Introduction
Hydrochloric acid is one of the most common and important chemical compounds in industry and research. Hydrochloric acid (HCl) is a chemical compound which is an aqueous solution of hydrogen chloride gas.
Hydrochloric acid was known to alchemists at the end of the 16th century, who obtained it by heating table salt with clay or with iron vitriol.
The aim of this article is to look at the basic chemical aspects of hydrochloric acid, its structure, properties and industrial applications. We will also look at its production methods and safety issues when working with it.
Structure and Properties of Hydrochloric Acid
Chemical Formula and Molecular Structure
Hydrochloric acid, HCl, is a strong inorganic acid, solution of hydrogen chloride in water. The molecular structure of hydrochloric acid is one positively charged hydrogen ion and one negatively charged chloride ion. The salts of hydrochloric acid are called chlorides.
Physical Properties of Hydrochloric Acid
Hydrochloric acid is a characteristic caustic smelling liquid, colorless or light yellow. It has a high density 1.18 g/cm³ and a boiling point about 48°C. In its pure form, hydrochloric acid is usually a solution with a concentration about 37%, also called concentrated hydrochloric acid. In air, it “smokes” as the released HCl forms tiny droplets with water vapor. Higher concentrations up to just over 40% are chemically possible, but the evaporation rate is so high that storage and handling require extra precautions, such as pressurization and cooling.
Chemical Properties of Hydrochloric Acid
Hydrochloric acid is one of the strongest of the mineral acid compounds. When it reacts with water, it is completely ionized. This makes hydrochloric acid strongly acidic and allows it to have a low pH value, the theoretical pH equals 0. It can react with bases to form soluble salts and water.
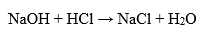
In addition, hydrochloric acid has corrosive properties and can react with many metals, including iron, magnesium, aluminum and zinc to form the corresponding salts and release hydrogen.
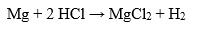
It can also react with metal oxides:
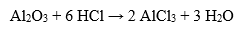
With strong oxidizing agents, hydrochloric acid behaves as a reducing agent, releasing chlorine gas:
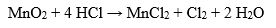
Interacts with ammonia to form thick white smoke consisting of tiny ammonium chloride crystals:
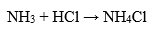

Hydrochloric Acid Synthesis
Hydrochloric acid can be produced by different methods, depending on the raw materials and the technological conditions. One of the main methods is the chloride method, which is based on the reaction of chlorine with hydrogen in the presence of a catalyst, for example platinum. The reaction is as follows:
As the reaction is exothermic, the installation is called an HCl oven or HCl burner. The resulting hydrogen chloride gas is absorbed in deionized water, resulting in chemically pure hydrochloric acid. This reaction can give a very pure product.
Another method is the sulphate method, which is based on the reaction of sulfuric acid with metal chlorides such as sodium chloride (NaCl) or iron chloride (FeCl3). This method is often used in the laboratory. The reaction is as follows:
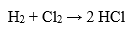

Hydrochloric Acid Applications
Production of chlorides
One of the main uses of hydrochloric acid is the production of various metal chlorides. The reaction is as follows:
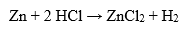
Pickling of Steel
Hydrochloric acid is also used extensively in industry to clean and treat metals. It is used to remove oxides, rust and other impurities from the surface of metal products. Hydrochloric acid reacts with the metal to form the corresponding salts and release hydrogen:
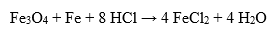
This process produces a clean metal surface and improves the properties of the metal before it is further processed and used.
Chemical Synthesis of Organic Compounds
Hydrochloric acid plays an important role in the chemical synthesis of organic compounds. Halogenation of organic compounds, for example:
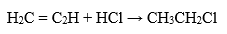
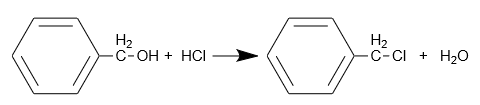
Water Treatment and pH Regulation
Hydrochloric acid is used in water treatment and pH regulation. It is used to neutralize alkalis, remove scales, deposits and for water disinfection. It can be used in drinking water treatment processes, wastewater treatment as well as in swimming pools and industrial cooling systems.
Laboratory Use
The strong mineral acids common in chemistry, hydrochloric acid is the least likely to undergo an oxidation-reduction reaction. It is one of the least dangerous strong acids, despite its acidity. Medium concentrated hydrochloric acid solutions are stable during storage, maintaining their concentration over time. These properties, as well as the fact that it is available as a pure reagent, make hydrochloric acid an excellent acidifying agent. Also, hydrochloric acid is not expensive. Hydrochloric acid is the preferred acid in titration.
The industrial applications of hydrochloric acid are vast and varied, and these examples represent only a few of its many uses. Its chemical properties and reactivity make it a valuable resource for many processes and manufacturing operations.

Safety and Storage of Hydrochloric Acid
Characteristics and hazards of hydrochloric acid
Hydrochloric acid is an acidic chemical compound with high corrosive activity. It is highly reactive and can cause serious hazards if handled improperly. The properties which make hydrochloric acid a hazard include its caustic nature, high toxicity and the possibility of fumes which may be irritating to the respiratory system.
Safe Storage Practices
For the safe storage of hydrochloric acid, special containers made from non-corrosive materials, such as glass or polymeric polyethylene tanks, should be used. These materials must be compatible with and unaffected by hydrochloric acid.
In addition, storage of hydrochloric acid should be carried out in special rooms or storage facilities with good ventilation. It is important to avoid contact with substances that can react with hydrochloric acid and cause dangerous situations.
Precautions
Personal protective equipment such as protective clothing, goggles, masks and gloves must be worn when handling acid. Work with acid must be carried out under a fume hood – toxic fumes must be quickly and efficiently removed from the room. If acid solution comes into contact with skin, rinse with warm water immediately. In case of contact with eyes, rinse with plenty of water. Spilled hydrochloric acid solution can be removed with water and an alkaline solution.
Conclusion
Hydrochloric acid is widely used in industries for production of metal chlorides, purification of metals, chemical synthesis of organic compounds, pH regulation and water treatment. Its importance in various industries emphasizes the need for efficient production, safe storage and handling.
Safety aspects related to the use and storage of hydrochloric acid have been addressed. The need for precautions and the use of special equipment are key aspects to prevent accidents and minimize risks.
Bibliography
- Anion Exchange Studies. IX. Adsorbability of a Number of Metals in Hydrochloric Acid Solutions Kurt A. Kraus, Frederick Nelson, and Gilbert W. Smith Cite this: J. Phys. Chem. 1954, 58, 1, 11–17 Publication Date: January 1, 1954 © American Chemical Society https://doi.org/10.1021/j150511a003
- A METHOD FOR PREPARING STANDARD HYDROCHLORIC ACID SOLUTIONS G. A. Hulett and W. D. Bonner Cite this: J. Am. Chem. Soc. 1909, 31, 3, 390–393 Publication Date: March 1, 1909 https://doi.org/10.1021/ja01933a011
- The Dissolution of Magnesium in Hydrochloric Acid B. Roald1 and W. Beck1 © 1951 ECS – The Electrochemical Society Journal of The Electrochemical Society, Volume 98, Number 7 Citation B. Roald and W. Beck 1951 J. Electrochem. Soc. 98 277 https://doi.org/10.1149/1.2778207
- Titration of Weak Bases in Strong Salt Solutions F. E. Critchfield and J. B. Johnson Anal. Chem. 1958, 30, 7, 1247–1249 Publication Date: July 1, 1958 © American Chemical Society https://doi.org/10.1021/ac60139a017
- Pulmonary effects of experimental graded aspiration of hydrochloric acid Ann Surg. Publication Date: 1969 July; 170(1): 74–86. https://doi.org/10.1097/00000658-196907000-00009