- The process of creating a drug - May 23, 2023
- How do scientists model mental disorders in animals? - May 23, 2023
What is a Chemical Structure?
Chemical Structure is the spatial arrangement of atoms in an organic molecule. Chemical structure determines the molecular geometry of the molecule. Russian сhemist A.Butlerov acknowledged that molecules are not random bunch of atoms and functional groups but are arranged in a concrete structure. The molecular structure is determined using a density-sensitive characteristic viscosity detector combined with a light scattering detector to provide molecular weight data.

Types of organic formulas
The molecular formula of a compound shows the exact number of each different atom (element), which is represented in one compound molecule. This formula is useful by reason that it allows to count the molecular weight of the compound, but can also be limiting. It doesn’t give you any information about molecule bonds. Also, might not even tell you about which functional groups are present, depending on how formula is written. Functional groups are parts of organic molecules which determine their individual reactivity with other substances; they were mentioned in a previous post «Classification of organic compounds».
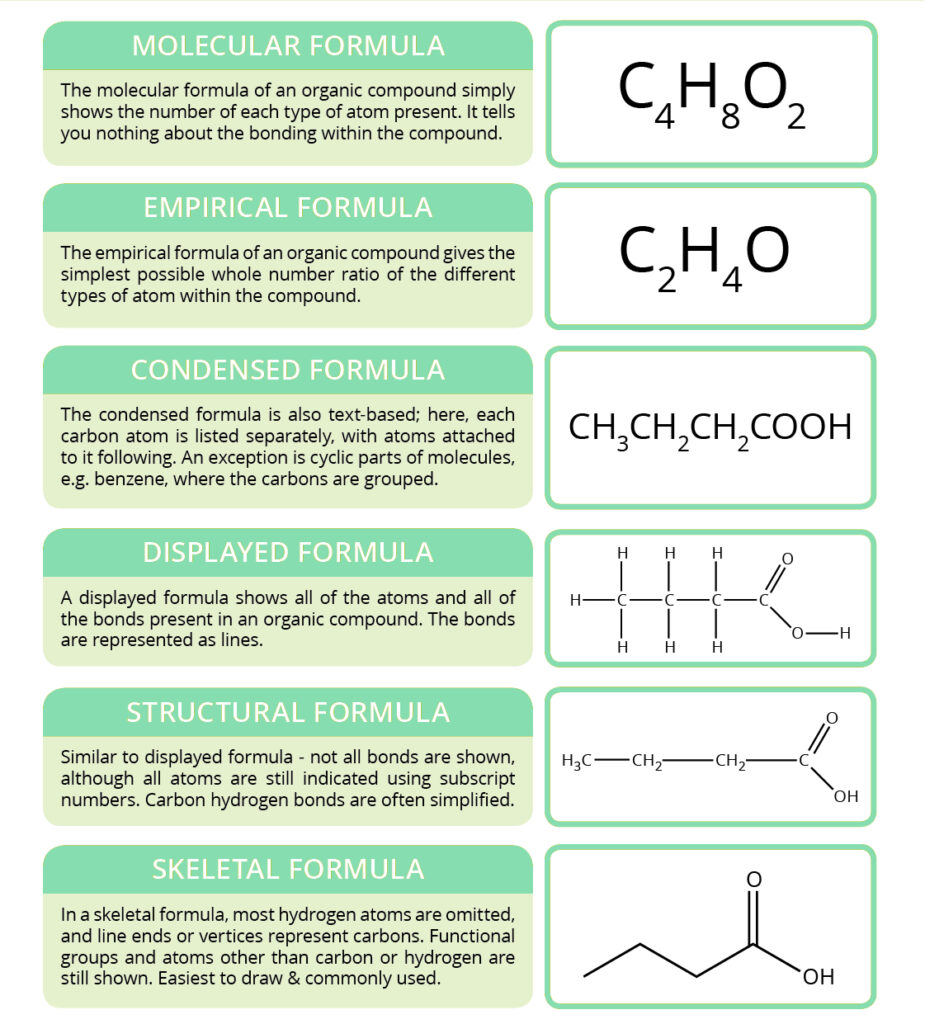
The empirical formula is the simplest elements ratio, which is presented in a molecule. They are used for some organic compounds – they help us sometimes determine the molecular formula in case we don’t know it. Nevertheless, they are much more frequently used for ionic compounds such as table salt, sodium chloride (NaCl). Ionic compounds have huge lattice structures with a large number of oppositely charged ions electrostatically attracted to each other. It wouldn’t be handy to try and write down the exact number of each type of atom present in a salt crystal, because the number would be giant. We just use the simple empirical formula instead.
The condensed formula is often used in texts, and was particularly used in early organic chemistry publications by reason that graphics application was limited. All carbon atoms in a molecule are written out in turn, with atoms attached to them written directly after. Brackets are used to indicate branching in a molecule, or a multiplicity of identical groups. The condensed formula is occasionally seen for simple cyclic compounds, but representing them with this formula usually looks confusing, so it’s unusual.
The displayed formula and structural formula are quite similar. The displayed formula represents the molecule by showing all atoms and all bonds between those atoms. Bonds are represented by lines, with the number of lines representing a type of a bond. Single bonds and double bonds (2 lines) are relatively common, triple bonds (3 lines) are also possible in a lot of molecules. These are evidently useful because they show the molecule in as much detail as possible. On the other side, they also take up a lot of space and time to draw. The structural formula attempts to tackle this by omitting some bonds, and grouping some atoms together, but it still isn’t the quickest way to represent organic molecules.
The skeletal formula is the simplest way of drawing organic molecules and it is commonly seen in textbooks and science articles. It represents the main carbon chain as a zig-zagging line, where the end of lines and vertices mean carbon atoms. All hydrogens are omitted, exept for they are part of a functional group, which are always shown. Heteroatoms are also always shown.
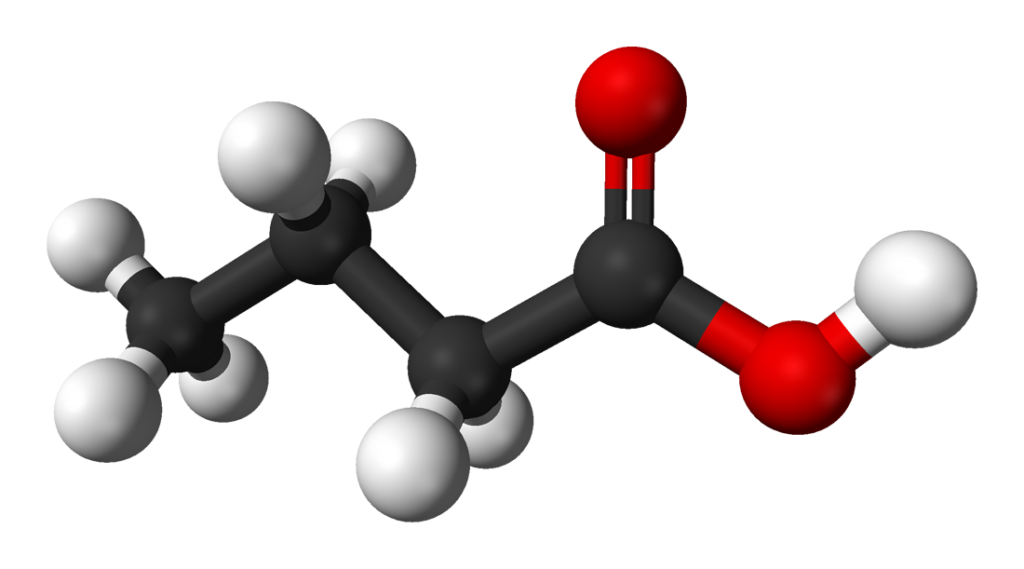
Butanoic acid 3D structural formula
Summary
In this article we have described what molecular structure and types of organic formulas are. This material is informational in nature and does not contain a manual.
Share and comments would be appreciated.
