.
History of the Grignard Reagents Development
The history of the development of Grignard reagents began with the work of Franz Grignard, a German chemist, who first described this type of organic compound in 1900. In his work, Grignard described a method for producing magnesium halogens (chlorides, bromides and iodides) and using them for synthesizing new organic compounds such as alkenes, alkanes, ketones and aldehydes. This method became known as the “Grignard reaction”.
In the 1950s, Grignard reagents were widely used in the synthesis of organic compounds, leading to a significant development in organic chemistry. Today, Grignard reagents remain an important tool for the synthesis of complex organic molecules and continue to attract the attention of chemists around the world because of their potential applications in medicine, electronics and other industries.
Basic Principles of Grignard Reagents
The basic principle behind the reactivity of Grignard reagents is their ability to act as strong nucleophiles due to the polarized carbon-magnesium bond. The carbon atom of the alkyl or aryl group in the Grignard reagent is negatively charged due to the electronegativity difference between carbon and magnesium. As a result, the carbon atom becomes highly reactive and can attack electrophiles, such as carbonyl compounds, to form new carbon-carbon bonds.
Another important principle of Grignard reagents is their high sensitivity to moisture and air. The polarized carbon-magnesium bond is very reactive towards water and oxygen, which can lead to the decomposition of the Grignard reagent and the formation of undesirable byproducts.
Grignard reagents also exhibit strong basicity due to the presence of the negatively charged carbon atom. This can lead to the deprotonation of acidic compounds, such as alcohols or carboxylic acids, to form alkoxides or carboxylates. The resulting alkoxides or carboxylates can then react with electrophiles to form new carbon-carbon bonds.
Overall, the basic principles of Grignard reagents involve their strong nucleophilicity, sensitivity to moisture and air, and strong basicity. These properties make Grignard reagents valuable tools in organic synthesis for the formation of new carbon-carbon bonds and the synthesis of a wide variety of organic compounds.
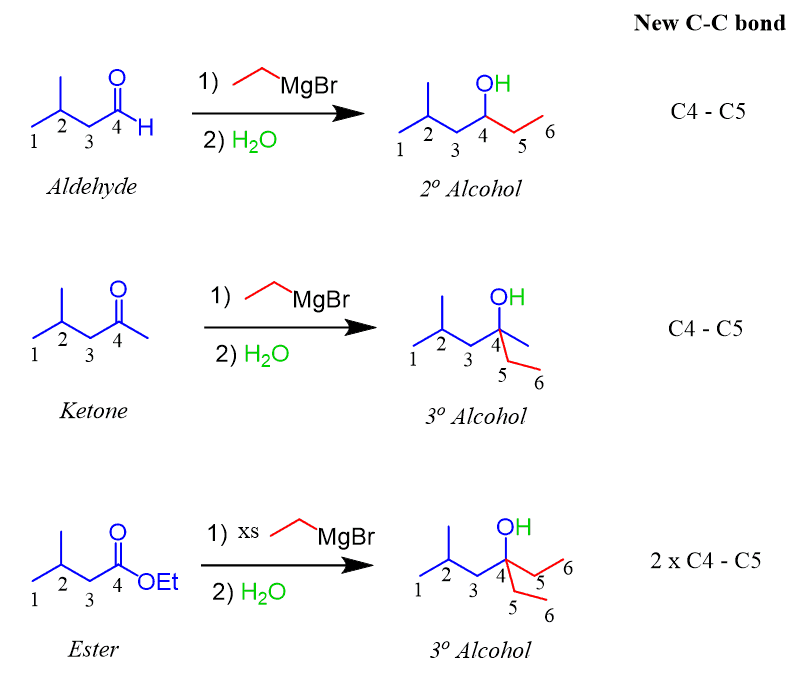
Structure of Grignard Reagents
Grignard reagents are magnesium halogens of the general formula RMgX, where R is an organic radical and X is a halogen (usually bromine or chlorine). The structure of Grignard reagents has a complex multi-dimensional structure.
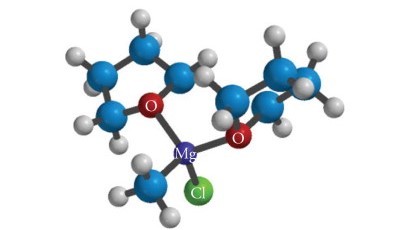
The magnesium ion (Mg2+) has an octahedral geometry, surrounded by six ether or THF molecules. The magnesium ions combine with each other through bromide or chloride ions to form dimers. Thus, each Grignard reagent contains two RMgX groups bonded through a magnesium ion.
Each RMgX group consists of radical R bonded to magnesium through a covalent bond and halogen X bonded to magnesium through an ionic bond. The radical R can be any organic radical, such as an alkyl, an aromatic radical, or a functional group such as a carbonyl group.
Mechanisms of Reactions with Grignard Reagents
Grignard reagents react with a wide range of organic compounds to form new bonds. The mechanisms of reactions with Grignard reagents depend on the nature of the reagent and reagent, as well as the reaction conditions.
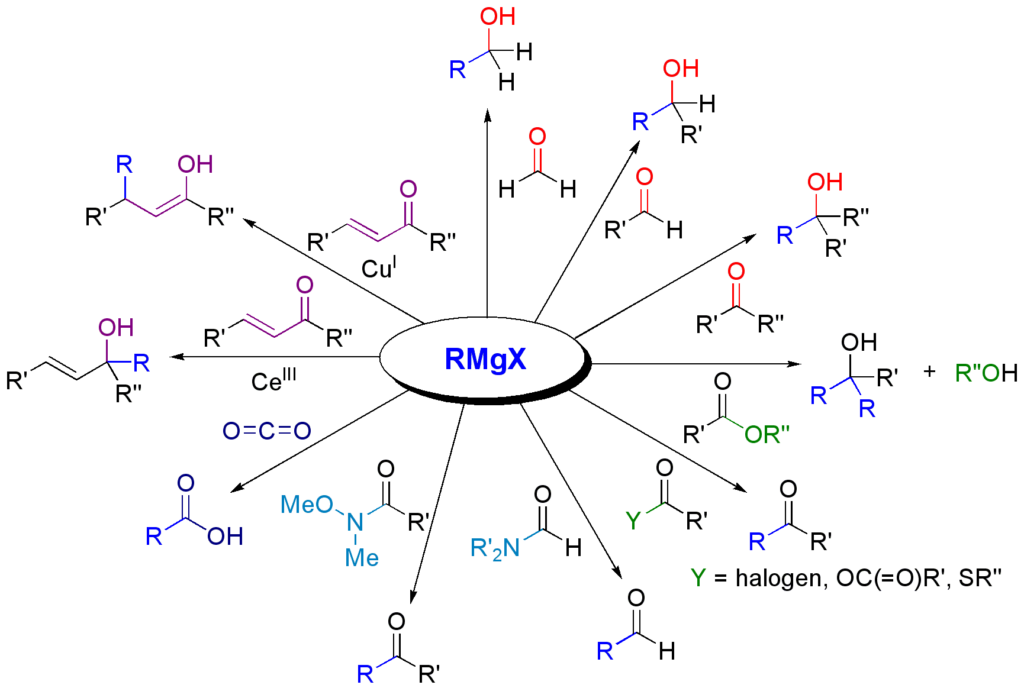
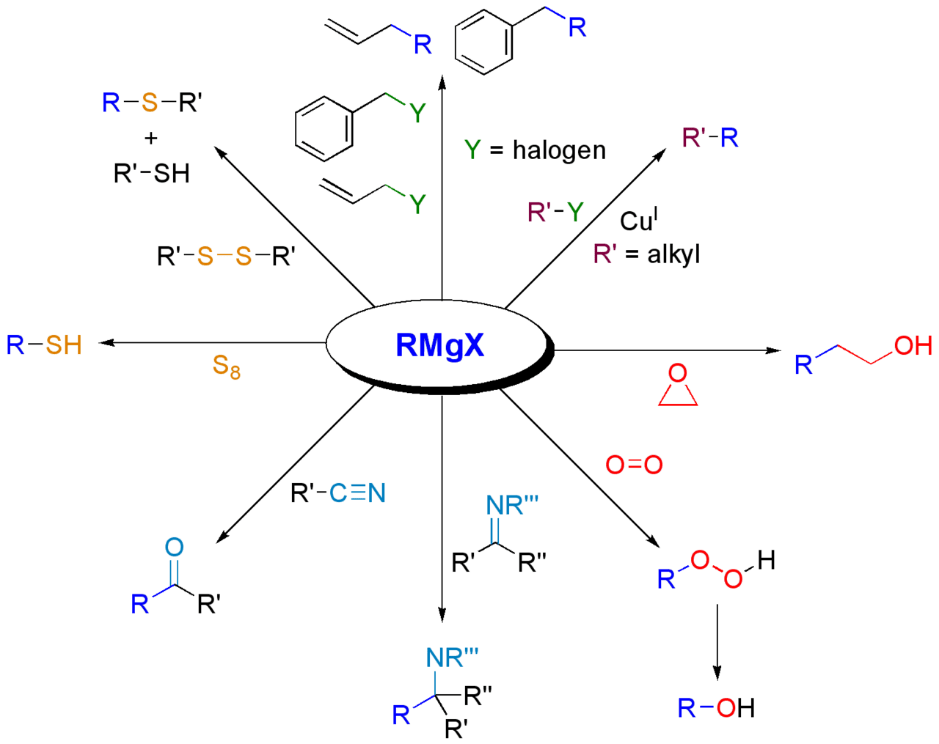
One of the most common reaction mechanisms of Grignard reagents is nucleophilic substitution, where the Grignard reagent attacks the electrophilic functional group in the organic compound. For example, a Grignard reagent can react with a ketone to form an alcohol:
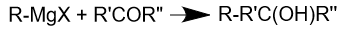
Another reaction mechanism with Grignard reagents is the addition to the double bond in an organic compound, forming a new single bond. For example, a Grignard reagent can react with an alkene to form an alcohol:
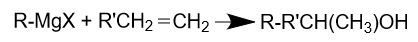
Grignard reagents can also react with halogens to form halogens:

It is important to note that reactions with Grignard reagents are stereoselective, i.e. a certain isomer can form with a higher degree of preference.
Application of Grignard Reagents
Synthesis of Organic Compounds
Grignard reagents are widely used for the synthesis of organic compounds. These compounds can have different functional groups, such as alkenes, amines, alcohols and carbonyl compounds.
The process of synthesizing organic compounds using Grignard reagents consists of reacting a Grignard reagent (usually an alkyl or aryl-magnesium halogen) with an organic compound containing an electrophilic functional group, such as a carbonyl group. The reaction leads to the formation of a new bond between molecules and the formation of complex organic compounds.
Synthesis of organic compounds using Grignard reagents can be used in many applications, including pharmaceuticals, plastics and polymers, catalysts and dyes, synthesis of new materials, etc.
Catalysts and Semiconductors
In addition to the synthesis of organic compounds, Grignard reagents are also widely used as catalysts and precursors for semiconductor materials.
The catalysts obtained with Grignard reagents can be used to activate various chemical reactions such as polymerization and oxidation. For example, nickel-based catalysts produced with Grignard reagents can be used in the manufacture of plastics and rubber products.
Grignard reagents are also used in the production of semiconductor materials to produce optoelectronic devices such as lasers and photodiodes or materials used in the production of solar cells, microelectronic devices and transistors.
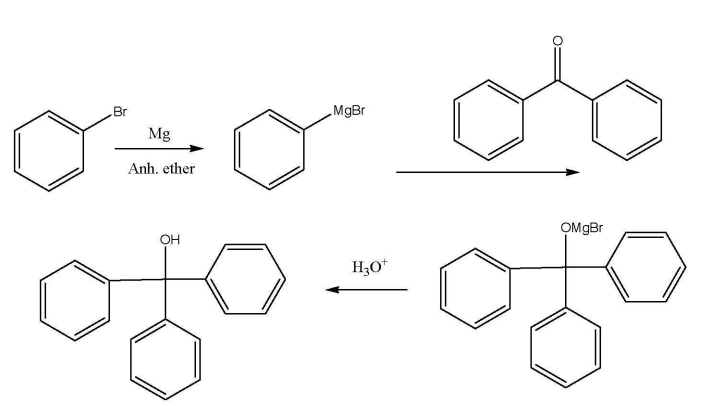
Example: Grignard Reaction, Synthesis of Triphenylmethanol
Generation of the Grignard Reagent:
- Assemble the 5mL conical vial, the Claisen head, and the Drierite-charged drying tube.
- Flask1: Fill up with dry diethyl ether.
- Flask 2: Fill Flask #2 with 76µl of bromobenzene (Approximate the volume assuming that 20 drops equals 1mL in order to minimize/avoid inhaling the vapors and adding water). Add 0.5mL of ether from flask 1. Cap and mix the contents.
- Flask 3: Using a pair of tweezers place 3 pieces of dried magnesium turnings (in excess) in flask 3. Add 100µl of the solution from flask 2, followed by a small grain of iodine (serving as a kind of catalyst, one chemical method to initiate sluggish Grignard reactions). At this point, the magnesium turnings should be covered by the added solution. If no, add enough ether to cover the magnesium, but not too much or it will be too dilute.
- With the dried stirring rod press gently on the magnesium turnings in order to break their surface and to expose the fresh, unoxidized magnesium metal to the reaction mixture.
- Immediately assemble the apparatus and start to heat it gently. Before adding the remaining bromobenzene solution, you should observe signs of reaction initiation in the flask 3. These signs include appearance of bubbles on the Mg surface, cloudiness and disappearance of the purple iodine color.
- NOTE: Since ether is a low-boiling solvent, it is important to keep the reaction mixture from running dry of ether. Keep a close eye on the level of the reaction mixture and replenish any lost ether by adding more fresh ether from flask 1 with needle. (Do not open the rubber cap). On the other hand, adding too much ether may drastically lower the concentrations of the reacting species and slow down or stop the reaction.
- After the reaction is initiated, quickly add the spin vane in order to have constant stirring and then add the remainder of the bromobenzene solution drop-wise while stirring and gently heating.
- After the addition of bromobenzene is complete, transfer 300 µL of dry diethyl ether into flask 2, mix and dissolve any leftover bromobenzene and add it to the reaction flask. Keep stirring for 5 more minutes.
II. Addition of Benzophenone: - Prepare a solution of 105 mg of benzophenone (you can use the same needle that you have been using, no need of rinsing) in 300µL of diethyl ether in flask 4.
- When the 5 minutes of stirring (from part 1) is completed, add the benzophenone solution to the reaction over a 30 second-period, drop- wise (reaction is very quick. You should add it slowly. Don’t get too hot). The rate of addition should be such that a gentle reflux is never exceeded (you should not see bubbling).
- Dissolve any remaining benzophenone with 300µL of dried ether and add it to the reaction flask to increase the yield. Keep stirring for 5-10 minutes. Detach the reaction flask and let cool.
- Add 1.5 ml aqueous HCl drop-wise and stir to neutralize the solution and make alcohol. (If you don’t have enough to see two layers, add more ether). Cap, shake and let settle. Separate the two layers.
- Extract any residual amount of the organic compounds from the aqueous layer by using two 0.5ml portions of ether. Do back extraction to collect triphenylmethanol which was very slightly soluble in water due to its –OH group. Combine all the organic layers and dispose of the aqueous layer.
- Dry the organic layer using sodium sulfate. Evaporate all of the solvent over a hot plate.
- At this point you are be left with a mixture of triphenylmethanol and biphenyl.
- In order to separate these: Pour 10ml of petroleum ether on the solid mixture. It will dissolve the biphenyl but not the triphenylmethanol. Decant the liquid & rinse the solids with another 2ml of petroleum ether.
Special Features of Working with Grignard Reagents
Grignard reagents can be hazardous to humans and require special care when working with them. They can be toxic and easily flammable in air, so special precautions are necessary.
When working with Grignard reagents, laboratory safety rules have to be performed, such as the use of laboratory gloves, protective glasses and robe, and working in a well-ventilated room. Contact of Grignard reagents with oxygen from the air must also be avoided, which may cause them to ignite.
When storing Grignard reagents certain storage conditions must be observed, such as storage in closed containers, separate from other chemicals, at a certain temperature and under conditions that exclude the possibility of ignition. Grignard reagents can be hazardous to the environment.
Conclusion
In conclusion, Grignard reagents are an important tool in modern organic chemistry and have a wide range of applications in various fields, including synthesis of organic compounds, catalysis and semiconductors as well as medicine.
However, working with Grignard reagents requires special attention to safety and adherence to the rules of laboratory practice. These reagents are toxic, flammable, air- and moisture-sensitive, therefore storage, transport and disposal regulations must be observed.
The use of Grignard reagents can greatly expand the possibilities for the synthesis of organic compounds, facilitate the preparation of many industrial and scientific products and accelerate the development of many branches of science and technology.
Bibliography
- The Grignard Reagents Dietmar Seyferth Organometallics 2009, 28, 6, 1598–1605 Publication Date: March 16, 2009 American Chemical Society URL: https://doi.org/10.1021/om900088z
- Mechanism of Grignard reagent formation. The surface nature of the reaction H. M. Walborsky Acc. Chem. Res. 1990, 23, 9, 286–293 Publication Date: September 1, 1990 URL: https://doi.org/10.1021/ar00177a004
- Handbook of Grignard Reagents Gary S. Silverman, Philip E. Rakita CRC Press, 25.04.1996 Pages: 736 URL: https://books.google.ru/books?hl=en&lr=&id=82CaxfY-uNkC&oi=&redir_esc=y
- Grignard reagent formation John F. Garst, Manuel P. Soriaga, Coordination Chemistry Reviews Volume 248, Issues 7–8, April 2004, Pages 623-652 URL: https://www.sciencedirect.com/science/article/abs/pii/S0010854504000335
- The constitution of the Grignard reagent A. D. Vreugdenhil, C. Blomberg Recueil des Travaux Chimiques des Pays-Bas Volume 82, Issue 5 1963 Pages 453-460 URL: https://doi.org/10.1002/recl.19630820505

