- The process of creating a drug - May 23, 2023
- How do scientists model mental disorders in animals? - May 23, 2023
Unlike simple low molecular weight molecules dissolution, a polymer one is different. The main distinctive feature of polymer dissolution is preliminary polymer swelling. This article describes the term “polymer” and discloses peculiar properties of polymer dissolution.
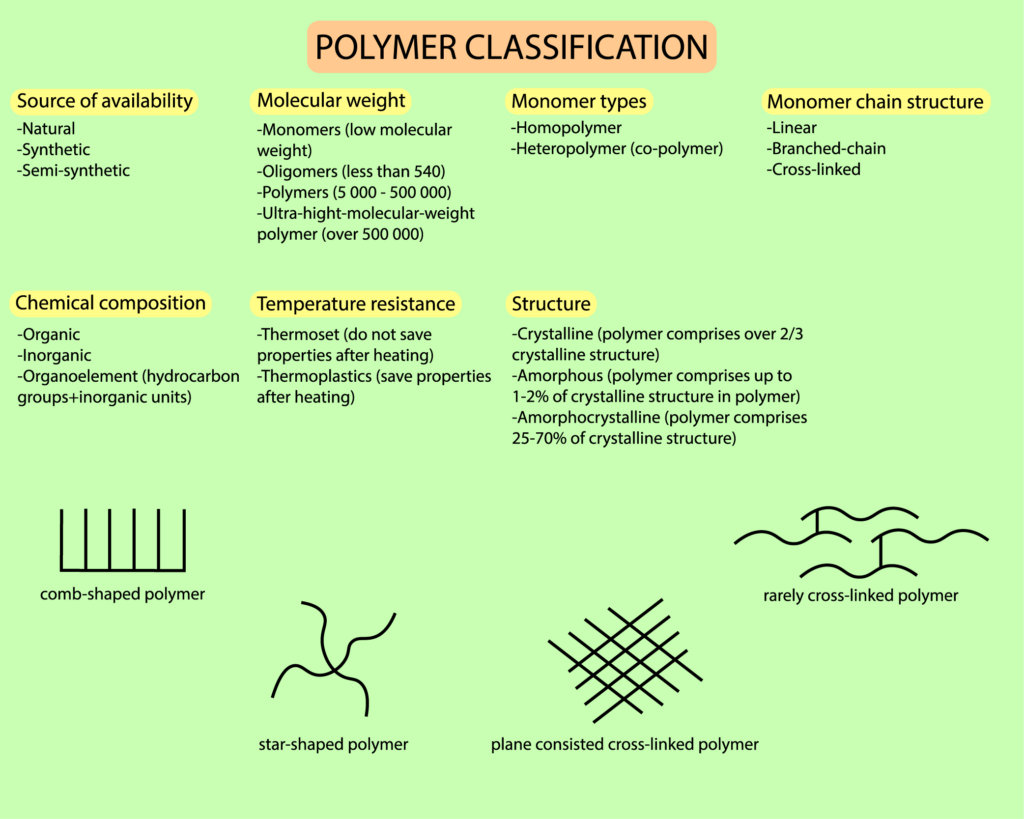
A polymer is a high molecular weight (several thousand to several million) compound that is comprised of a large number of repeating sub-units (atom groups). Units can have identical or different structure and are represented by building blocks linked together by chemical bonds. A polymer structure can exist as a linear chain (e.g. cellulose), branched chain (e.g. amylopectin) and three-dimensional one. Physical and chemical properties of polymers are largely determined by its chemical structure.
Polymer Сlassification
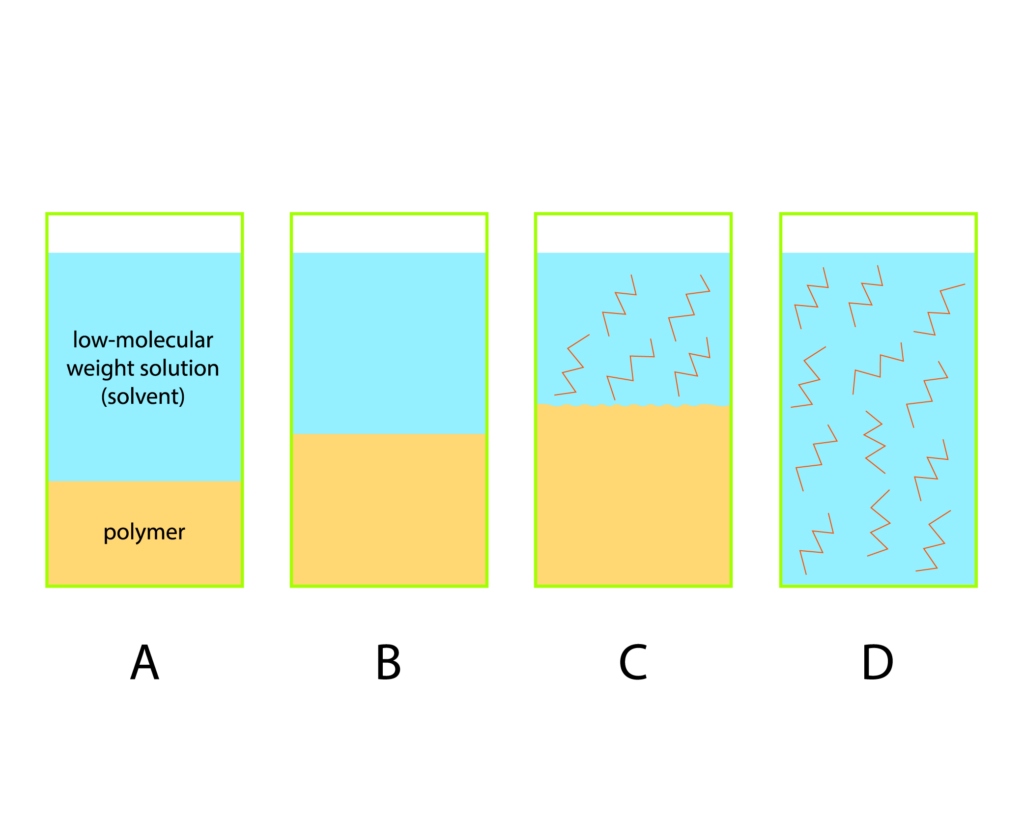
Polymer Dissolution
1. At the initial stage the system consists of two components: a polymer and a low-molecular-weight solution.
2. Transition a → b is characterized by intensive penetration of low molecular weight molecules into the polymer structure and the polymer chain salvation (accompanied by heat release). The polymer volume increases but the total volume of the polymer-solvent system decreases. This phenomenon is called contraction.
3. Transition b → c represents the initial stage of polymer macro-molecule distribution throughout the solvent volume and is characterized by a system entropy increase due to a possible conformations number increase.
Limited swelling finishes at stage b or c with the jelly formation. Further dissolution development stage is unconstrained swelling that leads to the polymer dissolution, i.e. the formation of the polymer solution.
4. The transition с → d occurs as a result of diffusion forces and is characterized by the significant system entropy increase. In this case, the polymer molecules are evenly distributed throughout the low molecular weight solvent volume to form a true solution (regular solution).
Factors Affected Polymer Dissolution
-Temperature and pressure (Le Chatelier principle);
-Polymer and solvent polarity. Swelling and dissolution can occur if the polarity of the polymer and the solvent are close to each other. If the polymer and the low molecular weight liquid polarity are significantly different then dissolution will not occur. However, there are some exceptions when a polymer can be dissolved in a solvent with a greater difference in polarity than that one of a polymer;
-Polymer structure. Flexible structures dissolve faster due to faster diffusion;
-The polymer molecular weight. The polymer molecular weight increasing leads to a reduction of the swelling and dissolution ability in the same solvent;
-pH. The lowest polyelectrolytes swelling and dissolution corresponds to the isoelectric point. Isoelectric point is the pH at which a molecule carries no net electrical charge or is electrically neutral in the statistical mean. Swelling and dissolution raises above and below this point.
-The presence of electrolytes. The addition of electrolyte ions to the solvent reduces the polar molecules solubility and impairs the swelling process.
-The dispersion degree. An increasing of the polymer dispersion degree promotes swelling due to the size of the individual particles decreases and their specific surface area increases.
-Age of the polymer. Freshly prepared or specially plasticized polymers swell faster than polymers after long-term storage.
Based on the mentioned above information, it can be understood that the polymer dissolution is a complex process. As opposed to a low-molecular-weight substance dissolution, a polymer dissolution depends not only on a molecule polarity. A polymer solubility is usually calculated using several formulas, followed by an experimental confirmation. Here are some examples of polymers and its solvents.
Polyethylene is insoluble at room temperature and does not swell in any of known solvents. It is soluble in cyclohexane and carbon tetrachloride at elevated temperature (80 °C). Also it can be dissolved under high pressure in superheated water to 180 °C.
Polystyrene is soluble in carbon disulfide, pyridine, acetone, toluene, dichloroethane, chloroform, carbon tetrachloride and esters. Insoluble in water.
Cellulose acetate is one of the most widely used polymer. It is soluble in acetic acid, methylene chloride, chloroform, dichloroethane, aniline and pyridine.
Polyacrylamide is soluble in water, morpholine, formamide, glycerine, ethylene glycol and glacial acetic acid. It swells in propionic acid, dimethyl sulfoxide and propylene glycol. Insoluble in alcohols and ketones.
Polyvinyl acetate is soluble in acetic acid and many organic solvents such as acetone, methanol, ethyl acetate, benzene, methylene chloride, etc.
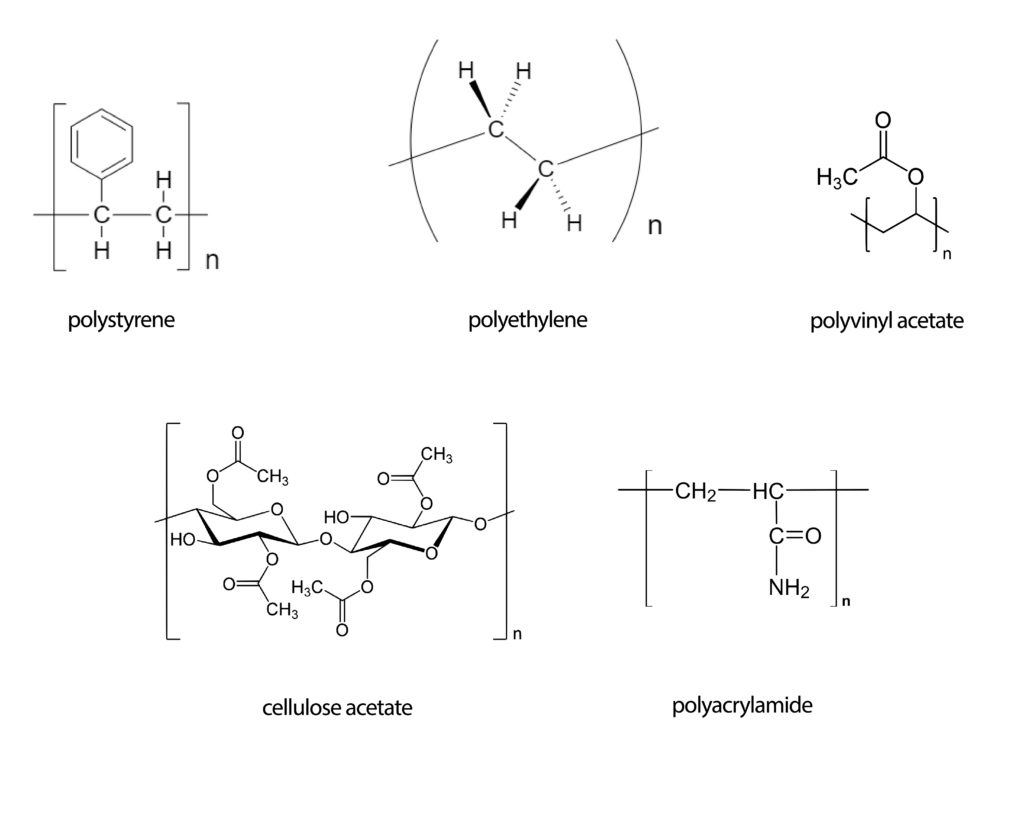
Fig. 3 Polymer structures from dissolution examples.
As you can see from this article, it is important to understand the dissolution details to dissolve the polymer substance.

