- The process of creating a drug - May 23, 2023
- How do scientists model mental disorders in animals? - May 23, 2023
Introduction
Isomerism is an essential concept of the organic chemistry. The ability of carbon atoms to form four covalent bonds, including those with other carbon atoms, reveals the possibility of the existence of several different compounds with the same elemental composition. Such compounds are called isomers. All isomers are divided into two large classes – structural isomers and stereoisomers.
Structural isomers correspond to different organic compounds structures with different sequence of atomic bonds and the same composition (same number of atoms of each element in molecular formula).
Stereoisomers are compounds that have the same composition and the same atomic sequence but the different atomic arrangement in space.
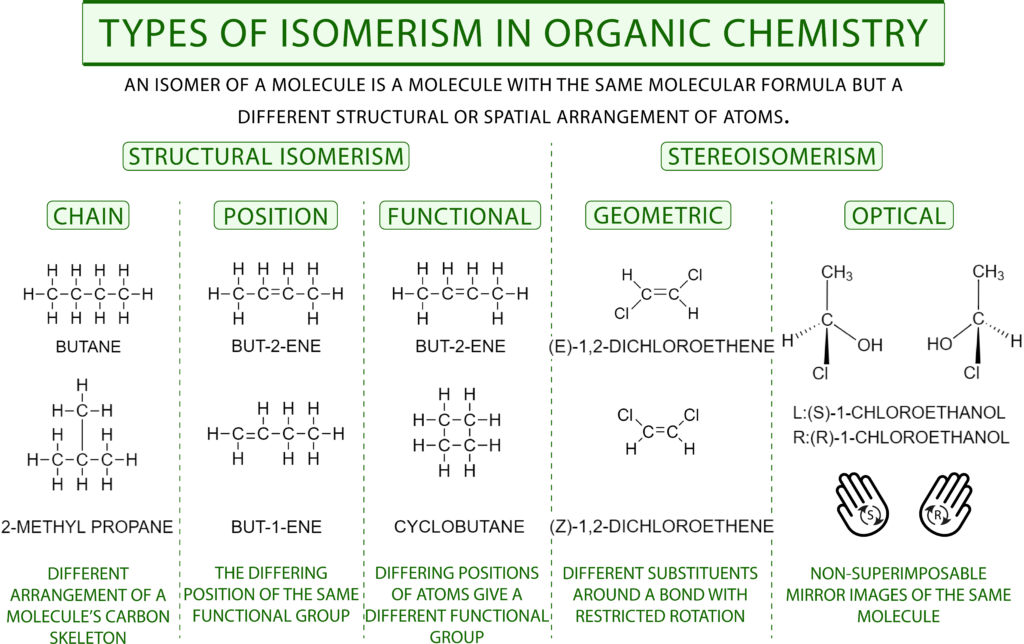
Structural isomers are divided into three groups
1) Functional group isomerism (interclass isomerism)
Compounds that contain different functional groups and belong to different organic compounds classes.

2) Isomerism of the carbon chain (carbon skeleton)
Compounds with different carbon skeleton.
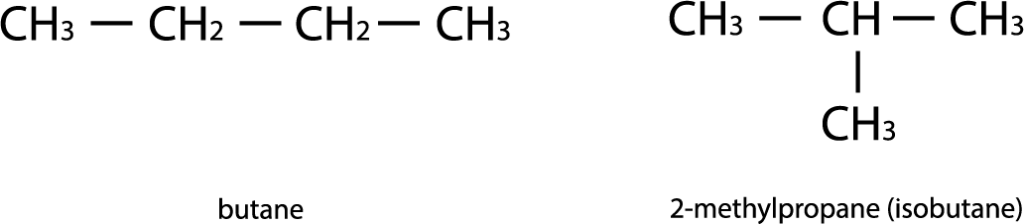
3) Position isomerism
A type of structural isomerism characterized by a difference in the position of the same functional groups or multiple bonds with the same carbon skeleton. Example: 2-chlorobutanoic acid and 4-chlorobutanoic acid.
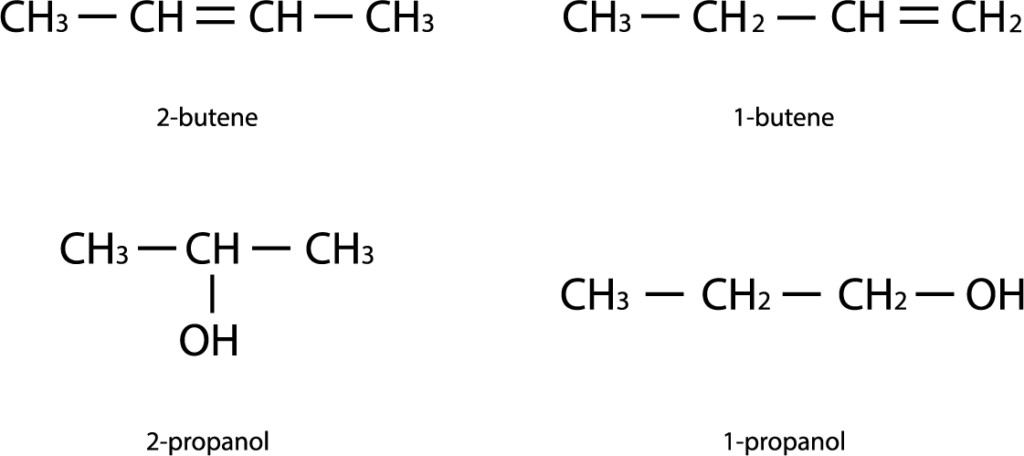
Stereoisomerism
1) Geometric isomerism is typical for compounds containing a double bond or a cycle. It is also possible in such molecules to draw a conventional plane so that the substituents at different carbon atoms could be located on one side (-cis) or on different sides (-trans) in relation to this plane. Geometric isomers are determined by the ability of an orientation changing. If the orientation of these substituents relative to the plane could be changed only by breaking one of the chemical bonds, then the geometric isomerism presents.
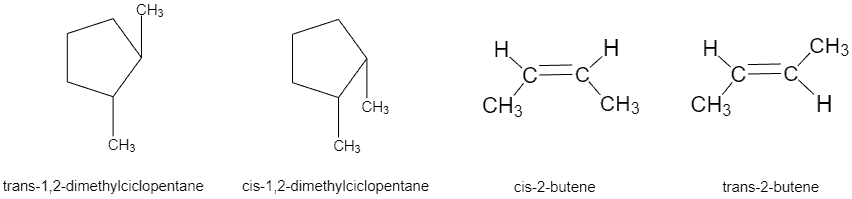
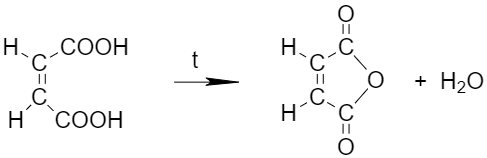
Geometric isomers can differ significantly in their physical and chemical properties. For example, the cis- and trans-isomers of butenedioic acid (maleic and fumaric acids, respectively) behave differently during a heating: maleic acid transforms into a cyclic anhydride, whereas fumaric acid does not form anhydride.
2) Optical isomers are considered to be such compounds which mirror images are incompatible with each other. There are two types of optical isomers: enantiomers and diastereomers. Enantiomerism is a characteristic of molecules that have one asymmetric carbon atom, i.e., an atom bonded to four different atoms or groups of atoms. Molecules of enantiomers relate to each other as a subject and incompatible mirror image.
Enantiomers have the same physical and chemical properties, but different rotation sign of polarized light. Lactic acid is the example of enantiomers:
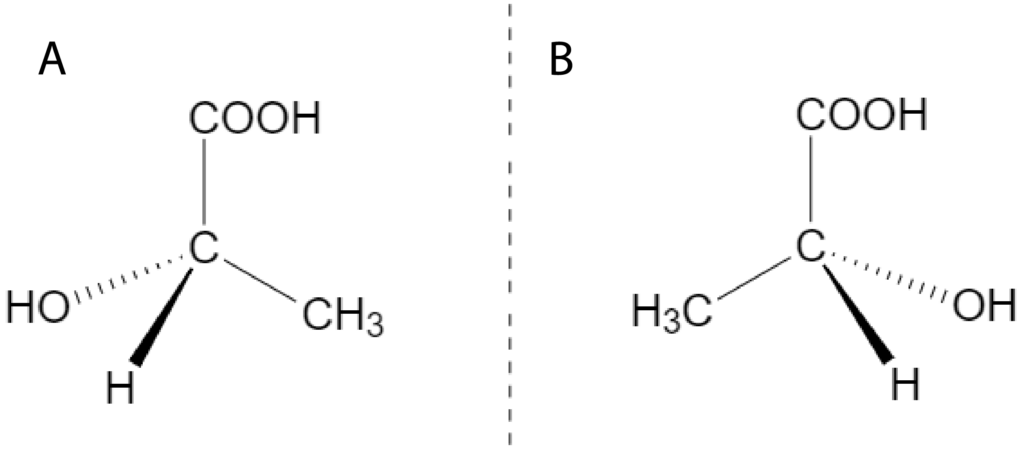
A – Dextrorotary (+) lactic acid that is produced in muscles during physical activity. Is used to be carbohydrate metabolism product.
B – Levorotary (-) lactic acid that is produced by some microorganisms.
Diastereomers molecules are stereoisomers, which not mirror images of each other. Diastereomers have different chemical and physical properties.
