Introduction
Organic compounds are classified into acids and bases. Mainly three theories of acids and bases are used in modern organic chemistry: Arrhenius theory, Brønsted–Lowry acid–base theory and Lewis’s theory. Despite there are three different approaches, they are not contradictory and are applied in combination with each other.
Application
The acid-base properties of organic compounds are of a greatest importance as they can provide critical information regarding the reactivity of a given compound. After determining whether the compound is an acid as opposed to a base it is possible to determine its strength by the value of pKa. The term pKa is referred to acid dissociation constant (acidity constant, acid-ionization constant) and it is a quantitative characteristic of an acid strength in a solution. The lower pKa, the stronger an acid. Also, the more readily a compound can donate a proton, the more acidic it is. Some functional groups are more acidic than others (such as carboxylic acids and phenols). The more stable a molecule is without a proton, the more easily it will give it up and become deprotanated. On the other hand, the more readily a compound donates electrons the more basic it is. All of these acid-base properties play an important role in the reactivity of organic compounds.
The main goal of the acid-base theory is to predict an acid-base interaction products and a probability of its occurrence. For these purposes quantitative characteristics of acid and base strength are applied. Acid-base interactions are widespread and widely used in laboratory, scientific and industrial practice.
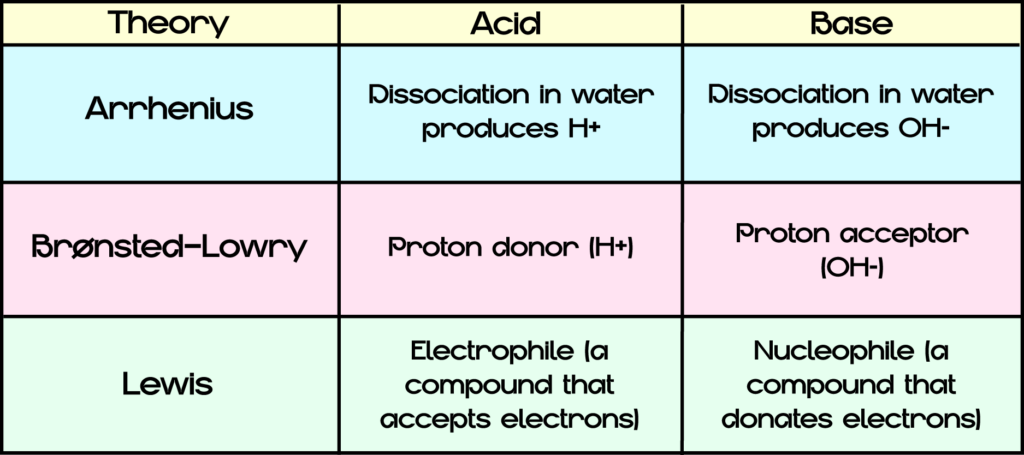
1. Arrhenius theory
According to the Arrhenius theory:
- Acids are electrolytes that dissociate with the formation of hydrogen ions, for example:
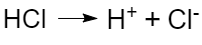
- bases are electrolytes that dissociate in solutions with the release of hydroxide ions (OH–), for example: NaOH→Na++OH–
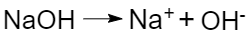
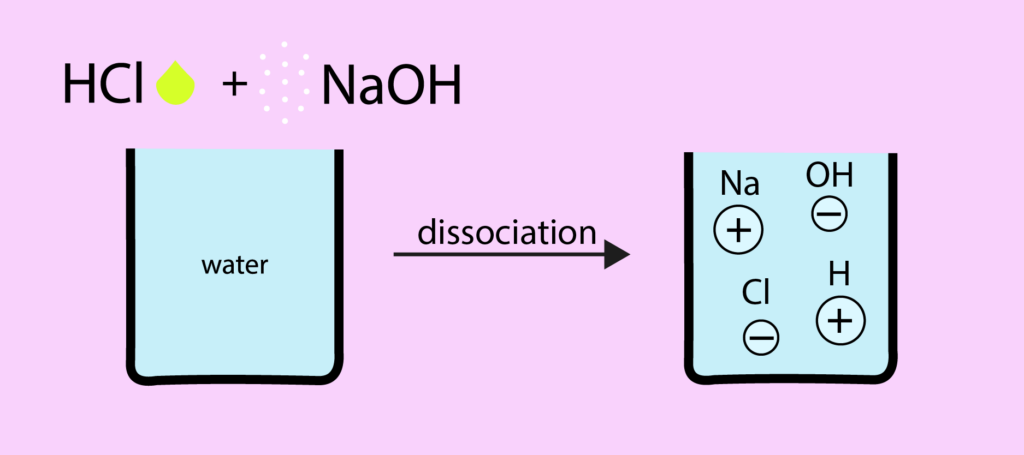
Since mixing of an acid aqueous solution and base aqueous solution produces a neutral solution, Arrhenius called such acid-base reaction a neutralization reaction.
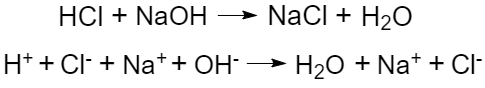
The hydrogen ion interacts with the hydroxide ion to form a molecule of water (the basic process underlying Arrhenius acid-base re actions):
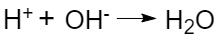
The Arrhenius’ theory has its disadvantages. For example, the interaction of gaseous ammonia with hydrogen chloride produces a solid ammonium chloride:
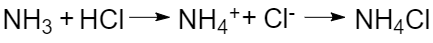
As a result of the reaction, hydrochloric acid transfers the hydrogen ion to ammonia, but this reaction takes place in the gas phase and not in aqueous solution (no hydroxide ion is involved), thus it cannot be classified as an acid-base reaction according to Arrhenius.
This problem was solved by the Brønsted-Lowry proton theory proposed in 1923.
2. Brønsted–Lowry acid–base theory
According to the Brønsted-Lowry theory, an acid is a donor, supplying its hydrogen protons to form a chemical bond, and the base acts as an acceptor, accepting these protons. The base provides one pair of its electrons to form the donor-acceptor bond. Thus, a covalent bond between an acid and base is formed by two electrons that originally belong to one atom, acting as an electron pair donor, and the free orbital of another atom, acting as an acceptor of the same pair. Usually, a covalent bond is formed by two atoms. Each of these atoms gives one electron to form covalent bond. However, both electrons are provided by the same atom according to the donor-acceptor covalent bond formation method.
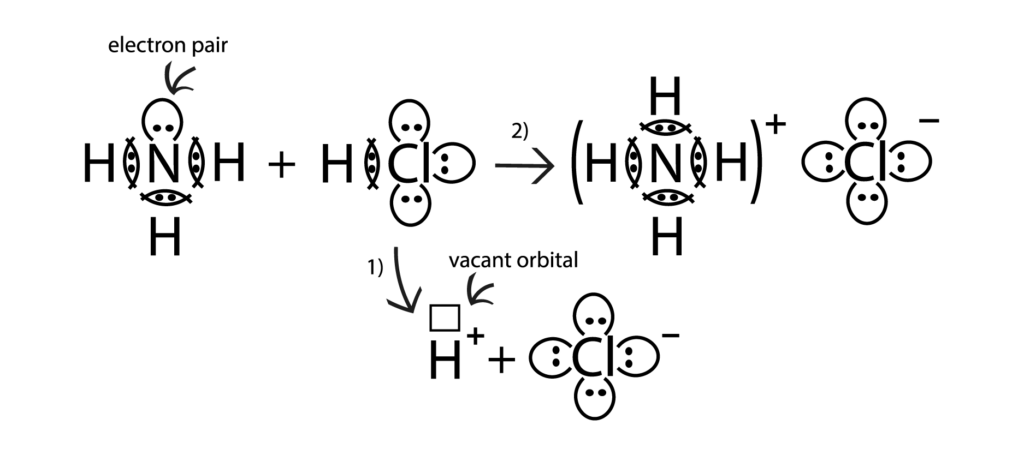
In the reaction between NH3 and HCl, hydrochloric acid is a proton donor, while ammonia acts as acceptors, since ammonia has one pair of unbounded electrons, which is used to create a donor-acceptor bond.
3. Lewis’ theory
Despite its merits, Brønsted’s theory, as well as Arrhenius’ theory, is not applicable to substances that perform acid properties and do not contain hydrogen. Therefore, the Lewis’ electron theory of acids and bases is more overall. According to this theory, an acid is a substance that accepts electron pairs (the electron acceptor); a base is a substance that supplies electrons for the formation of chemical bonds (the electron donor).
Performed theories of acids and bases do not contradict, but complement each other and have a deep inner connection. Thus, according to Brønsted, acids can be classified as a particular case of Lewis’ acids: a proton that is characterized by a high electron pair affinity can be considered an acid according to Lewis’ theory.
In accordance with Lewis, an acid is any particle that has an actual vacant orbital or a great potential for its formation. The vacant orbital of such particle is capable to be an acceptor of an electron pair that is provided by a base in an acid-base interaction. A base is a compound with an available electron pair, that is capable to provide it to an acid.
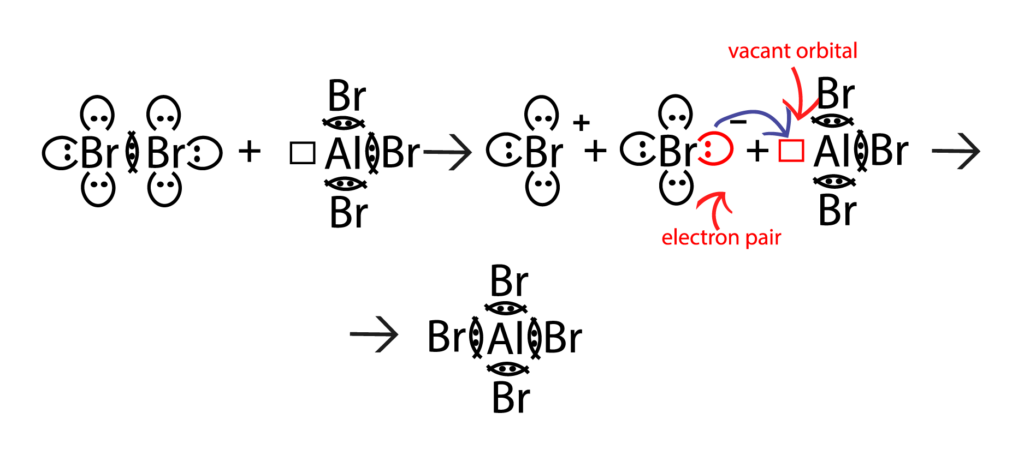
Various particles (not only proton) can act as acceptor of electron pair; therefore, this theory is more general. In organic chemistry, Lewis’ acids are typically carboxylic acids or their precursors.
