Introduction
Pyrrolidine (C₄H₉N) – is an important nitrogen-containing compound widely used in organic chemistry and industry due to its unique properties and reactivity. Pyrrolidine is found in tobacco and carrots, the pyrrolidine structure is a constituent of some alkaloids (e.g. nicotine), the amino acids proline and hydroxyproline. This five-membered heterocycle has been the object of intense research in recent decades, leading to the discovery of its chemical and physical properties, as well as the expansion of its applications. In this article, we present an overview of the main properties of pyrrolidine, its reactions and applications.
Pyrrolidine has interesting physical and chemical properties that determine its importance in organic synthesis and industry. It is a molecule with nitrogen in a pentagonal atomic structure, which gives it unique characteristics. These features make pyrrolidine an important component in a multitude of organic compounds and functional groups, playing a main role in the formation of complex molecular structures. For example, pyrrolidine used for synthesis alpha-pyrrolidinopentiophenone (a-PVP), also known as “bath salts” or “flakka”, synthetic psychostimulant of the cathinone class.
In addition to its activity in organic synthesis, pyrrolidine has found wide application in various industries. Its role in the production of pharmaceuticals, plastics, agrochemistry and other chemical products is becoming more important.
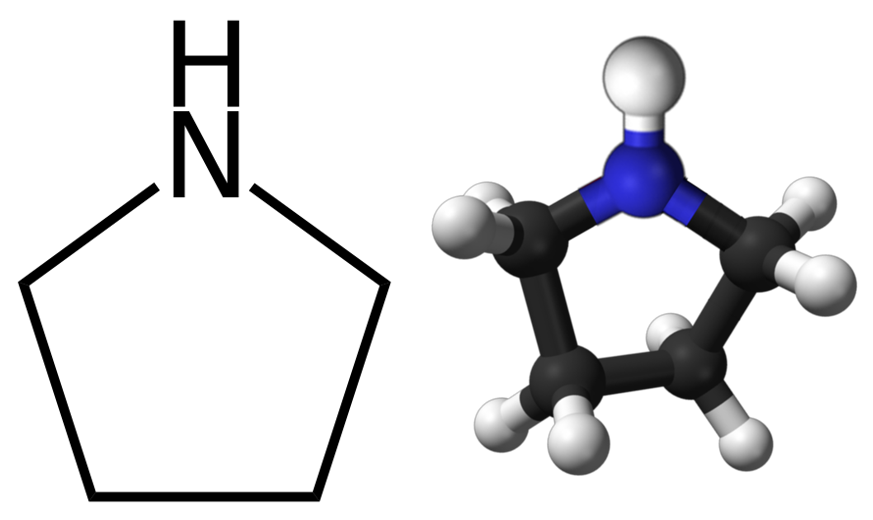
Physical and Chemical Properties of Pyrrolidine
Pyrrolidine (tetrahydropyrrole) is a nitrogen-containing heterocyclic compound, a secondary amine with a chemical formula C₄H₉N and a molecular weight 71.11 g/mol. According to its physical characteristics, pyrrolidine is a colorless liquid with a characteristic odor of ammonia, fumes in air. Its density at room temperature is 0.866 g/mL, its melting point is -63°C and its boiling point is about 87°C. Pyrrolidine has basic properties. Liquid pyrrolidine can form flammable vapor concentrations at ordinary temperatures. Pyrrolidine is soluble in water, diethyl ether, acetone, ethanol and slightly soluble in benzene, chloroform.
Synthesis of Pyrrolidine
There are several main methods for the synthesis of this five-membered nitrogen-containing heterocycle, which have their own characteristics and advantages.
1) One of the classical approaches to the synthesis of pyrrolidine, is the reduction of pyrrole. Pyrrolidine is formed by the reduction of pyrrole with hydrogen at 200°C on a nickel catalyst.
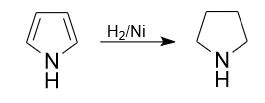
Reduction of 3-pyrroline with hydrogen at 200 °C on a nickel catalyst, also produces pyrrolidine. This approach makes it possible to obtain pyrrolidine of high purity.
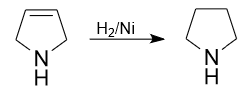
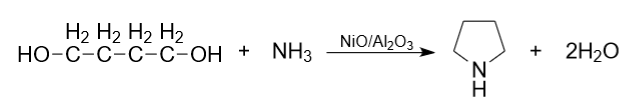
2) Also in industry, pyrrolidine is produced by the reaction of 1,4-butanediol and ammonia at a temperature of 165-200°C and a pressure of 17-21 MPa on a nickel oxide catalyst supported on aluminum oxide.
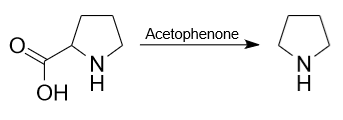
3) In laboratories, pyrrolidine is often prepared by decarboxylation of the amino acid L-Proline using acetophenone as a catalyst.
Details of the synthesis method are shown in the video in this article.
4) Another less common method for producing pyrrolidine is the reduction of succinimide. When carrying out the reaction, safety precautions should be observed. The reaction is carried out in the presence of sodium in ethanol.
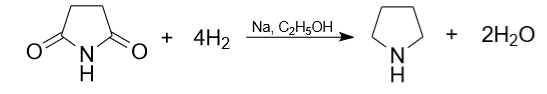
The synthesis of pyrrolidine presents many variants, each of them may be selected depending on the specific purposes and available reagents.

Chemical reactions of Pyrrolidine
Pyrrolidine has diverse reactivity, making it an important object of study in organic chemistry. In this section, we will look at some of the most important chemical reactions in which pyrrolidine is involved.
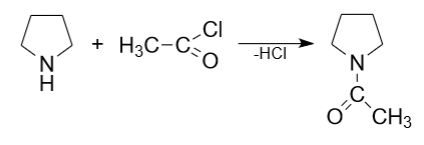
1) Pyrrolidine, being a good nucleophile, easily undergoes electrophilic substitution reactions with various electrophiles, such as alkyl halides and forms N-substituted pyrrolidines. N-acetyl pyrrolidine can be produced from pyrrolidine and acetyl chloride, hydrogen chloride must be removed from the reaction mass.
2) The reaction of pyrrolidine with nitrous acid has been identified. When carrying out the reaction, water must be removed from the reaction mass.

3) Reaction with cyclic ketones yields enamines. 1-Pyrrolidino-1-cyclohexenecan can be obtained from cyclohexanone with pyrrolidine, water must be removed from the reaction mass. First stage of the reaction: reduction of cyclohexanone to 1-cyclohexen-1-ol. The second stage of the reaction is the conversion of 1-cyclohexen-1-ol to 1-pyrrolidino-1-cyclohexene.

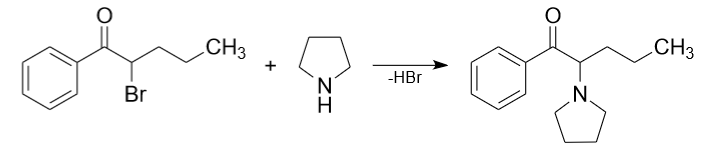
4) Sometimes in laboratories pyrrolidine is used for synthesis of alpha-pyrrolidinopentiophenone (a-PVP). Pyrrolidine reacts with 2-bromo-1-phenylpentan-1-one, hydrogen bromide must be removed from the reaction mass.
5) The industry also uses catalytic dehydrogenation of pyrrolidine at temperatures from 200°C to 300°C on palladium catalysts. Fixed-bed reactors are used as synthesis reactors. The reaction can be carried out without pressure or under pressure, preferably in a continuous mode, but also possibly in a batch mode.
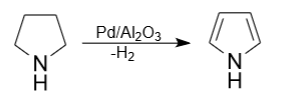
The variety of chemical reactions in which pyrrolidine participates makes it an important component of organic synthesis and expands its applications in various fields of chemistry and industry. These reactions allow the creation of a variety of organic molecules using pyrrolidine as a building block.

Applications of Pyrrolidine
Due to its unique chemical properties and reactivity, pyrrolidine has a wide range of applications in various fields of chemistry, pharmaceuticals and industry. In this section we will look at some of the main applications of pyrrolidine.
- Pharmaceuticals: Pyrrolidine is an important component of many drugs and biologically active molecules. Its heterocyclic structure allows the development of drugs with high selectivity. Can be included in the structure of antispasmodic, antiviral, antibiotics and nootropic drugs. For example, the nootropic drug piracetam is obtained from pyrrolidine.
- Chemical industry: Pyrrolidine is used in the chemical industry as an intermediate for the synthesis of various chemical products. It can be included in catalysts, polymer stabilizers, solvents and other chemical additives, improving the properties of the final products.
- Agrochemistry: Pyrrolidine is used in agriculture and agrochemistry as a component of various insecticides and herbicides. Its insecticidal properties help control crop pests and reduce the need for fertilizers. For example, the biofungicide pyrrolnitrin is obtained from pyrrolidine.
- Synthesis of polymers: Pyrrolidine can be included in the synthesis of polymers such as nylon to improve their mechanical and chemical properties. It is also used to produce polyvinylpyrrolidone.
- Extraction agents: Pyrrolidine is used as an extractant to extract chemically valuable compounds from raw materials. Its ability to form complexes with various molecules makes it a useful tool in the processing of raw materials.
This demonstrates the important role of pyrrolidine in modern chemistry and industry.

Health Effects of Pyrrolidine
Although pyrrolidine is widely used in chemical and pharmaceutical applications, its potential health effects must be considered. In this chapter we will look at some aspects of the health effects of pyrrolidine.
- Toxicity: Pyrrolidine may have toxic properties at high concentrations. Inhalation of pyrrolidine vapor may cause respiratory and eye irritation. Some studies have shown that high doses of pyrrolidine may be carcinogenic and teratogenic (harmful to the developing fetus). This highlights the importance of maintaining safety precautions when handling this compound.
- Dermatologic reactions: Skin contact may cause irritation and allergic reactions in some people. Therefore, it is important to use protective equipment.

Safety Precautions
Working with pyrrolidine, as with any chemical, requires certain precautions. In this chapter, we will discuss several important precautions when handling pyrrolidine.
- Personal protection: When working with pyrrolidine, always use appropriate personal protective equipment, such as chemical safety goggles, chemical resistant gloves, protective clothing and respirators. This will help minimize contact with the substance and protect against possible exposure.
- Ventilation: Work in a well-ventilated area or under exhaust ventilation to avoid inhalation of pyrrolidine vapors. Additionally use personal respiratory protection.
- Storage: Store pyrrolidine in glass containers, in special chemical containers designed for storing chemicals. The substance should be stored in a dark, dry room, away from sources of flame and heating devices.
Conclusion
In this article, we have discussed various aspects of pyrrolidine, from its physical and chemical properties to its wide range of applications. Pyrrolidine, as a five-membered nitrogen-containing heterocyclic compound, has unique chemical characteristics that make it an important component in organic chemistry.
The variety of chemical reactions in which pyrrolidine participates opens possibilities for its use in organic synthesis. It can be a «building block» for the creation of complex molecules and heterocyclic structures, which is important for pharmaceuticals and medicinal chemistry. Pyrrolidine has also found its place in the chemical, agrochemical and polymer industries, demonstrating its importance in various aspects of modern technology. In general, pyrrolidine remains a subject of research and development.
Bibliography
- Purification and Properties of Pyrrole, Pyrrolidine, Pyridine and 2-Methylpyridine R. Vernon Helm, W. J. Lanum, G. L. Cook, and J. S. Ball J. Phys. Chem. 1958, 62, 7, 858–862 Publication Date: July 1, 1958 https://doi.org/10.1021/j150565a023
- Pyrrolidine, piperidine, and pyridine alkaloids A. R. Pinder Nat. Prod. Rep., 1990,7, 447-455 https://doi.org/10.1039/NP9900700447
- Heterocyclic compounds such as pyrrole, pyridines, pyrrolidine, piperidine, indole, imidazol and pyrazines Yasuhiko Higashio, Takayuki Shoji Applied Catalysis A: General Volume 260, Issue 2, 8 April 2004, Pages 251-259 https://doi.org/10.1016/S0926-860X(03)00197-2
- General Pyrrolidine Synthesis via Iridium-Catalyzed Reductive Azomethine Ylide Generation from Tertiary Amides and Lactams Ken Yamazaki, Pablo Gabriel, Graziano Di Carmine, Julia Pedroni, Mirxan Farizyan, Trevor A. Hamlin and Darren J. Dixon Cite this: ACS Catal. 2021, 11, 12, 7489–7497 Publication Date:June 9, 2021 https://doi.org/10.1021/acscatal.1c01589
- Chapter 3 Pyrrolidine Alkaloids Georges Massiot, Clement Delaude The Alkaloids: Chemistry and Pharmacology Volume 27, 1986, Pages 269-322 https://doi.org/10.1016/S0099-9598(08)60309-0
- https://bbgate.com/threads/alpha-pvp-synthesis-1-10kg-scale-complete-video-tutorial.12/

