- The process of creating a drug - May 23, 2023
- How do scientists model mental disorders in animals? - May 23, 2023
Reactions of organic compounds are driven by the same laws of that one of inorganic compounds, but have some specific features. Inorganic reactions usually involve ions, whereas organic reactions usually involve molecules: some covalent bonds are destroyed and other covalent bonds are formed. The organic compounds reactions have longer reaction time rather than inorganic reactions and usually require additional conditions such as catalyst, elevated temperature or/and pressure. Organic reactions rarely result in high product yields. When the organic reaction is mentioned, it is referred to the prevailed one, but there are always other parallel side reactions. By selecting optimal reaction conditions, chemists try to make sure that the main reaction they want will be carried out completely. That is why the organic reaction yield is never 100%, but usually is less than 50%. The good yield of organic reactions is considered to be 70-80%.
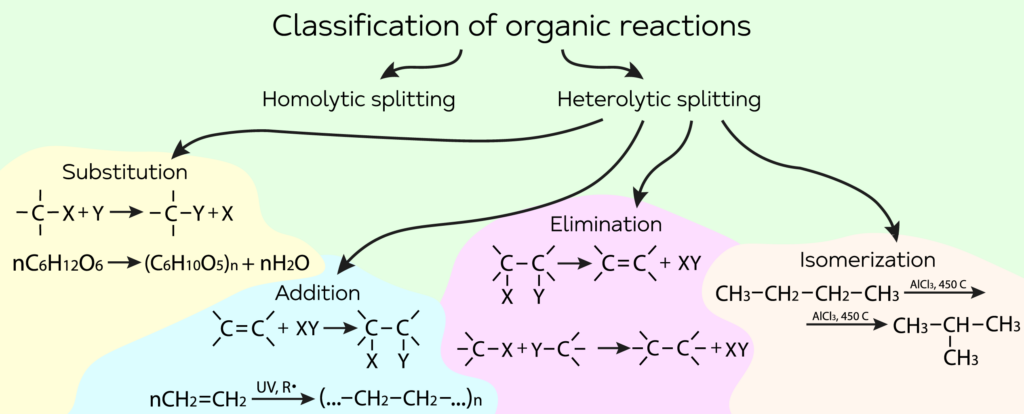
Organic reactions are classified according to the way of chemical bond splitting. Two large reaction groups can be distinguished: radical and ionic reactions.
Radical reactions
Radical reactions are processes that are proceeded with homolytic splitting of covalent bond. As a result of homolytic splitting, an electron pair, forming the bond, is separated with formation of two particles, where each particle (radical) receives one electron:
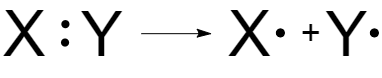
Fig. 1 Homolytic splitting
A neutral atom or particle with an unpaired electron is called a free radical.
Ionic reactions
Ionic reactionsare processes that are proceeded with heterolytic splitting of covalent bond. As a result of heterolytic splitting, the electron pair forming the bond is remained on one of the particles:
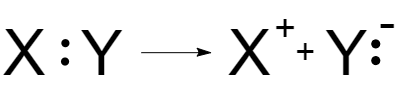
Fig. 2 Heterolytic splitting
As a result of heterolytic splitting, charged particles (nucleophilic and electrophilic) are formed.
Nucleophilic particle (nucleophile) is a particle that have an electron pair on the outer electronic level. A nucleophile is able to form a new covalent bond due to an electron pair.
Electrophilic particle (electrophile) is a particle that have a vacant orbital on the outer electronic level. An electrophile provides unfilled, vacant orbitals to form a new covalent bond with the electron pair of another particle.
Main types of ionic reactions:
- Substitution reactions
An atom or group of atoms replaces another atom or group of atoms in an organic compound molecule. Scheme or substitution reaction:
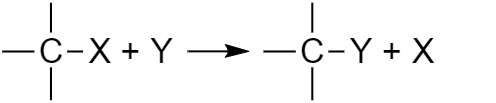
Fig. 3 Scheme of substitution reaction
Example:
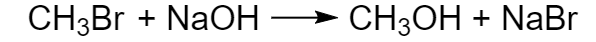
Fig. 4 Substitution reaction of bromomethane and sodium hydroxide
A particular case of substitution reaction is a polycondensation reaction. Polycondensation is a reaction of the polymer formation with a low-molecular-weight side product (for example water):
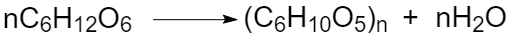
Fig. 5 Polycondensation of glucose to form starch
- Addition reactions
As a result of such reactions, a reagent is combined with a organic substance molecule through an unsaturated multiple (double or triple) bond:
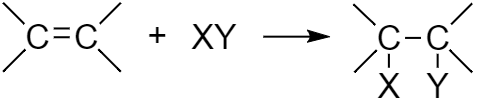
Keep in mind, that electrons are involved in different types of bounds in an unsaturated bonds. One shared electron pair is involved in the σ bond formation and the other shared electron pair is involved in the π bond formation. While addition reaction takes place, the less strong π bond is broken (not the σ bond). Example:
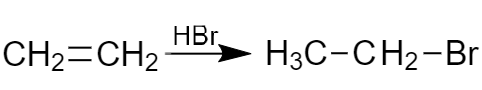
Fig. 7 Addition of hydrogen bromide to ethylene
A particular case of addition reaction is a polymerization reaction. A polymerization is a reaction of the polymer (macromolecule) formation where reagents are low-molecular-weight substances (monomers):
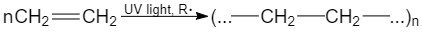
Fig. 8 Polymerization of ethene to form polyethylene
- Elimination reaction
In such reactions two σ bonds (С-X and C-Y) are broken and a new carbon-carbon π bond is formed:
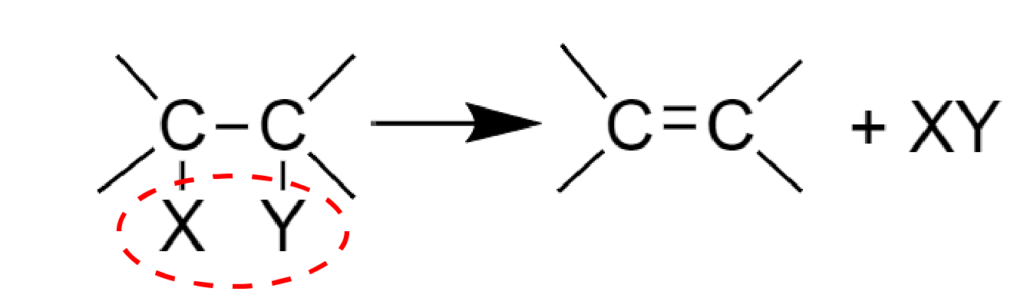
For example, intramolecular dehydration:
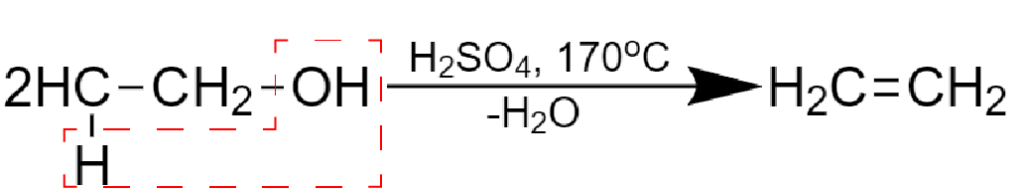
Fig. 10 Intramolecular dehydration
There is another possible mechanism, where two interacting molecules condense into one due to a XY product elimination:
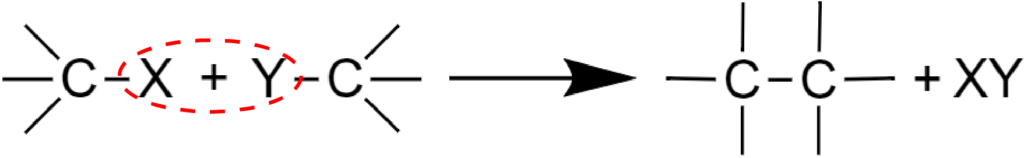
For example, intermolecular alcohols dehydration:
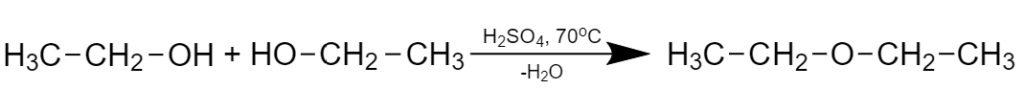
Fig. 12 Intermolecular dehydration
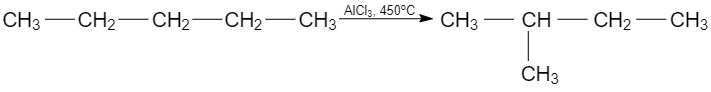
- Isomerization reaction
An isomerization is a reaction when molecules of one substance transform into molecules of another substance with the same quantity composition and same molecular formula.