- The process of creating a drug - May 23, 2023
- How do scientists model mental disorders in animals? - May 23, 2023
Thin layer chromatography (TLC) is a chromatographic process which is employed to distinguish the constituents of a combination through a thin stationary layer backed up by an inactive base. It may be conducted on the analytical scope as a means of tracking the advancement of a reaction, or on the preparative scale to cleanse modest amounts of a compound. TLC is an analytical instrument broadly utilized because of its uncomplicatedness, relatively low cost, elevated sensibility, and velocity of differentiation. TLC works on the same principle as all chromatography: a compound will have different attractions for the mobile and stationary stages, and this influences the speed at which it migrates. The objective of TLC is to acquire well defined, well segregated spots.
Retention Factor
Once a division has been accomplished, individual constituents manifest as spots spaced out vertically. Every spot has a retention coefficient (Rf) which is identical to the distance traversed over the total distance traversed by the liquid.
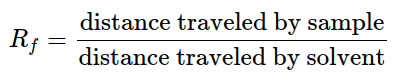
The Rf value can be deployed to discriminate between compounds due to their distinctiveness to each compound. When assessing two different compounds under the same circumstances, the compound with the greater Rf value is less polar as it does not cling to the stationary phase as long as the polar compound, which would have a lower Rf value. Rf values and repeatability can be influenced by a selection of different factors such as layer thickness, dampness on the TLC plate, vessel saturation, temperature, depth of mobile phase, nature of the TLC plate, sample size, and solvent parameters. These effects usually lead to an expansion in Rf values. Nonetheless, in the event of layer thickness, the Rf value would diminish as the mobile phase moves slower up the plate. If it is desired to articulate positions relative to the position of another substance, x, the Rx (relative retention value) can be determined:
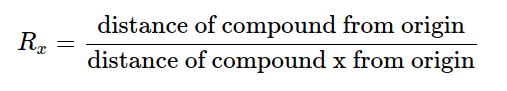
Although Rf can never exceed 1, Rx can be more rapid than the standard compound x.
Equipment
Plates (Stationary Phase)
Earlier, it was mentioned that TLC plates (also known as chromatoplates) can be made in the lab, but are more frequently purchased. Silica gel and alumina are the most typical stationary phases, though other options are accessible. Many plates include a component that glows under short-wave ultraviolet (254 nm). The backing of TLC plates is usually made up of glass, aluminum, or plastic. Glass plates are chemically inert and can withstand aggressive stains and heat, but can be fragile and hard to cut. Aluminum and plastic plates are simple to cut with scissors, however aluminum may not be able to handle strongly acidic or oxidizing stains, and plastic does not withstand the high temperature necessary to develop many stains. Additionally, aluminum and plastic plates are supple, which could cause flaking of the stationary phase. It is very important to never touch the surface of a TLC plate with your fingers as contamination from skin oils or deposits on gloves can distort results. Instead, they should be handled by the edges, or with forceps.
When choosing the stationary phase, the characteristics of your sample should be taken into account. Silica gel can only be used for amino acids and hydrocarbons. It is essential to remember that silica gel is acidic. Accordingly, silica gel does not offer good separation of basic samples and can cause harm to acid-labile molecules. This is also true for alumina plates in acidic solutions. It is important to note the differences between silica gel and alumina. Alumina is basic and will not separate sample sizes as large as silica gel would at a given layer thickness. Moreover, alumina is more chemically reactive than silica gel and so, more caution should be taken with compounds and compound classes to prevent decomposition and rearrangement of the sample.
Solvents (Mobile Phases)
Selecting the proper solvent is most likely the most significant component of TLC, and discovering the best solvent may necessitate a certain amount of trial and error. As with plate selection, keep in mind the chemical properties of the analytes. A usual starting solvent is 1:1 hexane:ethyl acetate. Changing the ratio can have a marked effect of Rf. Rf values range from 0 to 1 with 0 implying that the solvent polarity is very low and 1 implying that the solvent polarity is very high. When executing your experiment, you do not want your values to be 0 or 1 since your components that you are isolating have different polarities. If the value is 0, you need to amplify your solvent polarity since the sample is not moving and adhering to the stationary phase. If the value is 1, you need to decrease your solvent polarity since the compound was not able to separate.
If you are aware that one component of a mixture is insoluble in a given solvent, but another component is freely dissolved in it, it often gives good separations. How fast the compounds travel up the plate depends on two things:
- If the compound is soluble in the solvent, it will travel further up the TLC plate
- How well the compound likes the stationary phase. If the compound likes the stationary phase, it will stick to it, which will cause it to not move very far on the chromatogram.
You should be able to determine which by looking at the Rf value.
Acids, bases, and strongly polar compounds often create streaks rather than spots in neutral solvents. Streaks make it difficult to calculate an Rf and may occlude other spots. Adding a few percent of acetic or formic acid to the solvent can correct streaking with acids. Similarly for bases, adding a few percent triethylamine can improve results. For polar compounds adding a few percent methanol can also improve results.
The volatility of solvents should also be taken into consideration when chemical stains are to be used. Any solvent left on the plate may react with the stain and obscure spots. Many solvents can be removed by allowing them to sit on the bench for a few minutes, but very nonvolatile solvents may necessitate time in a vacuum chamber. Volatile solvents should only be used once. If the mobile phase is used repeatedly, results will not be consistent or reproducible.
Useful Solvent Сombinations
- A solvent that can be used for dividing mixtures of strongly polar elements is ethyl acetate : butanol : acetic acid : water, 80:10:5:5.
- To isolate strongly basic substances, make a mixture of 10% NH4OH in methanol, and then a 1 to 10% mixture of this in dichlormethane.
- Mixtures of 10% methanol or less in DCM can be advantageous for segregating polar components.
Pipettes
- Spots are dispensed onto the plate using fine glass pipettes. The pipette should be thin enough to create a precise spot, but thick enough to take up a suitable quantity of analyte. Here is a typical process to make TLC pipettes.
- Heat the tip of a glass capillary in the flame of a Bunsen burner until it is flexible and then spread out the ends until the middle part of the capillary is much skinnier. Break it in two and use the slender end to dispense spots.
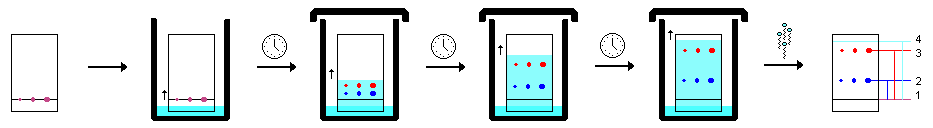
Spotting and Developing
Developing a TLC plate necessitates a developing container or receptacle. This can be as plain as a wide-mouthed jar, but more specialized glassware to fit large plates can be acquired. The holder should comprise an amount of solvent to cover the bottom only. It should also contain a piece of filter paper, or other absorbent stuff to fill the atmosphere with solvent fumes. Lastly, it should have a lid or other cover to reduce vaporization.
- Cut the plate to the right dimensions and, utilizing a pencil (never a pen!), lightly draw a straight line near the bottom. Use no unnecessary pressure when writing on a TLC plate as this will remove the stationary phase. It is crucial to use a pencil in place of a pen because inks usually spread up the plate with the solvent.
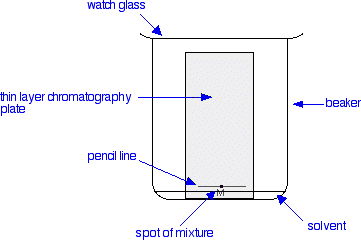
- Make use of TLC pipettes to deposit spots of analyte on the line. Guarantee sufficient sample is spotted on the plate. This can be done by utilizing the short-wave UV. A purple spot should be visible. If the spot is not perceivable, more sample needs to be placed on the plate. If a standard of the target compound is available, it is wise to create a co-spot by spotting the standard onto a spot of the unknown mixture. This verifies the identity of the target compound.

- Insert the plate into the chamber as evenly as doable and lean it against the side. Never let the bulk solvent rise above the line you drew. Allow capillary action to draw the solvent up the plate until it is close to 1 cm from the end. Never permit the solvent to migrate all the way to the end of the plate.

- Remove the plate and right away draw a pencil line across the solvent front.
- Utilize a short-wave UV light and circle the components shown with a pencil.
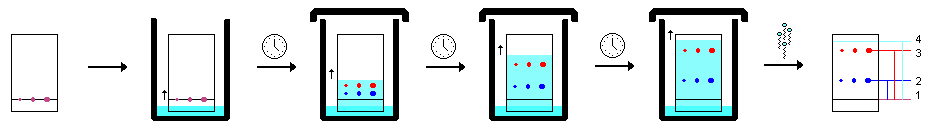
Scheme of eluting procedure
Visualizing
If fluorescent plates are employed, numerous compounds can be discerned by radiating the plate with short-wave UV. Dampening causes dark areas on the plate’s exterior. These dark spots ought to be encircled with a pencil. For compounds that are not UV active, a range of chemical dyes can be utilized. These can be quite general, or they can be exact for a particular molecule or functional group.

Iodine is among the most common dyes. Plates are placed in a vessel containing iodine crystals, or covered in silica gel with iodine dispersed throughout, for around one minute. The majority of organic compounds will be temporarily stained brown. Certain popular general use stains are Permanganate, ceric ammonium molybdate (CAM), and p-anisaldehyde. These can be kept in jars which plates are dipped into, or in spray bottles.

To develop a plate with potassium permanganate, spray or dip the plate and heat it with a heat-gun. Keep the plate face up 10 to 20 cm above the heat gun until the majority of the water evaporates. Then move the plate to 5 to 10 cm above the heat gun and heat it until white/yellow/brown spots appear. Overheating will turn the whole plate brown, masking the spots. If glass plates are used it is often simpler to observe spots through the backing because it is hard to overheat. CAM and p-anisaldehyde stained plates are developed similarly. Overheating CAM stained plates turns everything blue.
Common Problems in TLC
Common issues in TLC that should be circumvented. Usually, these issues can be addressed or evaded if educated on the right skills.
-Over-large Blotches: Spotting measurements of your example should be no bigger than 1-2 mm in width. The component blots will never be bigger than or littler than your sample source stain. In the case that you have an over-large blot, this could bring about overlapping of other component blots with comparable Rf values on your TLC plate. If combining takes place, it would be hard to separate the distinctive components.
-Uneven Progress of Solvent Front: Uneven progress of the mobile phase is a commonplace issue faced in TLC. Consequences would be mistaken Rf values due to the uneven progress of sample source spots. This uneven progress can be caused by a few elements listed below.
- No level base. When setting the TLC plate into the chamber, place the base of the plate on the edge of the chamber (frequently glass holder (e.g. flask)) and incline the top of the plate along the other side of the chamber. Also, make sure that the TLC plate is placed in the chamber equally. Do not slant the plate or seat it at an angle.
- Not enough solvent. There should be enough solvent (relies upon size of the chamber) to travel up the length of the TLC plate.
- Plate is not cut equally. It is suggested that a ruler is utilized so that the plate is cut equally.

Infrequently, water is utilized as a solvent since it creates an uneven bend front which is generally ascribed to its surface tension.
-Streaking: In the event that the sample spot is excessively focused, the substance will travel up the stationary phase as a streak instead of a solitary isolated spot. Put another way, the solvent cannot deal with the focused sample and in result, moves however much of the substance as could be expected up the stationary phase. The substance that it cannot move is left behind. This can be disposed of by weakening the sample arrangement. To guarantee that you have enough arrangement, utilize a short-wave UV light to check whether the spot is noticeable (generally purple in shading), as referenced before.
-Spotting: The sample should be over the solvent level. On the off chance that the solvent level covers the sample, the sample spot will be washed off into the solvent before it goes up the TLC plate. An illustration is shown beneath.
Pros and cons of TLC
TLC is easy to utilize and cost-effective. Undergraduates can be instructed on this technique and apply its analogous principles to other chromatographic methods. Little materials are required for TLC (chamber, watch glass, capillary, plate, solvent, pencil, and UV-light). Thus, when the ideal solvent is identified, it can be applied to other strategies such as High performance liquid chromatography. More than one compound can be detached on a TLC plate provided that the mobile phase is suited for each compound. The solvents for the TLC plate can be shifted easily and it is possible to employ multiple solvents dependent on your desired results. As mentioned earlier, TLC can be employed to confirm purity of a compound. It is very simple to verify the purity using a UV-light. The recognition of most compounds can be completed easily by examining Rf literature values. You can modify the chromatography conditions easily to amplify the optimization for resolution of a particular component.
TLC plates do not have long stationary phases. Accordingly, the length of separation is restricted contrasted to other chromatographic techniques. Additionally, the detection limit is much higher. If you would require a lower detection limit, one would need to use other chromatographic techniques. TLC acts as an open system, so factors such as humidity and temperature can be consequences to the outcomes of your chromatogram.

