- The process of creating a drug - May 23, 2023
- How do scientists model mental disorders in animals? - May 23, 2023
Melting point is a characteristic of solid crystalline substances. This is the temperature at which the solid phase becomes the liquid phase. Melting point definition is the thermal analysis that commonly used to distinguish solid crystalline chemicals. This method is applied in research and development spheres as well as in an analytical quality control in various industry segments to identify solid crystalline substances and to check their purity. This method very helpful for checking your synthesis precursors for synthesis product confirmation according with a literature data.
What Is Melting Point?
This phenomenon takes place while the substance is heated. All applied heating energy on the substance is consumed as heat of fusion and the temperature remains constant (see diagram below) within the melting process. During the phase conversion, the two physical material phases exist side-by-side.
Crystalline materials are composed of small particles that, in a regular three-dimensional arrangement, form a crystal lattice. The particles within the lattice are held together by lattice forces. When the solid crystalline material is heated, molecules and atoms become more mobile and start to move more actively until finally the crystalline attraction forces between them are no longer strong enough to hold them together. As the result, the crystalline structure is broken and the solid substance melts.
The stronger attraction forces between particles, the more energy is needed to broke them. The more energy is needed, the higher the melting point temperature. Thus, the crystalline solid substance melting temperature is an indicator of its lattice durability.
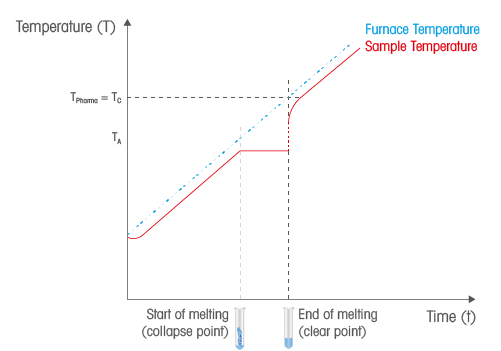
The Principle
A change in light transmission is arisen at the melting point. Compared with other physical values, the change in light transmission can easily be defined and can hence be used for melting point detection. Powdered crystalline substances are non-transparent in the crystalline state and transparent in the liquid state. This distinct difference in optical properties can be measured in order to define the melting point by recording the percentage of light intensity shining through the substance in the capillary, the transmittance, in relation to the measured furnace temperature.
There are different phases of the melting point process of a solid crystalline substance: At the collapse point, the substance is mostly solid and consists of only a small amount of molten material. At the meniscus point, most of the substance has melted but some solid material is still present. At the clear point, the substance has completely melted.

Melting Point of an Organic Compound
The Capillary Method.
Materials required:
• Liquid paraffin in a 100ml beaker (melt a candle)
• Powdered drug
• Thin walled capillary tube of 8-10cm length and 1-2mm diameter thread
• Glass or ceramic plate
• Thermometer stand with clamp stirrer, spatula, hot plate
The melting point quantification is usually carried out in thin glass capillary tubes with an internal diameter of 1-2 mm and a wall thickness of 0.1 – 0.2 mm.

Take a capillary tube and seal one end by heating it in the flame of the burner (can use camping burner). With help of the spatula or plastic card, make a heap of the powdered sample on the glass plate. Push the open end of capillary tube into the heap. You can help by spatula or plastic card. Some substance will enter into the capillary tube. Then, tap the sealed end of the capillary tube on the plate carefully and fill the capillary tube up to 2-3 mm. Attach the capillary tube to the thermometer utilizing the thread or rubber. Take the 100 ml beaker containing liquid paraffin and place it onto the hot plate. Grip the thermometer with the test tube to the iron stand and immerse them in the liquid paraffin bath. Start heating the liquid paraffin bath gradually and stir the bath carefully by the stirrer to ensure uniform heating. Note the temperature t1 when the substance starts melting. Continue heating and note the temperature t2 when the substance in the capillary tube is completely melted. The average of the two temperatures t1 and t2 gives the melting point of your sample.
Important: use dry and powdered sample for the melting point definition; the powder packing should be uniform without any big air gaps between the solid particles; the liquid paraffin bath have to be heated very slowly and paraffin is stirred gently to ensure uniform heating. The bulb of the thermometer and the capillary attaching to it should not touch the sides or the beaker bottom.
The Air Method
Materials required:
• Thermometer
• Spatul or plastic card
• Hot plate
• Powdered drug
• A foil roll
Cut a foil square and put it on the heating plate. Fold the foil in two layers and wrap around the thermometer bulb. Take couple small drug lumps and put them on the thermometer wrapping foil. Start heating it gradually (5-7 degrees per min). Write t1 temperature , when the substance fusing is started. Continue heating and write t2 temperature when the substance in the capillary tube is completely melted.
There are pictures of melting point experiment with methamphetamine. Literature melting point data for d- and l-methamphetamine hydrochloride is 170-175°C but a mixture of equal amounts of both optical isomers (racemic mixture) has a lower melting point 130-135°C. Experimental result shows 174°C, which is matched with literature data for one these (d- or l-) isomer.



Experiments with amphetamine and mephedrone were carried out as well.
Amphethamine



Literature melting point data for amphetamine sulphat is 280-281°C. Experimental result is 189°C.
Mephedrone



Literature melting point data for mephedrone hydrochloride is 217-219°C. Experimental result is 206°C.
Results and Discussion
The methamphetamine lumps melting point characterizes this powder as one of its (d- or l-) isomer. The amphetamine melting point isn’t matched with the literature data. There are three reasons: polluted by side products amphetamine, big method error, substituted substance. The mephedrone melting point isn’t matched 11°C, which can be explained by the method error.
Conclusion
Both methods have advantages and disadvantages. Capillary method takes a lot of efforts and materials to carry it out but you get more precise melting point experiment result. Air melting experiment quite easy to handle and not difficult to get materials but you get the result with big error and have to check it several times. In conclusion I want to say that you can chose more suitable method for your purpose and check the melting point of your sample.
Melting Points of Some Drugs and their Precursors:
Drugs:
Amphetamine sulphate, 280-281 °C;
Methamphetamine, 170-175 °C;
Mephedrone hydrochloride, 251.18 °C and Mephedrone hydrobromide 205.25 °C;
Cocaine hydrochloride, 197 °C;
Phencyclidine hydrochloride, 243-244 °C;
MDMA hydrochloride, 147-153 °C;
a-PVP hydrochloride, 162-173 °C;
MDA Hydrochloride, 187-188 °C;
Ephedrine hydrochloride, 217-220 °C;
Methcathinone hydrochloride, 188-191 °C;
2C-B hydrochloride, 236-238 °C;
Mescaline hydrochloride, 180-182 °C;
Methylone hydrochloride (MDMC), 236-238 °C;
DMT (free base) 42-47 °C;
DMT Fumarate 152 °C;
Psilocybin 220-228 °C;
Ergotamine 241-249 °C;
JWH-018 55-59 °C;
UR-144 68 °C;
JWH-1503 91-97 °C;
AM-2201 80 °C;
JWH-210 90 °C;
JWH-122 89 °C;
JWH-081 127 °C;
JWH-073 100 °C;
Methadone hydrochloride, 232-234 °C;
Diacetylmorphine hydrochloride (heroin) 229-233 °C;
Codeine monohydrate, 154-156 °C.
Precursors:
2,5-Dimethoxybenzaldehyde 50 °C;
2,5-Dimethoxy-4-methylbenzaldehyde 82-86 °C;
Piperonal 37 °C;
3,4,5-trimethoxybenzaldehyde 73-76 °C;
Phenyl-2-nitropropene 64-66 °C;
2-Bromo-4-methylpropiophenone 75-77 °C;
4-Cyano-2-dimethylamino-4,4-diphenylbutane 88-91 °C;

