Introduction to Laboratory Glassware
Laboratory glassware is an essential tool used in scientific research and experimentation. It is designed to withstand high temperatures, chemical reactions, and other rigorous conditions that are often encountered in laboratory settings. Laboratory glassware comes in a wide range of shapes and sizes, each with its unique properties and uses. From beakers to test tubes, flasks to pipettes, laboratory glassware is crucial for conducting experiments accurately and efficiently. High-quality laboratory glassware is critical for obtaining reliable results and avoiding contamination, which can lead to inaccurate data and results. Therefore, understanding the properties and uses of different types of laboratory glassware is vital for scientists and researchers working in various fields, including chemistry, biology, physics, and medicine. Not sure what the difference is between a round-bottom flask and a Florence flask, or a pipette and a burette? This topic is here to help. You can find information about the most commonly used glassware in drug manufacturing laboratories below.
Types of Laboratory Glassware and Their Uses
Laboratory beakers and glass rods
Flasks – tall, short, thin-walled cylindrical receptacles with or without a spout that can hold from 5 milliliters to 5 liters and constructed from various materials. Beakers are employed for pouring liquids, concocting solutions, and as receivers in several set-ups. It is not feasible to warm plain glass beakers in a flame, for the reason that this may lead to them shattering. Heating of heat-resistant glasses should only be done in a water bath or any other (sand, oil bath). Heat-resistant glass can tolerate temperatures up to 650 degrees. Laboratory glass rods are intended for stirring solutions in glass laboratory apparatus. Handy for other operations with chemicals.
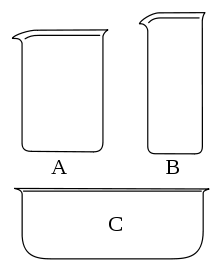
(B) A tall-form or Berzelius beaker.
(C) A flat beaker or crystallizer.


Test tubes
Test tubes are slender cylindrical receptacles with a curved base. They vary in diameter, height and material composition. They are generally employed for analytical and miniature chemistry processes. Moreover, graduated and centrifuge conical tubes also exist. Commonly, test tubes intended for routine chemical experiments are constructed of glass, due to its high temperature tolerance. Tubes made of expansion-resistant glasses, such as borosilicate glass and fused quartz, can withstand temperatures up to multiple hundred Celsius.
Chemistry tubes come in a great variety of sizes and widths, usually from 10 to 20 mm wide and 50 to 200 mm long. Generally, the top segment has a flared lip for easy pouring. The base is generally flat, round or conical. Some test tubes are designed to accept a stopper or a screw cap. A ground glass or white glaze area is sometimes present near the top for labelling with a pencil. Test tubes are extensively used by chemists for managing chemicals, mainly for qualitative experiments and assessments. Their curved base and straight walls reduce substance loss when pouring, permit them to be washed out easily, and enable straightforward observation of their contents. The lengthy, slim neck of the test tube hinders the dispersal of gases to the environment.
Test tubes are practical containers for heating tiny amounts of liquids or solids with a Bunsen burner or alcohol burner. The tube is ordinarily held by its neck using a clamp or tongs. By tilting the tube, its base can be heated to extreme temperatures in the flame, while the neck remains relatively cool, possibly allowing vapours to condense on its walls. A boiling tube is a large test tube particularly intended for boiling liquids. A test tube filled with water and upturned into a water-filled beaker is usually employed to capture gases, such as during electrolysis experimentation. A test tube with a stopper is often used for short-term storage of chemical or biological specimens.


Graduated Cylinders
Containers with graduations indicated on the external wall, intended for measuring specific amounts of liquids in laboratory environment, are called cylinders. They possess a slender cylindrical form. There are four forms of cylinders: one with an outlet, one with a glass stopper, one with a plastic stopper, and one with an outlet and plastic base. Besides cylinders, conical vessels with divisions on the walls, known as beakers, are also used for the same intention.

Laboratory flasks
Laboratory flasks are vessels or receptacles that belong to the category of laboratory equipment recognized as glassware. In laboratory and other scientific situations, they are usually referred to as flasks. Flasks come in a variety of shapes and sizes, but generally have a wider base and one or more cylindrical necks with an opening at the top. Laboratory flasks are sized according to their capacity, usually expressed in metric units such as milliliters (ml) or liters (l). Laboratory flasks were traditionally made of glass, but can also be made of plastic. At the neck of some glass flasks such as round-bottom flasks, retorts, or sometimes volumetric flasks, there are outer (or female) conical ground glass joints. Some flasks, particularly volumetric flasks, have a rubber stopper, bung, or cap to cover the hole at the top of the neck. These stoppers can be made of glass or plastic. Glass stoppers normally have a matching inner (or male) conical ground glass surface, but usually only of stopper quality. Flasks without stoppers or caps can be sealed with a rubber bung or cork stopper. Flasks can be used for creating solutions or for storing, containing, collecting, or sometimes volumetrically measuring chemicals, samples, solutions, etc. for chemical reactions or other activities such as mixing, heating, cooling, dissolving, precipitation, boiling (as in distillation), or analysis.
There are different varieties of lab flasks, each of which serves a distinct purpose in the laboratory. These flasks can be classified into:
Reaction Flasks
Reaction flasks, which are generally globular (i.e. round-bottom flask) and are complemented by their necks, at the terminations of which are ground glass joints to quickly and securely attach to the rest of the equipment (such as a reflux condenser or dropping funnel). The reaction flask is usually constructed of solid glass and is able to withstand large pressure fluctuations, allowing it to be kept in a reaction under vacuum, and pressure, sometimes both at the same time. There is at least one tubular section known as the neck with an opening at the tip. Two-, three- or four-necked flasks are also common. Round bottom flasks are available in a variety of sizes, from 5ml to 20l, with the sizes typically inscribed on the glass.
The necks of the flasks are usually furnished with conically shaped glass connections. These are standardized, and can take any compatible tapered (male) attachments. 24/20 is the norm for 250 ml or bigger beakers, while littler sizes like 14/20 or 19/22 are for smaller beakers. As a result of the curved bottom, cork rings are needed to stabilize the curved bottom flasks when upright. When in use, curved-bottom flasks are usually secured at the neck by clamps on a frame. In pilot plants, even bigger flasks can be seen. Examples of these are:
- Multiple-necked flasks, typically having between two and five, and rarely, six necks, each fitted with ground glass connections, are utilized in more intricate reactions that call for the precise blending of several reagents. They are employed for synthesis.

- A Schlenk flask is a globular container featuring a ground glass joint and a hose adapter with a vacuum stopcock. This valve permits the flask to be linked to a vacuum-nitrogen line via the hose, making it convenient to carry out a reaction in either a vacuum environment or a nitrogen atmosphere.

Distillation Flasks
Distillation flasks (Wurtz flasks) are designed to hold mixtures that are to be distilled, as well as to collect the outcome of the distillation. Distillation flasks can be found in a range of designs. Comparable to the reaction flasks, the distillation flasks as a rule have a single slim neck and a ground glass joint and are constructed of thinner glass than the reaction flask, so that they are simpler to heat. They may be globular, test tube-shaped or pear-shaped, also referred to as Kjeldahl Flasks, due to their utilization with Kjeldahl bulbs.

The Claisen Flasks
The Claisen flasks are typically employed for reduced-pressure distillation. This flask was constructed to minimize the chances of having to conduct the distillation again, caused by the splashing of the boiling liquid. It is comparable to a Würtz flask, though the unique marking of the Claisen flask is a U-shaped neck attached to the top of the flask. The flask itself is usually round-bottom or pear-shaped. The U-shape (or separation) is similar to that of a Claisen adapter, thus the name. This design makes it impossible for any splash of distilling liquid projected by bumping to reach the distillate.
Round-bottom Flasks
Round-bottom flasks are designed in the form of a cylinder rising out of the surface of a sphere. The flasks are typically lengthy in the neck area; sometimes they possess a slit on the neck, which accurately determines the capacity of the flask. They can be used in distillations, or to heat up a substance. These types of flask are also referred to as Florence flasks.
Applications:
- Heating and/or boiling of liquid.
- Distillation.
- Contain chemical reactions.
- Distilling flask in Rotary evaporators.
- Storage of the culture media.
- Preparation of gas-phase standards for flasks fitted with septa (requires volumetric calibration).
The bulbous bases on these varieties of flasks provide evener heating and/or boiling of liquid. Thus, round-bottom flasks are utilized in a multitude of situations where the contents are heated or boiled. Round-bottom flasks are employed in distillation by chemists as distilling flasks and collecting flasks for the distillate (see distillation diagram). One-neck round-bottom flasks are utilized as the distilling flasks in rotary evaporators. This flask shape is also more impervious to fracturing under vacuum, as a sphere more uniformly transmits stress across its surface.
Round-bottom flasks are regularly employed to contain chemical reactions run by chemists, particularly for reflux set-ups and laboratory-scale synthesis. Boiling chips are added to distilling flasks for distillations or boiling chemical reactions to enable a nucleation site for gradual boiling. This nucleation avoids an abrupt boiling surge where the contents may overflow from the boiling flask. Stirring bars or other stirring devices suited for round-bottom flasks are sometimes used. Round bottom flasks have lower stirring efficiency when compared with Erlenmeyer flasks, as they can’t take large stir bars and material can become caught at the base. For a reflux set-up, a condenser is usually affixed to the middle or only neck of the flask being used. Additional necks on a flask could permit a thermometer or a mechanical stirrer to be inserted into the flask contents. The additional necks can also permit a dropping funnel to be attached to let reactants slowly drip in. Special electrically powered heating mantles are available in various sizes into which the bottoms of round-bottom flasks can fit so that the contents of a flask can be heated for distillation, chemical reactions, boiling, etc. Heating can also be accomplished by submerging the bottom of the flask into a heat bath, water bath, or sand bath. Similarly, cooling can be accomplished by partial submerging into a cooling bath, filled with e.g. cold water, ice, eutectic mixtures, dry ice/solvent mixtures, or liquid nitrogen. For gas preparation where heating is required. Since the flask is round bottomed, heat is evenly dispersed throughout on heating.


An Erlenmeyer Flask
An Erlenmeyer flask, also referred to as a conical flask or a titration flask, is a kind of laboratory flask having a flat bottom, a conical body, and a cylindrical neck. Erlenmeyer flasks feature large bases, with sides that gradually ascend to a short vertical neck. They could be graduated, and often spots of ground glass or enamel where they can be marked with a pencil. It differs from the beaker in its slanted body and thin neck. Depending on the purpose, they could be made from glass or plastic, in a broad array of volumes. The mouth of the Erlenmeyer flask might have a beaded lip that can be blocked or covered. Alternatively, the neck could be fitted with ground glass or other connector for utilization with more specialized stoppers or connection to other apparatus. A Büchner flask is a common design modification for filtration under vacuum.
The slanting sides and thin neck of this flask allow the contents of the flask to be mixed by swirling, without the risk of spilling. These features similarly make the flask ideal for boiling liquids. Hot vapor condenses on the upper part of the Erlenmeyer flask, reducing solvent loss. Erlenmeyer flasks’ thin necks can also sustain filter funnels. The last two characteristics of Erlenmeyer flasks make them particularly appropriate for recrystallization. The sample to be refined is heated to a boil, and adequate solvent is added for full dissolution. The receiving flask is filled with a small amount of solvent, and heated to a boil. The hot mixture is filtered through a notched filter paper into the receiving flask. Hot vapors from the boiling solvent keep the filter funnel warm, avoiding premature crystallization. Like beakers, Erlenmeyer flasks are not normally suitable for exact volumetric measurements. Their stamped volumes are approximate within about 5% accuracy.

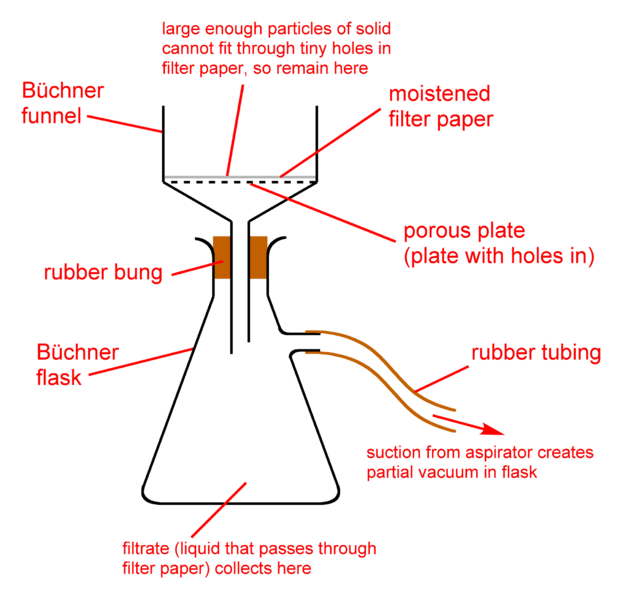
Büchner Flask and Funnel
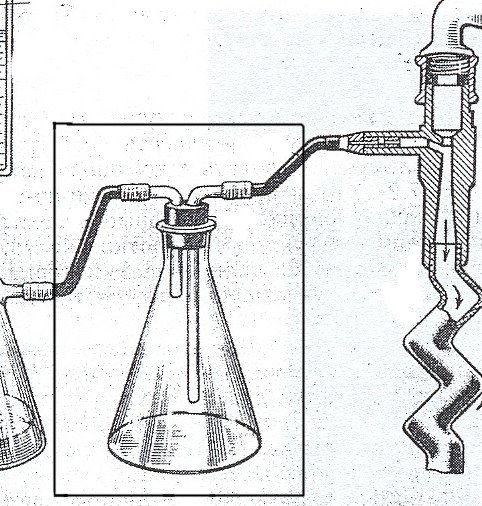
A Büchner flask, also known as a vacuum flask, filter flask, suction flask, side-arm flask, Kitasato flask or Bunsen flask, is a thick-walled Erlenmeyer flask with a short glass tube and hose barb protruding about an inch from its neck. The short tube and hose barb effectively act as an adapter over which the end of a thick-walled flexible hose (tubing) can be fitted to form a connection to the flask. The other end of the hose can be connected to a source of vacuum such as an aspirator, vacuum pump, or house vacuum. Preferably, this is done through a trap (Wolfe’s Flask), which is designed to prevent the sucking back of water from the aspirator into the Büchner flask.



The dense construction of the Büchner flask gives it the ability to bear the pressure variation while maintaining a vacuum inside. Usually, it is used with a Büchner funnel connected through a rubber stopper with a hole in it or a rubber adapter (a Büchner ring) at the bottle neck. The Büchner funnel holds the sample, separated from the suction by a filter paper. During filtration, the solution is contained within the flask, while the solid stays on the filter paper in the funnel.
In addition, the Büchner flask can be utilized as a vacuum catch in a vacuum line to ensure that no liquids are transferred from the vacuum pump (or other vacuum source) to the evacuated apparatus or the other way around.
Fritted Glass (Schott filter)
Funnels equipped with a Schott filter, composed of frittered glass, are utilized in chemical laboratories. Frittered glass is a finely porous glass, allowing liquids or gases to pass through. It is formed through the sintering of glass particles, resulting in a strong but porous material. The frit is implemented in lab glassware, such as filters, scrubbers, and spargers. Other uses for frittered glass in labs are for packing in chromatography columns and resin beds for special chemical synthesis. However, due to the glass particles being held together with small contact areas, frits cannot be employed in highly alkaline environments, which can dissolve the glass, thus leading to the frit eventually disintegrating.

Wolfe’s Flask
Wolfe’s flask acts as a barrier to prevent water from getting into the vacuum unit in the event of an unexpected “surge” of the pump caused by fluctuations in the water supply system, and also in the case of an unintended discharge of liquids from the facility and prevent them from directly entering the water-jet pump. A tube from the water-jet pump is connected to one side pipe, and a tube from the facility to the other side pipe. The infiltration of water into the facility is not allowed for many reasons. In some circumstances, for example when distilling high boiling liquids under vacuum, this could lead to an explosion.
Funels
Laboratory funnels are tools crafted for use in the chemical lab. There are various types of funnels tailored to these specialized needs. Filter funnels, thistle funnels (modeled on thistle flowers) and dropping funnels have stoppers to allow liquids to trickle into a flask. For solids, an apparatus with a wide, brief stem known as a powder funnel is more suitable since it does not obstruct quickly. When in use with filter paper, filter funnels, Buchner and Hirsch funnels are employed to extract fine particles from a liquid by filtration. For more challenging functions, the filter paper in the latter two can be substituted with a sintered glass frit. Separatory funnels are utilized in liquid-liquid extractions.
Plain funnels are manufactured with various sizes of necks. They are employed to pour liquids, to divide solids from liquids by filtration. Usually, a cone-shaped piece of filter paper is folded into a cone and set within the funnel. The solid-liquid blend is then poured through the funnel. The solid particles are too large to move through the filter paper, thus they stay on the paper, however the much smaller liquid molecules flow through the paper to a container located below the funnel, forming a filtrate. The filter paper is only used once. If just the liquid is of interest, the paper is discarded.

Dropping Funnels
A dropping funnel is an apparatus in a laboratory utilized to shift liquids. It has a stopcock which enables controlling the flow. This tool is beneficial when introducing reagents slowly, i.e. drop-by-drop, as rapid addition might cause side reactions or be too powerful.
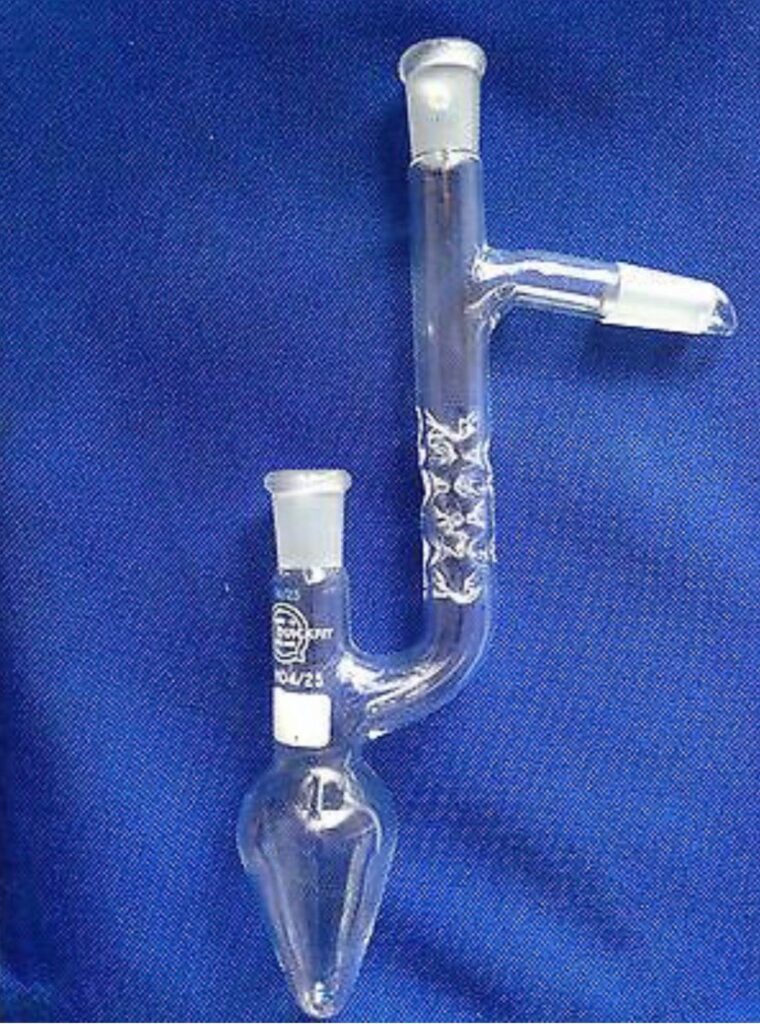
Generally, the funnel is linked with a ground glass joint which allows it to be linked to, e.g. a round bottom flask securely. This also implies that the funnel does not have to be affixed separately. Pressure-equalizing dropping funnels have a different slim-bore glass tube from the funnel’s bulb to the ground glass joint around the stem. This replaces the liquid amount lost in the bulb with the equivalent gas volume from the flask into which the reagent is streaming and is useful when dealing with air-sensitive reagents in a locked, inert-gas environment. Without this tube, or a different way to balance the pressure between a sealed receiving flask and the funnel’s bulb, the flow of fluid from the bulb will quickly come to a stop.


Pay attention to the valve, glass tube on the right, and sealed glass joint on this pressure-equalizing dropping funnel. An average dropping funnel does not include the pressure-equalizing glass tube on the right side.
Separatory Funnels
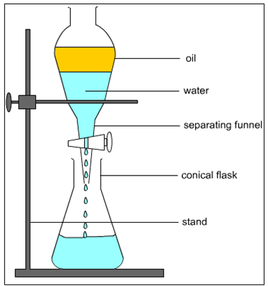
A separatory funnel, otherwise known as a divider funnel, separating funnel, or informally sep. funnel, is a bit of laboratory glassware utilized in liquid-liquid extractions to isolate (partition) the segments of a blend into two non-miscible solvent stages of various densities. Regularly, one of the stages will be aqueous, and the other a lipophilic natural dissolvable like ether, MTBE, dichloromethane, chloroform, or ethyl acetate. Every one of these solvents structure a definite separation between the two fluids. The more thick liquid, normally the aqueous stage except if the natural stage is halogenated, sinks and can be depleted out through a valve far from the less thick liquid, which stays in the separatory funnel. A separating funnel takes the state of a cone with a hemispherical end. It has a stopper at the top and stopcock (tap), at the base. Separating funnels utilized in research centers are typically made of borosilicate glass, and their stopcocks are made of glass or PTFE. Typical sizes are between 30ml and 3l. In modern science they can be a lot bigger and for a lot bigger volumes centrifuges are utilized. The inclining sides are intended to improve the recognizable proof of the layers. The stopcock-controlled outlet is planned to deplete the fluid out of the funnel. On head of the funnel there is a standard taper joint which fits with a ground glass or Teflon stopper. To utilize a separating funnel, the two stages and the blend to be isolated in arrangement are added through the top with the stopcock at the base shut. The funnel is at that point shut and shaken delicately by turning around the funnel a few times; if the two arrangements are blended together too forcefully, emulsions will shape. The funnel is then turned around, and the stopcock cautiously opened to discharge abundance vapor pressure. The separating funnel is set aside to permit for the total division of the stages. The top and the base stopcock are then opened and the lower stage is discharged by gravitation. The top should be opened while discharging the lower stage to permit pressure equalization between the inside of the funnel and the environment. When the base layer has been eliminated, the stopcock is shut and the upper layer is poured out through the top into another compartment.
A separating flask is deployed. The organic layer (yellow, upper layer) has a lower gravity than the aqueous layer (green, lower layer). The aqueous layer is being poured into the container.

Funnels used in organic chemistry can be employed to perform reactions which include:
- Halogenation,
- Nitration,
- Alkylation,
- Acylation,
- Reduction,
- Organomagnesium synthesis, etc.
Prior to using the separating funnel, the valve portion is coated with petroleum jelly or a specialized lubricant (vacuum lubricant) to make it easier to open, then a solution is loaded into the funnel itself plus (if applicable) a solvent, which is employed to pre-rinse the reaction flask. The liquid should not surpass 2/3 of the funnel’s capacity (usually ranging from 1/5 to 1/3), then it is stoppered and agitated. Subsequently, by turning the stopper down and securing it, the tap has to be opened. This is essential for the funnel’s airspace to be saturated with solvent fumes and the pressure in the funnel to remain stable. When the vapor pressure of the solvent is constant and the dissolved gases have been eliminated, the funnel should be vigorously swirled, eventually the funnel is placed into the stand’s rings and the liquids are allowed to separate. Afterward, the stopper is opened and the bottom layer is drained through the tap, while the top layer (if required) is poured through the funnel’s neck.
Сondensers
In chemistry, a condenser is a laboratory device used to transform vapors into liquids by cooling them. They are frequently employed in laboratory operations such as distillation, reflux, and extraction. With distillation, a mixture is heated until the more volatile components evaporate, the vapors are condensed, and collected in a different receptacle. In reflux, a reaction involving volatile liquids is performed at their boiling point, to hasten it; and the vapors that come off are condensed and sent back to the reaction vessel. In Soxhlet extraction, a hot solvent is infused onto some powdered material, such as ground seeds, to leach out some insoluble element; the solvent is then mechanically distilled out of the resulting solution, condensed, and infused again. Multiple types of condensers have been designed for various applications and processing volumes. The simplest and oldest condenser is merely a long pipe through which the vapors are directed, with the outside air providing the cooling. More regularly, a condenser has a separate tube or outer chamber through which water (or some other fluid) is circulated, to give a more effective cooling.
Look at the Distillation and distillation systems topic for more information.
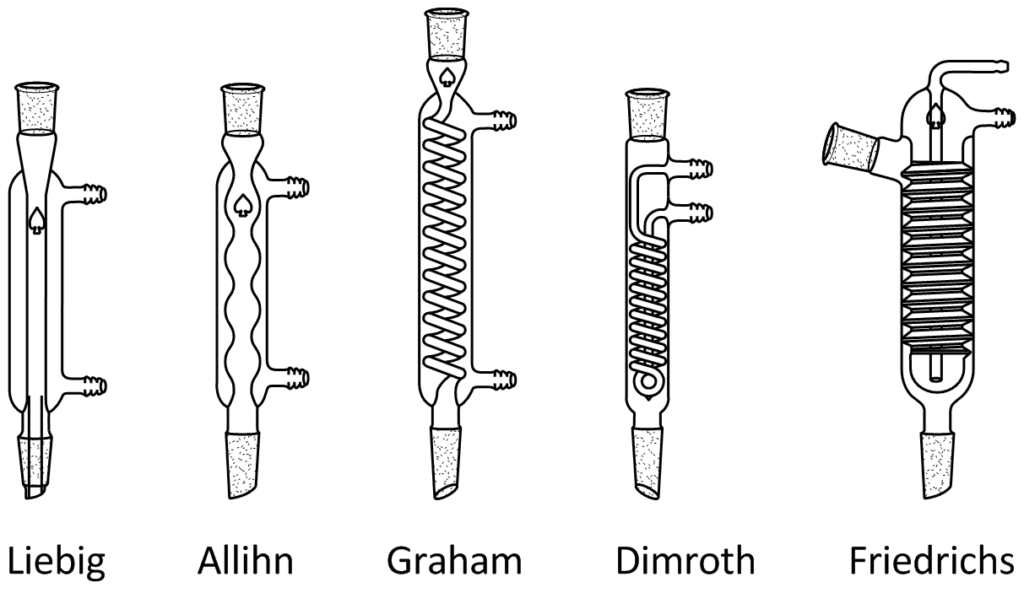
A reflux condenser is a type of laboratory glassware used to chill vapors. It is composed of a glass tube enclosed by a glass cylinder. The tube links the fractioning column and flask, carrying hot vapor created from heating. Water is present in the glass cylinder, and the water is pumped in and out of the cylinder through its side arms. The water cools the vapor within the tube and causes it to condense. Two sorts of reflux condensers exist. As the vapor condenses, it flows back into the reaction flask, thereby cutting down the amount of solvent lost during the reaction. Moreover, the reaction can be conducted for an extended time due to the recycling of the solvent back into the reaction flask. The condenser is mainly used in distillation, a process in which two liquids are separated through heating. The liquid with the lower boiling point will vaporize first and eventually become liquid again in the condenser. When the condenser returns the liquid to the reaction flask, it is referred to as a reflux condenser. There are two main kinds of reflux condensers: air cooled and water cooled. The frequent air cooled reflux condensers are the air condenser and the Vigreux condenser. The simplest water cooled reflux condenser is the Liebig condenser. The Dimroth condenser and the Graham condenser are other water cooled reflux condensers. The air cooled reflux condenser has only one glass tube, and the vapors condense on the glass as they are cooled by the air. Some air cooled reflux condensers are filled with glass beads to facilitate the condensation process. The Vigreux condenser has a series of indentations designed to increase the area for the vapor to condense on. The water cooled reflux condenser has two glass tubes. The inner tube carries the hot vapor, while the outer tube contains the water. Water is employed to cool the vapor. The Liebig condenser has a straight inner tube, while the Graham condenser features a spiral inner tube. There is a double spiral tube inside the Dimroth condenser.
Ground Glass Joints and Adapters
This particular type of glassware, generally referred to as Quickfit, contains an extensive selection of parts integrating standard-taper ground-glass joints. These joints are interchangeable with any of the same size, allowing for the assembly of an array of experiments without the use of rubber stoppers, corks, and so on. Whenever the sizes of the components of the glassware don’t match, adapters for reduction and extension can be put to use. Below are images of a typical selection of jointed glassware.
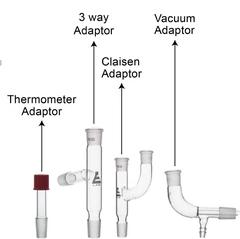
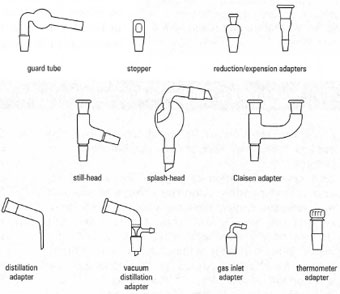
The ground-glass joint on the glassware is distinguished based on the diameter at its maximum width (inside diameter) and the length of the ground-glass part of the joint. Therefore, a 14/23 joint has an inner diameter of 14 mm and a length of 23 mm. Other usual joint sizes that you will often come across are 19/26, 24/29 and 35/39. The joint size is always inscribed on the glass close to or on the joint. For clear reasons, joints are classified as ‘female’ and ‘male’.

Jointed glassware is much pricier than regular glassware due to the accuracy needed for constructing the joints. If the joints become immovable and can’t be taken apart, the glassware is no longer usable, and you may encounter the issue of a sealed flask containing a flammable organic liquid, which someone has to open!
There are two primary reasons for “locked” joints:
- Employing solutions of potassium hydroxide or sodium hydroxide in liquid or other liquids, which corrode the glass.
- Incarcerating substances, including solids and mixtures of solids, in the ground-glass joints.
If you are utilizing jointed glassware with strong alkalis (NaOH, KOH), you must lubricate the joints. In many cases, a straightforward hydrocarbon-based lubricant, such as petroleum jelly, will be enough, as it is effortlessly eliminated from the joints by wiping with a cloth damp with a hydrocarbon solvent (petroleum spirit, b.pt. 60- 80 °C). Abstain from silicone-based grease, since this is hard to remove, soluble in some organic solvents and may taint your reaction products. To grease a joint, put a little smear of grease on the upper part of the ‘male’ joint, press it into the ‘female’ joint with a twisting movement and the joint should become ‘clear’ from the top to about halfway down. If more than half the joint has become ‘clear’, you have utilized an excessive amount of grease: isolate the joints, clean with a solvent-soaked cloth and rehash the procedure. To stay away from catching chemicals in the ground-glass joints, fill flasks etc. utilizing a long-stemmed filter funnel or paper cone, which extends past the joint into the flask.
The Claisen Adapter
The Claisen adapter can be placed on top of a round bottom flask to divide one port into two, For instance, join one upper portion of the Claisen adapter to a condenser and one to an extra funnel or accept an adapter for temperature measurements in a distillation set-up; This Claisen adapter has two upper external joints to affix any lab ware with inner joints, and a lower internal joint to go into a boiling flask with external joint. The sizes of the three joints are the same 24/40. Labor Glass Claisen adapter is constructed of high-quality borosilicate glass and tempered at 800 degrees Celsius, can be heated directly in an open flame and can endure regular laboratory thermal changes in chemistry processes such as heating and cooling.


Design
The Claisen adapter can be affixed to the top of a round bottom flask to divide one aperture into two. For instance, secure one outer joint of the Claisen adapter to a condenser and one to an auxiliary funnel or absorb a thermometer adapter for temperature readings in a distillation setup; This Claisen adapter has two external upper joints to link any laboratory glassware with internal joints, and a lower inner joint to fit into a boiling flask with an outer joint.
Use
This type of round-bottom flask is typically employed in situations that require multiple outlets. Its design is well-suited for a reflux procedure, with one opening fitting a glass condenser and the other accommodating an extra funnel. Usually, it is utilized in a distillation set-up, installed on the distillation flask. The extra neck can be utilized to add water to the boiling flask during the distillation process.
The 3-joint Claisen adapter is equipped with three 24/40 standard taper connections for conveniently and securely connecting laboratory glassware. The upper two ports are female to affix the distillation head and a supplementary funnel or powder funnel.
Conclusion
In conclusion, laboratory glassware is a critical component in many laboratory experiments and procedures. The variety of glassware available is designed to meet specific needs, and each type of glassware has its unique features and functions. Proper handling and cleaning of laboratory glassware are essential to ensure accurate and reliable results in experiments. Therefore, it is crucial for laboratory professionals to have a good understanding of laboratory glassware’s properties and applications to ensure their safe and effective use in scientific research. Overall, laboratory glassware is a fundamental tool in scientific research, and its importance cannot be overstated.
Bibliographic Information
DAVIDSON, J. British Laboratory and Scientific Glassware. Nature 107, 331–332 (1921). https://doi.org/10.1038/107331b0
Use of Glass Apparatus in the Laboratory and in Industry. Nature 164, 907 (1949). https://doi.org/10.1038/164907a0
Espahangizi, Kijan, et al. “The Twofold History of Laboratory Glassware.” Membranes, Surfaces and Boundaries: Interstices in the History of Science, Technology and Culture (2011): 17-33.
Works, Corning Glass, and N. Y. Corning. Laboratory Glassware. The Works, 1951.
Pyrex, Jobling England. “LABORATORY GLASSWARE.”

