Introduction
Lithium aluminium hydride (lithium alanate or LAH) an important chemical compound due to its properties, reactions, and applications. Being a metal hydride, it has attracted the attention of researchers and industrial chemists. It was discovered by Schlesinger, Finholt, and Bond in 1947.
The chemical activity of lithium aluminum hydride makes it a powerful reducing agent that plays an important role in the synthesis of organic compounds and modification of various materials. Attention should be paid to the variety of reactions in which it participates: from reactions with aldehydes to reactions with aromatic nitro compounds.
In this article we will look at the main characteristics of lithium aluminum hydride, as well as its applications in various fields of science and technology.
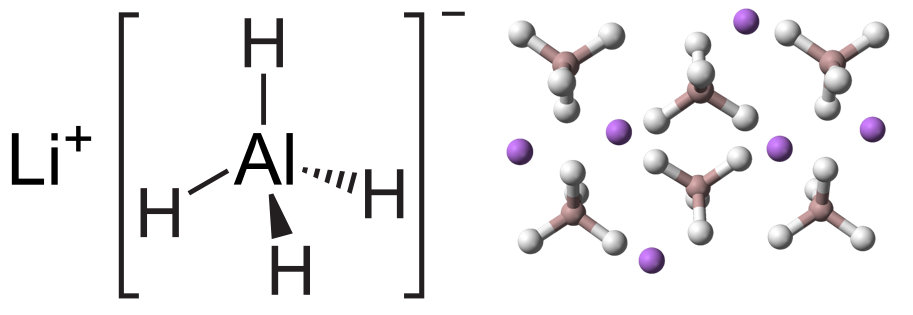
Physical and Chemical Properties of Lithium Aluminium Hydride
Lithium aluminium hydride (LAH) is an inorganic compound with the chemical formula LiAlH₄ and a molecular weight 37.95 g/mol. These are odorless white or gray crystals. Its density is 0.917 g/cm3. Its hygroscopic and reacts with water, releasing hydrogen. It is soluble in tetrahydrofuran – 2.96 mol/L (at 25°C), diethyl ether – 5.92 mol/L (at 25°C), dioxane – 0.03 mol/L (at 25°C).
This compound is a strong reducing agent used in organic synthesis. More potent than other commonly used agents such as sodium borohydride due to weaker Al-H bonds compared to B-H. Reduces esters, carboxylic acids and ketones to alcohols, nitro compounds and amides (as well as glycosamides) to amines. Reactions involving lithium aluminum hydride are usually carried out under anhydrous and inert conditions to avoid unwanted side reactions.
Synthesis of Lithium Aluminium Hydride
The synthesis of lithium aluminium hydride typically involves the reaction between lithium hydride and a metal halide. One common method is the reaction between lithium hydride and aluminum chloride in diethyl ether. This method for the synthesis of lithium aluminum hydride is complex because the reaction has several directions and can lead to the evolution of hydrogen gas at room temperature.

Another method that can be used in production is the initial production of sodium aluminum hydride from elemental substances. Sodium aluminum hydride is produced from the elements under high pressure hydrogen at 200 °C using a triethylaluminum catalyst.
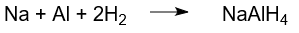
When sodium aluminum hydride suspended in diethyl ether, it reacts with lithium chloride to form lithium aluminum hydride by an exchange reaction between lithium chloride and sodium aluminum hydride.


Chemical Reactions of Lithium Aluminium Hydride
Lithium aluminium hydride is a reducing agent, and its chemical reactions are characterized by its ability to donate hydride ions. The most common and significant reactions involve the reduction of various functional groups in organic compounds. Here are some main chemical reactions of lithium aluminium hydride:
Decomposition of lithium aluminum hydride occurs at temperatures above 125°C with the formation of lithium hydride, aluminum, and hydrogen.
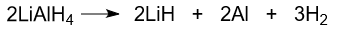
Lithium aluminum hydride reacts with water. As a result of hydrolysis aluminum hydroxide, lithium hydroxide, and hydrogen are formed.
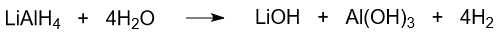
Lithium aluminum hydride reacts with dilute acids in the cold. For example, when dilute hydrochloric acid reacts with lithium aluminum hydride, lithium chloride, aluminum chloride and hydrogen are formed.

Lithium aluminum hydride is widely used to reduce carbonyl compounds such as aldehydes, ketones, carboxylic acids, and esters to the corresponding alcohols. The hydride ion LiAlH₄ attacks the carbonyl carbon, resulting in the formation of alcohol. For example, when lithium aluminum hydride reduces acetaldehyde in the presence of water, ethanol, lithium hydroxide, and aluminum hydroxide are obtained.

Lithium aluminum hydride can reduce nitro compounds to amines. The hydride ion reduces the nitro group to an amino group. For example, 1-phenyl-2-nitropropene can be reduced to amphetamine in 1,3-dioxane.
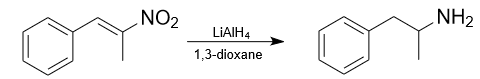
It’s important to note that the reactions involving lithium aluminium hydride are highly exothermic and should be conducted under inert conditions to prevent side reactions. Careful handling and proper safety precautions are essential when working with lithium aluminum hydride.

Applications of Lithium Aluminium Hydride
Lithium aluminum hydride is widely used in organic synthesis. It is an important reagent in the production of pharmaceuticals and agrochemicals. In addition, lithium aluminum hydride is used in the reduction of functional groups in polymers and in the preparation of certain metal hydride compounds. Its role in these processes highlights its importance in the synthesis of complex organic molecules and materials. Here are some examples of its applications:
Reduction in Organic Synthesis:
Lithium aluminum hydride is widely used to reduce carbonyl compounds, including aldehydes, ketones, carboxylic acids, and esters, to their respective alcohols. This process is fundamental in the synthesis of pharmaceuticals, chemicals, and organic intermediates.
Polymerization Reactions:
Lithium aluminum hydride is used to restore functional groups in polymers. For example, for polymethyl methacrylate and polymethyl acrylate.
Preparation of Metal Hydrides:
Lithium aluminum hydride is used in the preparation of various metal hydrides, facilitating the development of materials with unique properties, such as hydrogen storage materials and battery hydrides. For example, lithium hydride.
Reduction of Inorganic Compounds:
Lithium aluminum hydride can reduce certain metal halides to produce the corresponding metal hydrides. This process is significant in the synthesis of specific metal hydride compounds for research and industrial applications.

Health Effects of Lithium Aluminium Hydride
Lithium aluminum hydride is a strong reducing agent and is highly reactive. Although it has important applications in organic synthesis and other fields, it also poses health and safety risks. Here are some health effects associated with exposure to lithium aluminum hydride:
Irritating:
Lithium aluminum hydride may cause irritation to the skin, eyes, and mucous membranes. Contact with skin or eyes may cause irritation, redness, and burns. Inhalation of dust may cause respiratory irritation.
Toxicity:
Lithium aluminum hydride may be toxic if swallowed or inhaled. Symptoms of exposure may include nausea, vomiting, abdominal pain, respiratory distress, and central nervous system damage. In severe cases, exposure can cause serious health effects.
Prolonged or repeated exposure to lithium aluminum hydride may cause allergic reactions in some people, which may include dermatitis or breathing problems.
Flammability and Reactivity with Water:
Lithium aluminum hydride reacts violently with water to produce flammable hydrogen gas. This reaction can be dangerous and may result in thermal burns, fires, or explosions. Special precautions are required to prevent contact with moisture during handling and storage.
Safety Precautions
Handling lithium aluminum hydride requires strict precautions due to its reactivity and potential hazards. Here are some important precautions when working with it:
- Personal Protective Equipment (PPE):
Wear appropriate personal protective equipment, including chemical-resistant gloves, safety goggles, and a lab coat. Depending on the specific procedures, additional protective clothing such as a face shield and fire-resistant clothing may be required. - Ventilation:
Work in a well-ventilated area, preferably under a fume hood, to minimize inhalation exposure. Use exhaust ventilation systems to control the release of vapors and prevent the accumulation of hydrogen gas. - Handling Precautions:
Operate under inert gas conditions (such as argon or nitrogen) to prevent exposure to moisture and oxygen, which may react violently with lithium aluminum hydride. - Storage:
Store lithium aluminum hydride in a cool, dry place, away from incompatible materials. Use designated storage containers and ensure proper labeling.
Compliance with these precautions is necessary to minimize the risks associated with handling lithium aluminum hydride.
Conclusion
In conclusion, lithium aluminum hydride is a chemically important compound with unique reactivity that has found applications in various scientific and industrial fields. Its role as a powerful reducing agent has greatly influenced organic synthesis, facilitating the production of pharmaceuticals and materials.
However, the use of lithium aluminum hydride is associated with certain problems and safety considerations. The high reactivity of the compound requires careful handling under inert conditions to prevent unwanted reactions, especially with moisture and oxygen. Precautionary measures, including the use of personal protective equipment and proper ventilation, are critical to reducing potential health and safety risks.
Bibliography
- Amphetamine synthesis via 1-phenyl-2-nitropropene LAH reduction
Url: https://bbgate.com/threads/amphetamine-synthesis-via-1-phenyl-2-nitropropene-lah-reduction.14/ - Use of Lithium Aluminium Hydride in the Study of Carbohydrates M. ABDEL-AKHER & F. SMITH Nature volume 166, pages1037–1038 16 December 1950
Url: https://doi.org/10.1038/1661037a0 - Hydrogenolysis of carbohydrate acetals, ketals, and cyclic orthoesters with lithium aluminium hydride – aluminium trichloride S. S. Bhattacharjee and P. A. J. Gorin Canadian Journal of Chemistry April 1969
Url: https://doi.org/10.1139/v69-194 - The thermal decomposition of lithium aluminium hydride William Edward Garner and E. W Haycock Royal Society Volume 211 Issue 1106 06 March 1952
Url: https://doi.org/10.1098/rspa.1952.0046 - Reactions of lithium and sodium aluminium hydride with sodium or lithium hydride. Preparation of a new alumino-hydride of lithium and sodium LiNa2AlH6 Pierre Claudy, Bernard Bonnetot, Jean-Pierre Bastide, Jean-Marie Létoffé Materials Research Bulletin Volume 17, Issue 12, December 1982, Pages 1499-1504 26 July 1982
Url: https://doi.org/10.1016/0025-5408(82)90204-5 - Preparation of unsaturated long-chain alcohols by means of lithium aluminium hydride: some typical members of the series S. P. Ligthelm, E. von Rudloff and Donald A. Journal of the Chemical Society Sutton Issue 0, 1950 p.3187-3190
Url: https://doi.org/10.1039/JR9500003187

