- The process of creating a drug - May 23, 2023
- How do scientists model mental disorders in animals? - May 23, 2023
Introduction
Morphine, a naturally occurring alkaloid, has captivated scientists and medical professionals for centuries. Derived from the opium poppy plant, morphine has a rich history of therapeutic use as a potent analgesic. It is on the World Health Organization’s List of Essential Medicines. In this article, we delve into the intriguing world of morphine, exploring its chemistry, effects, synthesis, and wide-ranging applications.
Morphine Chemistry: Chemical and Physical Properties
Morphine or (5α,6α)-7,8-Didehydro-4,5-epoxy-17-methylmorphinan-3,6-diol, an opiate alkaloid derived from the opium poppy plant, possesses a fascinating array of chemical and physical properties that contribute to its unique characteristics. At the molecular level, morphine is classified as a benzylisoquinoline alkaloid and features a complex structure composed of 17 carbon atoms, 19 hydrogen atoms, 1 nitrogen atom, and 3 oxygen atoms. Its molecular formula, C17H19NO3, highlights its intricate composition.
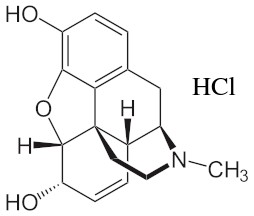
In its pure form, morphine presents itself as a white, odorless crystalline powder. It exhibits sparing solubility in water, meaning it dissolves only to a limited extent. However, morphine readily dissolves in alcohol and acidic solutions, enabling various methods of administration and formulation. These properties allow for the preparation of morphine solutions, suspensions, and solid dosage forms for oral, intravenous, or intramuscular administration.
- Trade names: States; MS Contin; MST Continus; Oramorph; Sevredol; (5α,6α)-7,8-Didehydro-4,5-epoxy-17-methylmorphinan-3,6-diol;
- CAS number: 57-27-2;
- Molar mass: 285.343 g/mol;
- Melting point: 255 °C;
Morphine Synthesis
The initial complete synthesis of morphine, developed by Marshall D. Gates, Jr. in 1952, continues to serve as a prominent illustration of total synthesis. Due to the intricate stereochemical nature and the resulting synthetic difficulties associated with this complex compound, Michael Freemantle has expressed skepticism regarding the feasibility of achieving a chemically synthesized version that would be cost-effective enough to rival the production cost of morphine derived from the opium poppy.
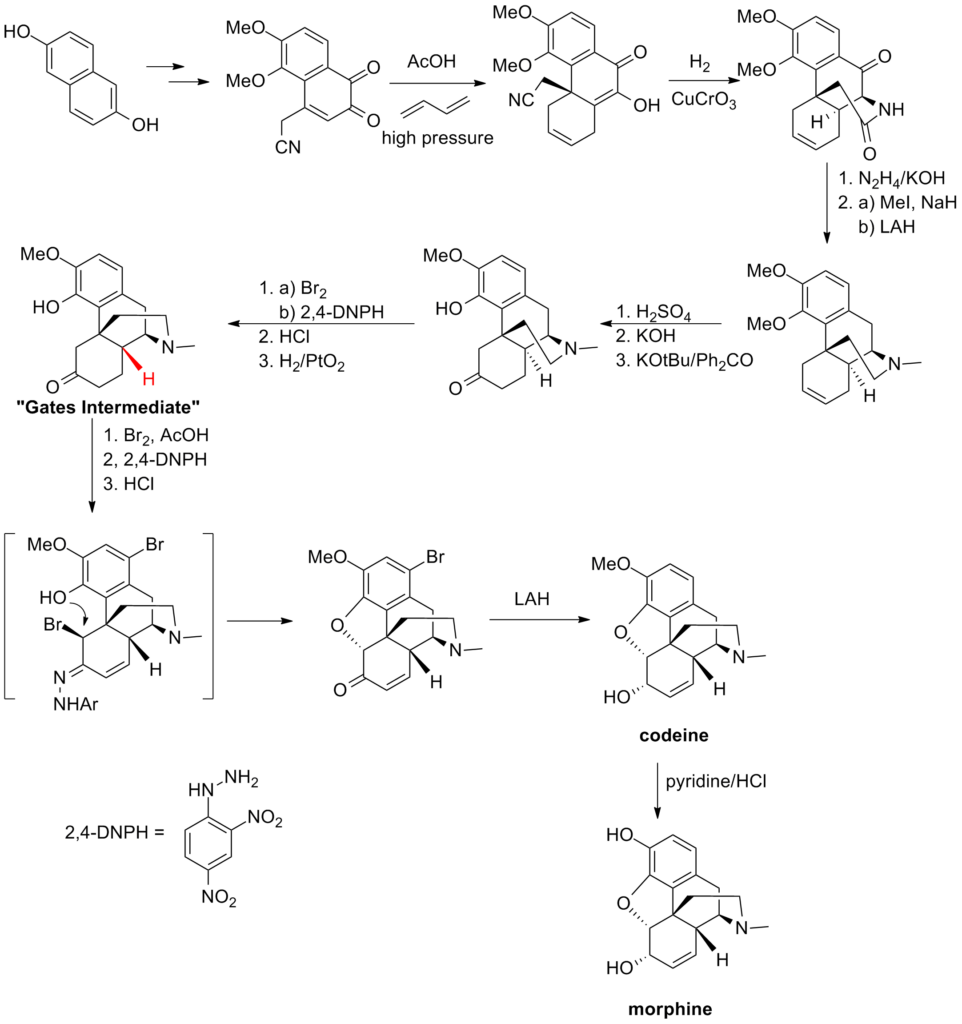
The synthesis was devised by utilizing the dihydroxynaphthalene compound depicted below to construct a cyanodiketone. The crucial stage involved a Diels-Alder reaction under high pressure with butadiene, which formed the fundamental fused ring system. Through reductive coupling between the hydroxyl and cyano groups, the ethylamine bridge was formed, and subsequent modifications (including a challenging epimerization of the highlighted stereocenter) led to the formation of the “Gates Intermediate.” Subsequent syntheses published at that time aimed to enhance the synthesis of this intermediate and refine the final stages.
Pharmacology of Morphine
Morphine exerts its effects by attaching to and activating three opioid receptors known as the κ-opioid (KOP), μ-opioid (MOP), and δ-opioid (DOP) receptors, acting as an agonist. This mechanism is based on the functional resemblance of opioids to the body’s natural endorphins. Endorphins play a role in pain relief, inducing sleepiness, and generating feelings of pleasure and enjoyment. They are believed to be released in response to pain, intense exercise, orgasm, or excitement. By mimicking these natural endorphins, morphine produces euphoria, pain relief, and anxiety reduction.
The human body naturally produces small amounts of morphine, which acts as an immunomodulator. Endogenous morphine preferentially binds to the μ3 opioid receptor.
Morphine, hydromorphone, and oxymorphone undergo minimal metabolism via CYP450 enzymes and are primarily metabolized by UDP-glucuronosyltransferases (UGTs), specifically the UG2B7 isozyme. Approximately 90% of morphine is converted into metabolites, mainly morphine-3-glucuronide (M3G) (45-55%) and morphine-6-glucuronide (M6G) (10-15%). Among these, M3G has low affinity for opioid receptors and lacks analgesic activity. On the other hand, M6G binds to the same receptor sites as morphine but exhibits higher affinity and potency.
The effects of morphine can be reversed by opioid receptor antagonists like naloxone and naltrexone. The development of tolerance to morphine can be hindered by NMDA receptor antagonists such as ketamine, dextromethorphan, and memantine. In long-term pain management, rotating morphine with chemically dissimilar opioids can slow down the development of tolerance, especially with agents that have significantly incomplete cross-tolerance with morphine, such as levorphanol, ketobemidone, piritramide, methadone, and its derivatives. These drugs also possess NMDA antagonist properties. It is believed that either methadone or dextromoramide, as strong opioids, exhibit the most incomplete cross-tolerance with morphine.
Effects of Morphine
Physical effects
- Sedation
- Sensations of profound physical pleasure – The sensations of profound physical pleasure induced by this substance are remarkably potent in comparison to other opioids like codeine or tramadol. This experience can be characterized as an overwhelming sense of physical comfort, warmth, affection, and ecstasy.
- Muscle relaxation
- Constipation
- Cough suppression
- Decreased libido
- Difficulty urinating
- Itchiness
- Nausea
- Pain relief – Many report that they are still aware of pain, however the pain is no longer of interest.
- Pupil constriction
- Respiratory depression
- Skin flushing
- Appetite suppression
- Orgasm suppression
Cognitive effects
- Psychological euphoria – Morphine elicits a psychological euphoria that is significantly more pronounced than other opioids like codeine or tramadol, yet milder in comparison to substances like heroin, hydrocodone, and oxycodone. This sensation can be characterized as an intense and encompassing experience of emotional bliss, satisfaction, and joy.
- Anxiety suppression
- Thought deceleration
- Compulsive redosing
- Dream potentiation
- Irritability – Although opioids are recognized for their mood-enhancing properties, they can also paradoxically heighten the user’s susceptibility to irritable stimuli. This may present as aloofness and abrupt episodes of anger and aggression, commonly referred to as “opiate rage.” Such occurrences tend to be more prevalent during the decline of the experience or with prolonged and intense usage.
Visual effects
- Suppressions
- Double vision – At elevated doses, the eyes undergo involuntary unfocusing and refocusing. As a result, a blurred and double vision effect ensues, persisting regardless of the point of visual focus. This visual disturbance can reach such intensity that tasks like reading or driving become impractical or even impossible.
- Hallucinatory States
- Internal hallucination – During the state of nodding caused by heavy doses, individuals may encounter a condition of partial awareness and hypnagogia, leading to dream-like states and vivid imagery up to level 3. This phenomenon is frequently accompanied by indistinct geometric patterns.
Diverse Applications of Morphine
Morphine, renowned for its potent analgesic properties, finds extensive utility across a broad spectrum of applications within the medical, pharmaceutical, and research fields. Its multifaceted nature makes it a valuable asset in the pursuit of effective pain management and other therapeutic interventions.
In the medical domain, morphine stands as a cornerstone in the treatment of severe pain. It plays a crucial role in managing acute pain resulting from surgical procedures, traumatic injuries, and post-operative recovery. Additionally, morphine finds indispensable application in palliative care, providing much-needed relief for patients with advanced illnesses, such as cancer, where pain management is paramount to improve quality of life.

Pharmaceutical industries harness the power of morphine in the formulation of various medications. It serves as a fundamental building block for the synthesis of other opioids, including codeine and oxycodone. These derivatives exhibit varying levels of potency and are employed in different contexts, ranging from mild to severe pain relief. Morphine’s versatility also extends to the treatment of respiratory diseases, where it aids in suppressing cough reflexes and improving patient comfort.
Research and development efforts continue to unlock new applications for morphine. Its role in scientific studies and discoveries is extensive, with ongoing investigations exploring its potential in neurology, addiction research, psychiatric disorders, and beyond. Morphine serves as a valuable tool to better understand the human body’s response to pain, addiction mechanisms, and the intricate workings of the central nervous system. Such insights pave the way for the development of innovative treatments and therapies.
Moreover, morphine’s significance extends beyond its primary analgesic function. It plays a vital role in anesthesia during surgical procedures, helping induce sedation and reduce pain sensations in patients. This application ensures safe and pain-free surgical interventions, contributing to positive patient outcomes.
Comparative Analysis: Morphine vs. Other Opioids in Effects
When examining the effects of opioids, such as morphine, it becomes essential to understand the distinctions between different substances within this class of drugs. A comparative analysis between morphine and other opioids sheds light on their varying pharmacological profiles, efficacy, and potential side effects.
Morphine, a prototypical opioid, is known for its potent analgesic properties. It acts primarily on the central nervous system by binding to specific opioid receptors, resulting in pain relief and a sense of euphoria. However, its effects extend beyond pain management. Morphine also produces relaxation, sedation, and a feeling of well-being. These effects contribute to its significant therapeutic value in managing severe pain and promoting patient comfort.
Comparatively, other opioids such as codeine, oxycodone, and fentanyl exhibit variations in their pharmacological characteristics and effects. Codeine, for instance, is considered a milder opioid and is often used for moderate pain relief and as an antitussive agent to suppress cough reflexes. Oxycodone, on the other hand, possesses stronger analgesic properties than codeine and is frequently prescribed for more severe pain conditions. Fentanyl, a highly potent synthetic opioid, is renowned for its rapid onset and high potency, making it particularly useful in acute pain management and during surgical procedures.

While all opioids share the ability to alleviate pain, each presents a unique profile of effects and side effects. Morphine, for example, commonly produces adverse effects such as nausea, constipation, sedation, and respiratory depression. These side effects must be closely monitored, especially in higher doses or when used for an extended period. Codeine and oxycodone can also induce similar side effects, albeit with varying intensities. Fentanyl, given its high potency, poses a greater risk of respiratory depression and requires careful administration due to its potential for rapid onset and overdose.
Furthermore, individual patient characteristics, including age, medical history, and genetic factors, can influence the response to opioids. Some individuals may exhibit increased sensitivity to certain opioids, while others may require higher doses to achieve adequate pain relief. Healthcare providers must consider these factors when selecting the most appropriate opioid and dosage for each patient.
Legal Framework of Morphine
Internationally, morphine is categorized as a Schedule I substance in accordance with the Single Convention on Narcotic Drugs. It is included in the WHO Model List of Essential Medicines, which comprises vital medications necessary for a basic healthcare system.
Here are the regulations regarding morphine in various countries:
- Australia: Morphine is classified as a Schedule 8 drug under the State and Territory Poisons Acts.
- Austria: Medical use of morphine is legal under the AMG (Arzneimittelgesetz Österreich), but it is illegal to sell or possess it without a prescription under the SMG (Suchtmittelgesetz Österreich).
- Canada: Morphine is classified as a Schedule I drug under the Controlled Drugs and Substances Act.
- France: Morphine falls under the strictest category of controlled substances based on the French controlled substances law of December 1970.
- Germany: Morphine is a controlled substance listed under Anlage III of the BtMG. It can only be prescribed on a narcotic prescription form.
- Japan: Morphine is considered a narcotic under the Narcotic and Psychotropic Drugs Control Act (麻薬及び向精神薬取締法).
- Netherlands: Morphine is classified as a List 1 drug under the Opium Law.
- Russia: Morphine is categorized as a Schedule II controlled substance.
- Sweden: Morphine is legal for medical use but is a controlled substance.
- Switzerland: Morphine is a controlled substance explicitly mentioned under Verzeichnis A. Medicinal use is permitted.
- Turkey: Morphine is designated as a ‘red prescription’ substance and is illegal to sell or possess without a prescription.
- United Kingdom: Morphine is listed as a Class A drug under the Misuse of Drugs Act 1971 and is classified as a Schedule 2 Controlled Drug under the Misuse of Drugs Regulations 2001.
- United States: Morphine is classified as a Schedule II controlled substance under the Controlled Substances Act, with the main Administrative Controlled Substances Code Number (ACSCN) of ACSCN 9300. Manufacturing quotas are imposed on morphine pharmaceuticals in the US, excluding production for extremely dilute formulations and its use as an intermediate or chemical precursor for other drugs.
Conclusion
Morphine stands as a cornerstone in the field of pain management and pharmaceutical sciences. Its chemical properties, synthesis methods, and diverse applications continue to delight researchers and medical professionals alike. By delving into the chemistry, effects, synthesis, and legal framework of morphine, we gain a deeper understanding of this powerful substance and its contributions to healthcare and scientific advancements. However, it is imperative to use and administer morphine responsibly and with caution to mitigate the risks associated with its potential for abuse and addiction.
Bibliography
- Gates, Marshall, and Gilg Tschudi. “The synthesis of morphine.” Journal of the American Chemical Society 78.7 (1956): 1380-1393. https://pubs.acs.org/doi/abs/10.1021/ja01588a033.
- Linares, Célia Lloret, et al. “Pharmacology of morphine in obese patients: clinical implications.” Clinical pharmacokinetics 48 (2009): 635-651. https://link.springer.com/article/10.2165/11317150-000000000-00000
- Behar, M., et al. “Epidural morphine in treatment of pain.” The lancet 313.8115 (1979): 527-529. https://www.sciencedirect.com/science/article/pii/S0140673679909474
- https://erowid.org/pharms/morphine
- https://psychonautwiki.org/wiki/Morphine
- https://en.wikipedia.org/wiki/Morphine


