- The process of creating a drug - May 23, 2023
- How do scientists model mental disorders in animals? - May 23, 2023
Abstract
This article provides information on 4,4′-DMAR, a new psychoactive substance that is a derivative of aminorex, an amphetamine-like appetite suppressant. Aminorex was withdrawn from the market in the 1960s due to its severe cardiovascular toxicity. The article covers general information about 4,4’-DMAR, its physico-chemical properties, recreational use and pharmacology, effects and symptoms of use, street names, prices and approximate dosage, legal status, and synthesis. The article concludes that 4,4′-DMAR has a high potential for abuse and toxic effects, and has been linked to several fatalities. Information from user websites suggests that the substance is used for its desired effects such as euphoria, increased sociability and energy, alertness, and increased confidence, but may cause untoward effects such as increased heart rate, hyperthermia, sweating, agitation, jaw clenching, nausea, and psychosis.
Serotonin vs. Dopamine – 7 Key Differences Between Pleasure and Happiness
General Information About 4,4’-DMARSynthetic Cannabinoid [1,2]
Other synonyms names of 4,4’-DMAR are: 4,4′-Dimethylaminorex; 4,4-Dimethylaminorex; 4,5-Dihydro-4-methyl-5-(4-methylphenyl)-2-oxazolamine; Serotoni; ST; 4-methyl-euphoria; p-methyl-4-methylaminorex; 4-Methyl-5-(p-tolyl)-4,5-dihydrooxazol-2-amine; p-methyl-4-methylaminorex; 4-methylaminorex p-methyl derivative; p4-DMAR; 4-methyl-U4Euh; 4-M-4-MAR.
IUPAC Name of 4,4’-DMAR: 4-methyl-5-(4-methylphenyl)-4,5-dihydro-1,3-oxazol-2-amine
CAS numbers are 1445569-01-6 form not specified and 364064-08-4 free base of the (4S,5S)-enantiomer
Analogs of 4,4’-DMAR are Aminorex; Rexamino; 4′-Fluoroaminorex (4′-FAR); Clominorex; Fluminorex; Methylenedioxyaminorex; 2C-B-aminorex (2C-B-AR); N,N-Dimethylaminorex (N,N-DMAR); Pemoline; Thozalinone; Fenozolone; Cyclazodone; N-Methylcyclazodone; 3-Methylaminorex; 4-Methylaminorex (4-MAR); 4-Ethylaminorex (4-EAR); 4,N-Dimethylaminorex (4,N-DMAR); 3,4-Dimethylaminorex (3,4-DMAR); 2′-Fluoro-4-methylaminorex (2′-F-4-MAR); 3′-Fluoro-4-methylaminorex (3′-F-4-MAR); 4′-Fluoro-4-methylaminorex (4′-F-4-MAR); 4′-Chloro-4-methylaminorex (4C-MAR); 4′-Bromo-4-methylaminorex (4B-MAR); 4′-Methoxy-4-methylaminorex (4′-MeO-4-MAR); 3′,4′,5′-Trimethoxy-4-methylaminorex (TM-4-MAR); 3′,4′-Methylenedioxy-4-methylaminorex (MDMAR) [2]
Physico-Chemical Properties of 4,4’-DMAR [1, 2, 3]
- Molecular Formula C11H14N2O
- Molecular Weight 190.24
- Boiling point 270.8 °C at 760 mmHg
- Flash Point: 117.6 °C
- Color/Form A crystalline solid; The hydrochloride salt form of 4,4′-DMAR is a crystalline solid at room temperature
- Solubilit: DMF: 30 mg/ml; DMSO: 30 mg/ml; Ethanol: 30 mg/ml; PBS (pH 7.2): 10 mg/ml
Structural formula present on Figure 1.
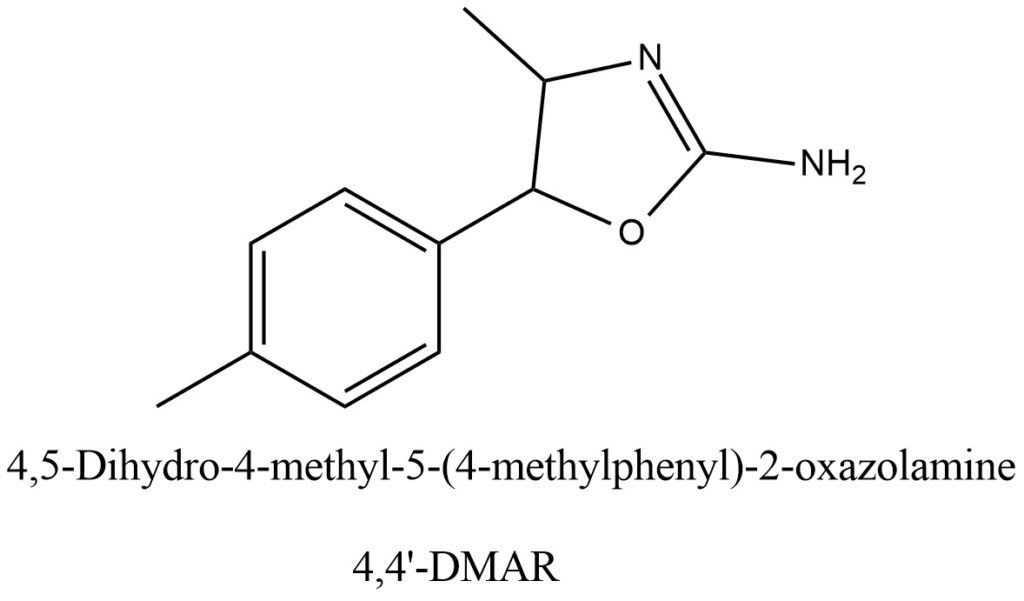
Figure 1. Structure of 4,4’-DMAR
owder and crystalline solid possible of the 4,4’-DMAR can be seen in the pictures provided in Figure 2 and Figure 3.


4,4’-DMAR in various forms can be seen in the pictures Figure 4, Figure 5 and Figure 6
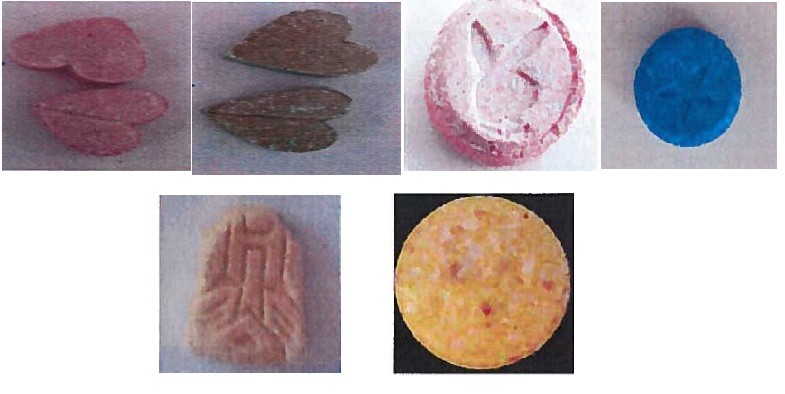
Figure 4. 4,4’-DMAR in Hungary


General Information of 4,4’-DMAR in Recreational Use and Pharmacology [4]
4,4′-DMAR is a recently discovered substance that belongs to the di-substituted 2-amino-5-phenyl oxazoline family. This compound contains a methyl group substitution both on the 4th position of the phenyl ring and the oxazoline ring. It is a derivative of aminorex and 4-methylaminorex (4-MAR), two synthetic stimulants that are strictly controlled under the 1971 United Nations Convention on Psychotropic Substances, with 4-MAR listed in Schedule I and aminorex listed in Schedule IV.
A study has been conducted to investigate the pharmacological properties of (±)-cis-4,4′-DMAR. This study analyzed the effects of (±)-cis-4,4′-DMAR on dopamine, noradrenaline, and serotonin release at the dopamine transporter (DAT), noradrenaline transporter (NET), and serotonin transporter (SERT) using rat brain synaptosomes. The results were then compared with those of d-amphetamine, aminorex, and (±)-cis-4-MAR. All substances were found to exhibit potent releasing activity at DAT, with considerable potency at NET. SERT activity, on the other hand, varied significantly across the four substances, with (±)-cis-4,4′-DMAR showing the highest potency in releasing serotonin. To date, no research has been conducted on the pharmacology of (±)-trans-4,4′-DMAR [5].
Effects and symptoms of 4,4’-DMAR Use [5]
There have been no studies conducted on the subjective effects of 4,4′-DMAR in humans, and the available information is limited to a small number of self-reported experiences from user websites. The onset of the drug is said to be fast, with effects lasting for several hours. The subjective effects are reported to be similar to those of other stimulant-type drugs. However, reports of non-fatal acute toxicity events include hyperthermia, pupil dilation, muscular spasms, seizures, increased perspiration, cardiac and respiratory arrest, agitation, confusion, convulsions, unconsciousness, and paranoid features. The presence of other substances may account for some of the reported effects. The activity of 4,4′-DMAR on catecholamine transporters may also be relevant, especially if the molecule is associated with recreational drugs that alter dopamine and norepinephrine levels. Psychotic episodes can occur if 4,4′-DMAR is taken in combination with other catecholamine-releasing agents such as amphetamine-type stimulants and cocaine. Cardiovascular issues may also result from excessive systemic levels of norepinephrine released. Furthermore, the risk of experiencing a serotonergic syndrome may increase if 4,4′-DMAR is combined with compounds affecting serotonin release or reuptake, such as MDMA or selective serotonin reuptake inhibitors. Some users have reported using 4,4′-DMAR with other drugs, especially alcohol, 6-APB, and phenylpiracetam. Alcohol was reported to potentiate the effects of 4,4′-DMAR, while 6-APB was associated with the drug to cope with social anxiety issues. The chemical structure of 4,4′-DMAR and its analogs is shown in Figure 7.
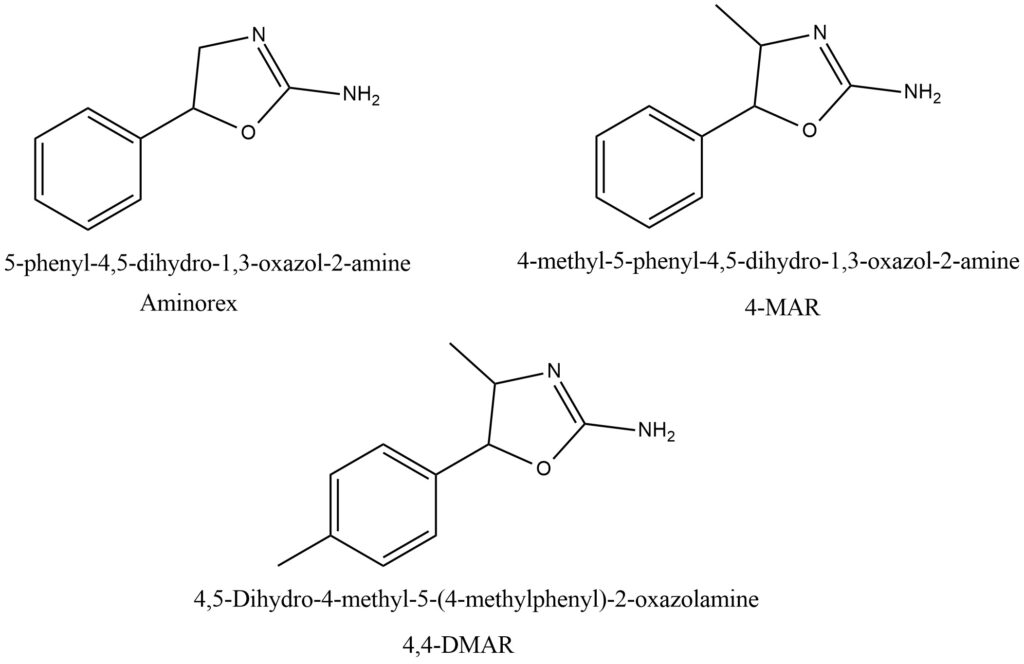
Figure 7. Chemical structures of the three psychostimulants aminorex, 4-methylaminorex (4-MAR) and para-methyl-4-methylaminorex (4,4’-DMAR)
Street Names, Prices and Approximate Dosage [5, 6]
The initial website that offered 4,4′-DMAR as a “research chemical” marketed it solely in powdered form, with various quantities available for purchase ranging from 500 mg (priced at EUR 18.10) to 100 g (priced at EUR 220). Although primarily sold as ecstasy on the black market, 4,4′-DMAR is also known by its street name “ST-60,” with the “ST” potentially standing for “Serotoni” and the “60” indicating a 60 mg dose (see Figure 5). In most instances, 4,4′-DMAR was reported as the sole active ingredient in drug samples, but in approximately 20% of cases, it was found in conjunction with other substances, particularly stimulants like pentedrone, mephedrone, RH-34, 5-APDB, bk-MPA, ethylphenidate, UR-144, and creatine monohydrate. One sample also contained a combination of 4,4′-DMAR and four different cathinones (methcathinone, MPPP, pentedrone, and alpha-PVP), but no quantitative analyses were available at the time [5]. Overall, 27 deaths linked to 4,4′-DMAR were reported in Hungary (eight deaths) and the United Kingdom (19 deaths) between June 2013 and February 2014. Of the decedents, gender and age data were available for 26 individuals, with 19 males (four from Hungary and 15 from the United Kingdom) and seven females (four from Hungary and three from the United Kingdom), ranging in age from 16 to 43 years old.
Limited information from user websites suggests that various doses of 4,4′-DMAR are used. Some sites report “low doses” of 10-15 mg when insufflated or 10-25 mg when taken orally, while others suggest dosages of 30-100 mg or oral doses between 60-200 mg and 65 mg when insufflated. Users have reported taking 4,4′-DMAR on its own or in combination with other new psychoactive substances or controlled drugs.
All 27 deaths related to narcotics involved the detection of one or more new psychoactive substances or controlled drugs post-mortem. Oral doses have been reported ranging from 10-200 mg, with insufflated doses varying from 10-65 mg. Oral consumption is common, with users directly ingesting tablets or powder, or swallowing powder wrapped in cigarette papers. The desired effects of 4,4′-DMAR include euphoria, increased sociability and energy, alertness, and increased confidence. However, users may also experience increased heart rate, hyperthermia, sweating, agitation, jaw clenching, facial spasms, stimulation, nausea, dysphoria, dilated pupils, psychosis, and hallucinations.
Users commonly take sedatives and anxiolytics to reverse the long-lasting stimulant effects of 4,4′-DMAR, which can last up to 16 hours. The desired and untoward effects of the drug are comparable to those observed with other stimulant-type drugs, such as MDMA and mephedrone, which share similar pharmacological properties. Combining 4,4′-DMAR with other drugs, such as synthetic cathinones, amphetamines, and cocaine, has been widely reported and has resulted in several toxicity events and fatalities.
The optimal dose, frequency of redosing, and combination of different routes of administration (ROAs) are widely debated among users. Most users indicated their preferred dosages and ROAs, with 76% preferring oral use, 12% reporting intranasal or vaping ROAs, and 12% reporting a multiple redosing practice involving both oral and intranasal routes. The formulations described were powder and pellets. Oral doses ranged from 10 to 120 mg, intranasal doses varied from 25 to 30 mg, and vaporized doses were in the 10-to 60-mg range. About 25% of users reported a slow onset of psychoactive effects.
Legal Status
DEA Schedule I substances are classified as having no accepted medical use in the United States, a high potential for abuse, and no accepted safety for use under medical supervision. In Germany, these substances are listed under Anlage II and can only be traded by authorized parties, but they cannot be prescribed. In the UK, they are classified as Class A drugs, while in the US they fall under Schedule I. Moreover, they are illegal in both the Czech Republic and Sweden.
Synthesis of 4-methyl-5-(4-methylphenyl)-4,5-dihydro-1,3-oxazol-2-amine [7]
A synthetic method for producing 4,4′-DMAR involves five reaction steps using 4′-methylpropiophenone as the starting material. This precursor is easily obtainable from chemical suppliers. The method begins with the alpha-bromination of 4′-methylpropiophenone, followed by reaction with sodium diformylamide to form the N,N-diformamide intermediate. Acidic hydrolysis of this compound yields 4-methylcathinone, which can then be reduced to 4-methylnorephedrine. The final step involves the conversion of this intermediate to either (±)-cis-4,4′-DMAR using cyanogen bromide, or (±)-trans-4,4′-DMAR using potassium cyanate (as shown in Figure 8).
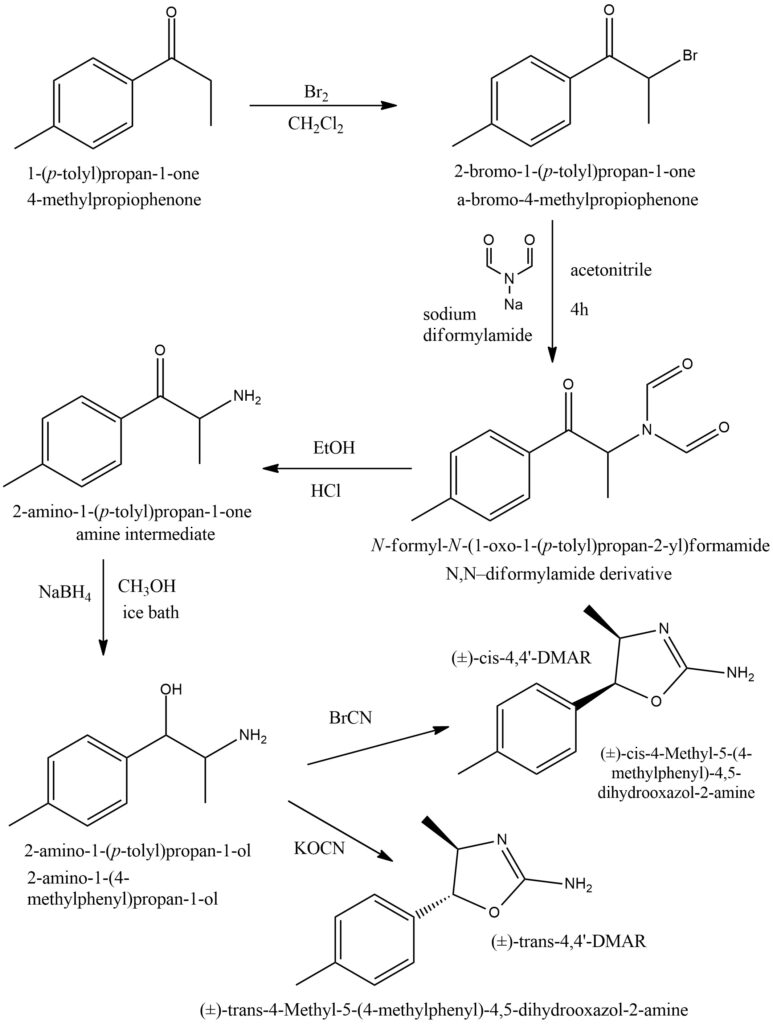
Figure 8. General scheme of 4,4’-DMAR synthesis
Conclusion
4,4′-DMAR is a novel psychoactive substance with high abuse potential and toxic effects, known to have stimulant properties. It is a di-substituted 2-amino-5-phenyl oxazoline substance derived from aminorex and 4-methylaminorex, both synthetic stimulants controlled under the 1971 United Nations Convention on Psychotropic Substances. An in vitro study suggests that (±)-cis-4,4′-DMAR acts as a potent substrate-type releaser at transporters for dopamine, norepinephrine, and serotonin. It has been found in tablet and powder form, with markings resembling ecstasy tablets’ markings. Information from user websites suggests that oral and insufflated doses of 10 to 200 mg have been used. The desired effects of 4,4′-DMAR include euphoria, increased sociability and energy, alertness, and increased confidence, while the untoward effects range from increased heart rate, hyperthermia, sweating, agitation, jaw clenching, nausea, and psychosis. The production of 4,4′-DMAR appears to be straightforward, using 4′-methylpropiophenone as the precursor. This substance has been linked to several fatalities and may be used on its own or in combination with other new psychoactive substances and/or controlled drugs. Sedatives and anxiolytics have been reported to reverse its long-lasting effects.
Drug Trafficking and its Penalties in the UAE
Bibliography
- https://pubchem.ncbi.nlm.nih.gov/compound/20741615
- https://en.wikipedia.org/wiki/List_of_aminorex_analogues
- https://www.caymanchem.com/product/15281
- Anna Rickli, Karolina Kolaczynska etc. Pharmacological characterization of the aminorex analogs 4-MAR, 4,4′-DMAR, and 3,4-DMAR. NeuroToxicology, 2019, Vol. 72, Pages 95-100. https://doi.org/10.1016/j.neuro.2019.02.011 https://www.sciencedirect.com/science/article/abs/pii/S0161813X18305230
- 4,4′-DMAR. EMCDDA–Europol Joint Report on a new psychoactive substance: 4,4′-DMAR (4-methyl-5-(4-methylphenyl)-4,5-dihydrooxazol-2-amine) https://www.emcdda.europa.eu/publications/joint-reports/4-4-DMAR_en
- Barbara Loi, Mire Zloh etc. 4,4′‐Dimethylaminorex (“4,4′‐DMAR”;“Serotoni”) misuse: AWeb‐based study. Hum Psychopharmacol Clin Exp. 2017;32:e2575. https://doi.org/10.1002/hup.2575 https://onlinelibrary.wiley.com/doi/full/10.1002/hup.2575
- Simon D. Brandt, Michael H. Baumann Characterization of a novel and potentially lethal designer drug (±)-cis-para-methyl-4-methylaminorex (4,4′-DMAR, or ‘Serotoni’). Drug Test. Analysis 2014, 6, 684–695. DOI 10.1002/dta.1668 https://analyticalsciencejournals.onlinelibrary.wiley.com/doi/abs/10.1002/dta.1668


