The iodine experiment is a chemical reaction-based identification procedure for starch. In this experiment, iodine and starch create a definite blue-black colored combination.
What does iodine test for?
This test can be used to detect the occurrence of starch in a sample. It can furthermore differentiate between monosaccharides and polysaccharides (glycogen, dextrin, and amylase).
Goals of Iodine Experiment
Verification of the existence of starch in the specimen supplied, employing the Iodine trial.
Biology description:
A starch-iodine test is a method for ascertaining the presence of starch. When a few droplets of potassium iodide solution are added to the specimen, the mixture changes to a dark blue or black hue. This is due to the production of polyiodide chains following the combination of starch and iodine. The amylose in starch builds up helices that bind with iodine molecules, forming the shade. If starch is broken down or split into smaller carbohydrate molecules, the blue-black color will not be created. As such, this test can also suggest the ending of hydrolysis when no color alteration comes about. Synonym: Iodine test.
Mechanism of Iodine Test
Before comprehending the concept of the iodine test, let us quickly grasp starch and its chemistry. Starch is a polysaccharide. Glucose created by plants is saved as starch. Starch is prevalent in potatoes and grains (oats, barley, rice, wheat). Natural starch consists of two monomeric components: Amylose (10-20%) and Amylopectin (80-90%). Chemically both monomers are composed of D-glucose subunits. Nonetheless, the arrangement of the D-glucose unit in Amylose and Amylopectin is dissimilar.
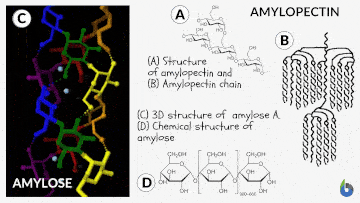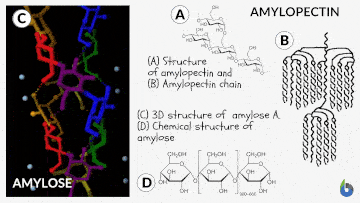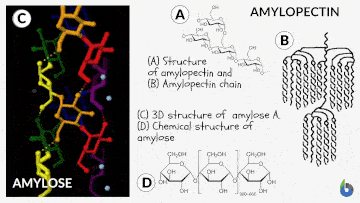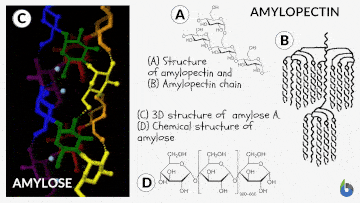
The Iodine test chemistry
The underlying concept of the iodine test is that Amylose combines with starch to generate a blue-black complex in the presence of iodine. The helical architecture of Amylose allows for a charge transfer (CT) complex with iodine, with iodine being within the helical structure of the Amylose. Therefore, for this test, an aqueous solution of molecular iodine (I2) and potassium iodide (KI) is used, commonly known as Lugol’s iodine or the KI3 solution.
I2 + KI = K[I]3 solution
Let us now contemplate the role of iodine in Lugol’s iodine test. Molecular iodine, or iodine molecule, is insoluble in water and so potassium iodide is used to make a laboratory reagent. Potassium iodide breaks down to form iodide ions, which join together to form triiodide ions (I3–). These triiodide ions then form polyiodide ions (In–) when in solution.
In essence, it is the I3– chemistry that creates the In– ions. The polyiodide ions are negatively charged, and can be triiodide (I3–), pentaiodide (I5–), or heptaiodide (I7–). These polyiodide ions serve as charge donors and form a complex with amylose. The Lugol’s iodine solution seen in a laboratory setting is brownish in color. However, when the charge transfer complex of polyiodide ions and amylose absorbs light energy, electrons become excited and reach a higher energy level. This energy is perceived as a blue-black color by the human eye.
In order to perform the starch detection test, the following supplies are necessary:
-Sample to be tested
-Iodine solution or Lugol’s reagent*
-Test tubes
-Tube rack
-Water bath
-Pipette
Note: What is an iodine solution? It is a water-based mixture of molecular iodine (5%) blended with potassium iodide (10%). The iodine solution has a brown hue and must be kept in a dark location.
Procedure of Iodine Test
The steps of the Iodine Test:
1. Obtain two test tubes and label them as the test sample and the control sample.
2. Place a small quantity (solid sample: 500 mg – 1000mg; liquid: 1ml) of the sample in a clean and dry test tube labeled as the test sample.
3. Put 1ml of purified water in the second clean and dry test tube identified as the control sample.
4. Pure 2-3 drops of Lugol’s iodine solution into each of the test tubes and stir them.
5. Observe the shade developed in both of the test tubes.
6. Then, warm the test tubes in a water bath until the color fades.
7. Let the test tubes cool off fully and inspect the color in both of the test tubes.
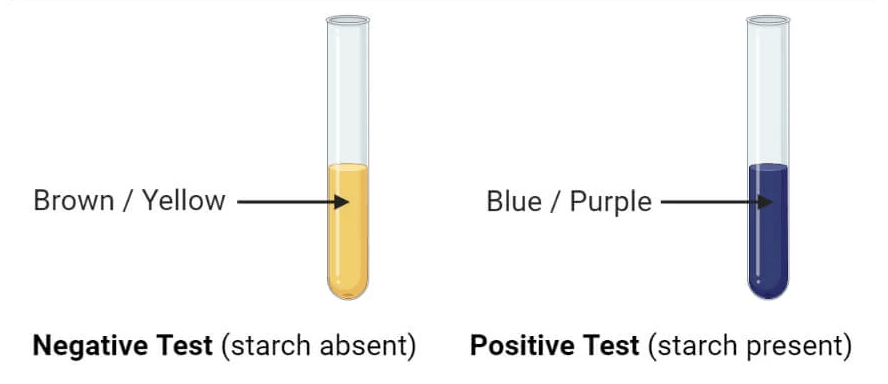
Outcome and Analysis of Iodine Test
-The emergence of a blue-black or violet hue implies a positive test, suggesting the existence of starch.
-If the color remains unchanged, then the outcome is negative, signifying the lack of starch.
What is the goal of the iodine test? Why is it a vital laboratory process?
An iodine test can be used to detect starch in a given sample.
The test can help to differentiate starch from monosaccharides, disaccharides, and other polysaccharides.
The iodine test is used to distinguish between starch, glycogen, and carbohydrates.
Blood iodine testing is used to identify hyper– or hypothyroidism in a patient.
The concept of iodine test for starch is also employed in starch hydrolysis test.
The iodine test serves as the basis of iodometric titrations wherein a starch indicator is employed.

Figure 4: Positive Test Example
Iodine Test Limitations
One of the key drawbacks of the iodine test is that the test is qualitative. This implies that the presence or absence of the starch in the sample can be identified. However, it is impossible to calculate the quantity of starch present in the sample utilizing the iodine test.
Another shortcoming is that starch hydrolysis occurs under acidic conditions. Therefore, the iodine test would be ineffective for acidic samples.
The iodine test cannot be performed on a very dark-colored sample as the color changes will be imperceptible in such samples.
Important Notes
-Lugol’s iodine solution is delicate to light and ought to be securely held in a dim chestnut shaded jug in a dull spot.
-The iodine experiment is an unmistakable indicative device for starch just. For example, cellulose won’t change tone with Lugol’s iodine solution.
-The assessment is impressionable to variances in temperature.
