In chemistry, recrystallization is a procedure utilized to cleanse chemicals. By dissolving both impurities and a compound in an appropriate solvent, either the sought-after compound or contaminants can be removed from the solution, leaving the other behind. It is named for the crystals regularly shaped when the compound precipitates out. As an alternative, recrystallization can allude to the natural development of bigger ice crystals at the cost of littler ones.
The technique of purification is based on the rule that the dissolvability of most solids rises with expanded temperature. This implies that as temperature increases, the measure of solute that can be broken up in a solvent increases.
An impure compound is dissolved (the impurities must also be soluble in the solvent), to prepare a highly concentrated solution at a high temperature. The solution is cooled. Decreasing the temperature causes the solubility of the impurities in the solution and the substance being purified to decrease. The impure substance then crystallizes before the impurities- assuming that there was more impure substance than there were impurities. The impure substance will crystallize in a purer form because the impurities won’t crystallize yet, therefore leaving the impurities behind in the solution. A filtration process must be used to separate the more pure crystals at this point. The procedure can be repeated. Solubility curves can be used to predict the outcome of a recrystallization procedure.
Recrystallization works best when
- the amount of impurities is little
- the solubility curve of the desired solute rises quickly with temperature
Single solvent crystallization
This section gives a visual representation of the refinement of approximately 1g of aged NBS – bromosuccinimide (NBS), which was discovered as an orange powder inside its container. The crystallization process utilizes water as the solvent, which poses no fire hazard and therefore a hotplate is employed.

If a crystallization is to be done using combustible organic solvents, it is advisable to use a steam bath and in certain circumstances essential (when employing diethyl ether, acetone, or low-boiling petroleum ether). This technique should be followed as a rule of thumb for the operation, and some major contrasts between using water and organic solvents are discussed in a subsequent segment.

Get ready
• Move the unclean solid to be crystallized into a suitably sized Erlenmeyer flask (Fig.2 a). If the solid is granulated, first crush it with a glass stirring rod.
• It is not suggested to carry out crystallizations in a beaker. The narrow neck of an Erlenmeyer flask allows for simpler swirling and minimized evaporation during the process as solvent vapors rather condense on the walls of the flask (they “reflux” on the sides of the flask). The limited opening of an Erlenmeyer likewise permits a flask to be more effortlessly covered during the cooling stage, or even potentially sealed for long crystallizations. A round-bottomed flask is not perfect for crystallization as the shape of the flask makes it hard to recover solid at the end of the process.
• It is essential that the flask be not excessively full or too empty during the crystallization. If the flask will be more than half-full with hot solvent, it will be hard to stop the flask from boiling over. If the flask will contain solvent to a height less than 1cm, the solution will cool too rapidly. It is normal to use between 10-50 times as much solvent as sample, and a rough guide is to use a flask where the sample just covers the bottom in a thin layer.
• Put some solvent in a beaker or Erlenmeyer flask alongside a few boiling stones on the heat source, and bring to a gentle boil. Utilize a beaker if the solvent will be poured and an Erlenmeyer flask if the solvent will be pipetted. If a hot filtration step is expected for later in the procedure, also prepare a ring clamp containing a funnel with fluted filter paper (Fig.2 b).
Add the Minimum Quantity of Hot Solvent
• When the solvent is boiling, hold the beaker with a hot hand protector (Fig.2 d), cotton gloves, or a paper towel holder made by folding a sheet of paper towel into a long rectangle (Fig.2 c). To the side of the heat source, pour a small part of boiling solvent into the flask containing the impure solid, to coat the bottom of the flask. If the crystallization is being done on a small scale (utilizing a 50ml Erlenmeyer flask or smaller), it may be simpler to use a pipette to transfer portions of the solvent to the flask.
• It’s typical to not put the dry solid on top of the heat source before adding solvent or the solid may decompose. When the solid is distributed in a small quantity of solvent, it can then be placed on the heat source.

• Place the beaker containing the impure solid and fluid on the heat source. Utilize some process to avoid bumping (boiling stones if intending to “hot filter”, a boiling rod), and bring the mixture to a temperate boil (Fig.3 a).
• Include fluid in parts (Fig.3 b), twirling to support dissolution, until the solid simply melts (Fig.3 d). For 100mg-1g of compound, add 0.5-2ml parts each time. Note that it may take time for a solid to totally dissolve as there is a kinetic viewpoint to dissolution. Each addition should be allowed to come completely to a boil before adding more fluid, and some time should be allowed between additions. Not permitting time for dissolution and then adding too much fluid is a major source of blunder in crystallization.
It is common for drops of liquid to be witnessed during the heating procedure (Fig.4). This is when the material “oils out”, or liquefies before it dissolves. If this happens, the liquid drops are now the compound you are crystallizing, so keep on adding fluid in parts until the liquid drops totally dissolve as well.
• Monitor the solution cautiously to assess whether the size of the solid pieces (or liquid drops) vary with extra fluid: if they don’t they may be an insoluble impurity. Inclusion of excessive fluid in an attempt to dissolve insoluble impurities will adversely affect the recovery. If insoluble solid impurities are present, the solution should be filtered (insert a hot filtration step at this point). Colored impurities can also be eliminated at this point with charcoal.

a) Oily droplets of acetanilide in a solution decolorized with charcoal, b and c) Oily droplets containing methyl red and acetanilide in water.
Allow the Solution to Slowly Cool
• After the solid has been dissolved, take the flask away from the heat source using a heat resistant glove, oven mitt, or paper towel holder, and set it apart to cool. Take away the stirring rod or stirring bar if previously used for bump protection (boiling stones can be gotten out of the solid at a later time if used).
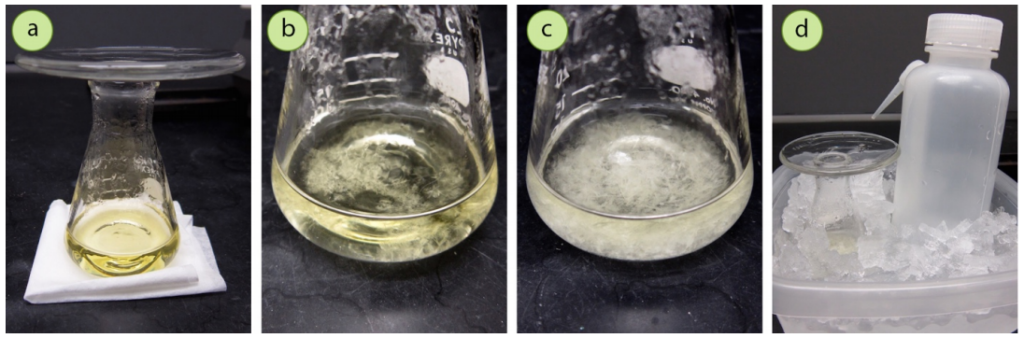
• To encourage a gradual chilling, put the flask on top of a material that does not transmit heat well, for instance a folded paper towel. Cover the neck of the Erlenmeyer flask with a watch glass to keep heat and solvent (Fig.5 a). Let the mixture come to room temperature gradually.
• As the solution cools, finally solid crystals should form (Fig.5 b). If the solution is only lukewarm to the touch or hazy and no crystals have appeared, utilize a glass stirring rod to scratch the glass and set off crystallization.
• After crystallization has started, the crystals should slowly develop as the temperature decreases. An ideal crystallization usually takes between 5-20 minutes to totally crystallize, depending on the size. Quick crystallization in less than 5 minutes is too fast.
• When the solution is at room temperature, place the flask into an ice bath (ice-water slurry) for 10-20 minutes to reduce the compound’s solubility further and maximize crystal formation (Fig.5 d). Also put a portion of solvent in the ice bath, to be used later for rinsing during suction filtration.
• Utilize suction filtration to rescue the solid from the mixture.
Mixed solvent crystallization
This section depicts the crystallization of a nearly 1g measure of trans-cinnamic acid. Trans-cinnamic acid is miscible in methanol and insoluble in water, and this crystallization utilizes a combination of methanol and water to give a 74% recuperation.

Presupposing that the experimenter carrying out this practice has previously done or studied a single-solvent crystallization, the following steps should be taken:
• Establish two compatible solvents that can be implemented for the crystallization (Fig.7 a): the sought-after substance should be soluble in one liquid (called the “soluble solvent”) and insoluble in the other liquid (designated the “insoluble solvent”).
• Move the impure solid to be crystallized into an Erlenmeyer flask of the appropriate size (Fig.7 b).
• Place some of the “soluble solvent” into the flask (Fig.7 c), add a stirring rod (or stirring stones if preferred), then warm above a steam bath (Fig.7 d). A hotplate can be used cautiously if using the mixed solvents methanol/water or ethanol/water.

• Introduce increased amounts of the “soluble solvent” until the solid is fully dissolved (Fig.8 a). Allow each addition to reach boiling point before adding more.
• Introduce the “insoluble solvent” in increments while heating until the mixture is slightly hazy (Fig.8 c).
• Gradually add the “soluble solvent” while warming until the solution is clear once more (Fig.8 d).

• Take the flask away from the heat source, eliminate the boiling rod and place the flask on a paper towel folded multiple times. Put a watch glass over the mouth of the Erlenmeyer flask and let the solution gently cool down to the room temperature (Figure 9a).
• As the solution cools, it should produce solid crystals (Figure 9b). Use a glass stirring rod to scrape the flask and encourage crystallization if needed. Put the crystals in an ice-water bath for 10-20 minutes and get the solid by suction filtration.

Hot filtration
A hot filtration is typically utilized in certain types of crystallization, when a solid contains contaminants that cannot be dissolved in the crystallization solvent. It is also required in crystallization when charcoal is employed to extract intensely colored impurities from a solid, as charcoal is so fine that it can’t be removed by decanting. A hot filtration is undertaken by first pouring a few milliliters of solvent through a funnel featuring a “fluted filter paper”. A fluted filter paper has multiple indentations and high surface area, which facilitates a rapid filtration. The funnel is allowed to become warm, while the mixture to be filtered is brought to a boil. The boiling mixture is then dispensed through the filter paper in portions (Fig.10 b and d).

It is advisable to attach a ring clamp to the filtration funnel, although it is possible to just rest the funnel on the flask. If a ring clamp is not available, a curved paper clip can be placed between the flask and funnel to allow air to release from the lower flask as the liquid drains (Fig.10 c and d). Without the clamp, the apparatus is more likely to tip, so employing a clamp is much safer.
In the event of a hot filtration, where solutions crystallize when cooled, it is critical that the funnel is kept warm by contact with the hot solvent vapors, or else crystals may form on the filter paper or in the funnel stem (Fig.11).

Crystallization of solids on the filter paper may block the set-up and result in a reduction of yield (as the filter paper will be discarded afterwards). Crystallization within the stem impedes filtration, potentially acting as a plug in the bottom of the funnel. One benefit of hot filtration is that the hot solvent in the filter flask aids in dissolving any crystals that prematurely form in the stem of the funnel. If hot filtration is employed, it is recommended to utilize a short-stemmed funnel (Fig.12 a) or stemless funnel (Fig.12 c) if available, in preference to a long-stemmed funnel (Fig.12 b), as material is less likely to precipitate in a short or nonexistent stem.


It is crucial that a solution is filtered expeditiously before it has time to cool in the funnel, making “fluted filter paper” (Fig. 13 b and c) the preferable choice to the quadrant-folded filter paper employed with gravity filtration (Fig. 13 a). The numerous folds on the fluted filter paper result in greater surface area and swifter filtration. The pleats also make room between the filter paper and glass funnel, letting displaced air escape the flask more simply as liquid drains.

a) Quadrant-folded filter paper (not recommended for hot filtration), b) A fluted filter paper after folding, c) Unfolded fluted filter paper during hot filtration.
Step-by-Step Procedures
Hot filtration is regularly employed in combination with crystallization, and this practice should be implemented after the dissolution step, but prior to allowing the solution to gradually cool.

Gather the Filtration Equipment
• Acquire a stemless or short-stemmed funnel (Fig.14 a), and attach it to a ring clamp secured to a ring stand or latticework (or alternatively, use a bent paper clip for the purpose shown in Fig.14 b).
• Pleat a filter paper of appropriate size for your funnel into a zigzag shape (instructions are in Fig. 15 and the completed pleat is in Fig.14 a). When placed in the funnel, the paper should not be shorter than the top of the funnel, or the liquid may slip over the filter paper when poured.
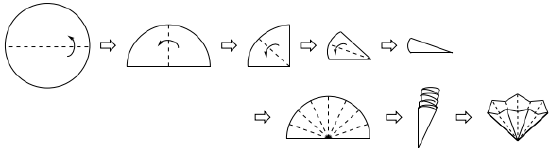
Fig.15 Forming a pleated filter paper. The dotted lines indicate spots to bend and fold the filter paper. The arrows point out the direction of folding.
• With the correct size of Erlenmeyer flask situated beneath the funnel and on the heat source, add a few milliliters of heated solvent into the funnel (Fig.14 d).
a) If relying on a ring clamp, adjust the clamp to create a tiny opening between the flask’s mouth and bottom of the funnel: this allows air to pass through when liquid enters the flask. If the opening is too wide, hot gases will be released without heating the funnel.
b) If not using a ring clamp, place a curved paper clip between the flask and funnel (Fig.14 b).
• Enable the solvent to vaporize and heat the whole set-up. If utilizing charcoal, execute that action now.

Filter the Solution in Portions
• When the filter flask is quite hot, and the solution to be filtered is boiling, dispense the boiling mixture into the filter funnel in sections. Keep the flask adjacent to the filter paper in the funnel as you dispense (Fig. 16 a).
• Safety note: the flask may be quite hot, and hot vapors may burn your hand as you pour (pour sideways so your hand is not above the funnel). If the flask is too hot to be handled with your hands, use a “paper towel holder” to hold the flask (Fig. 16 a):
a) Layer a sliver of paper towel several times such that the resulting strip is roughly one inch wide. If desired, secure the strip together using a few strips of tape.
b) When handling a flask, the paper towel holder should be below the lip of the flask. In this way, liquid will not be absorbed by the paper towel when pouring (towel stays dry in Fig. 16 a), but becomes damp with the too wide towel in Figure Fig. 16 c).

• When not decanting the mixture to be filtered, relocate the flask to the heat source (Fig. 17 a).
• When the mixture is totally filtered, station the bare flask on the workbench (precautionary note: do not warm an empty flask, or it may break). Check the funnel: if crystals are visible on the filter paper (as in Fig. 17 b), rinse with a few ml of boiling reagent to dissolve them. A rinse is not required in Fig. 17 c.
• Observe the filtrate (the fluid that has gone through the filter paper). If charcoal was utilized and the filtrate is grey, or you can detect minor black grains, then charcoal passed through the filter paper either through a puncture or by utilizing the improper filter mesh size.
