- The process of creating a drug - May 23, 2023
- How do scientists model mental disorders in animals? - May 23, 2023
Introduction
Refractometry, a fundamental concept in optics and analytical science, is central to our grasp of how light interacts with various materials. This article delves into the theory and practical applications of refractometry, examining how the refractive index, a unitless value, defines a material’s ability to bend light. We’ll explore the essential principles of Snell’s law, refractometers, and their pivotal role in gauging the purity and composition of substances. Additionally, we’ll discuss the interplay of light and matter, showcasing a real-world experiment that highlights the significance of refractometry in today’s scientific landscape.
Refractometry Theroy
In the field of optics, the refractive index (or simply, refraction index) of an optical medium is a unitless value that provides insight into the medium’s capacity to bend light.
The refractive index dictates the extent to which light alters its course, or refracts, upon entering a substance. This phenomenon is elucidated by Snell’s law of refraction, expressed as n1 sin θ1 = n2 sin θ2, where θ1 and θ2 represent the angle of incidence and angle of refraction, respectively, when a ray traverses the boundary between two materials with refractive indices denoted as n1 and n2. Furthermore, these refractive indices establish the degree of light reflection at the interface, ascertain the critical angle for total internal reflection, govern intensity (as per Fresnel’s equations), and determine Brewster’s angle.
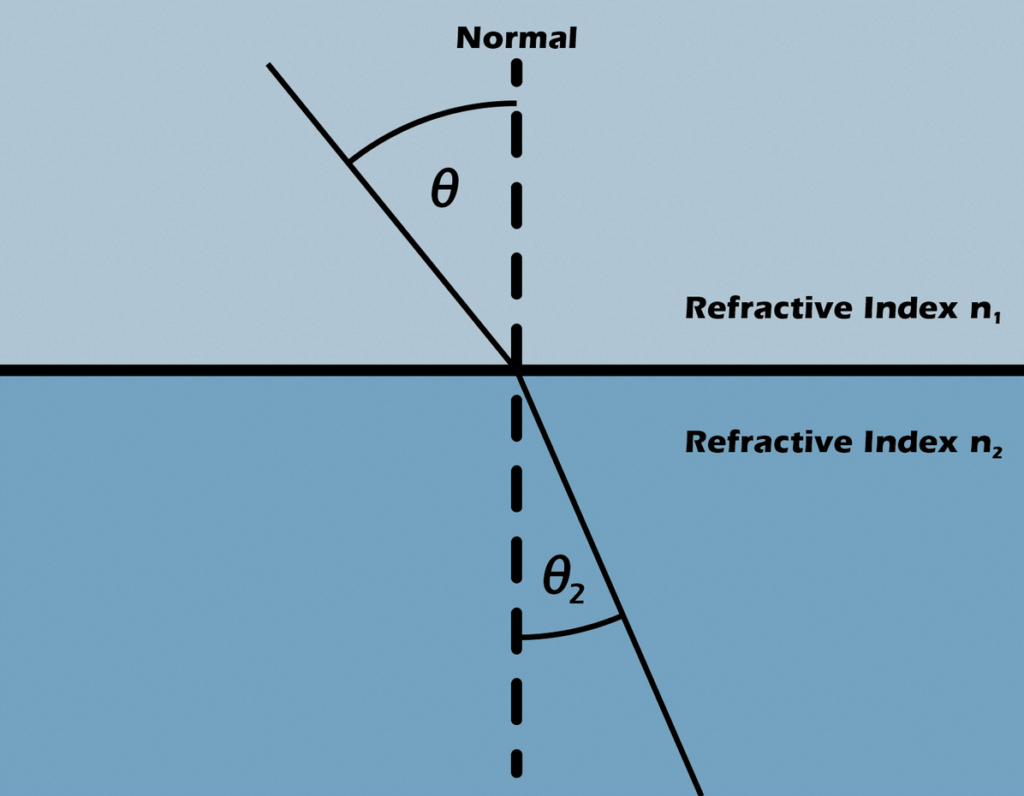
The absolute refractive index n of an optical medium is defined as the ratio of the speed of light in vacuum, c = 299792458 m/s, and the phase velocity v of light in the medium,
n = c/v
Refractometry, also known as Refractive Index Measurement, is the process of gauging the refractive index of substances to evaluate their composition or purity. This analytical method quantifies the alteration in the path of light as it traverses a specific material. Snell’s Law, commonly referred to as the Law of Refraction, elucidates the mathematical principles governing this phenomenon.
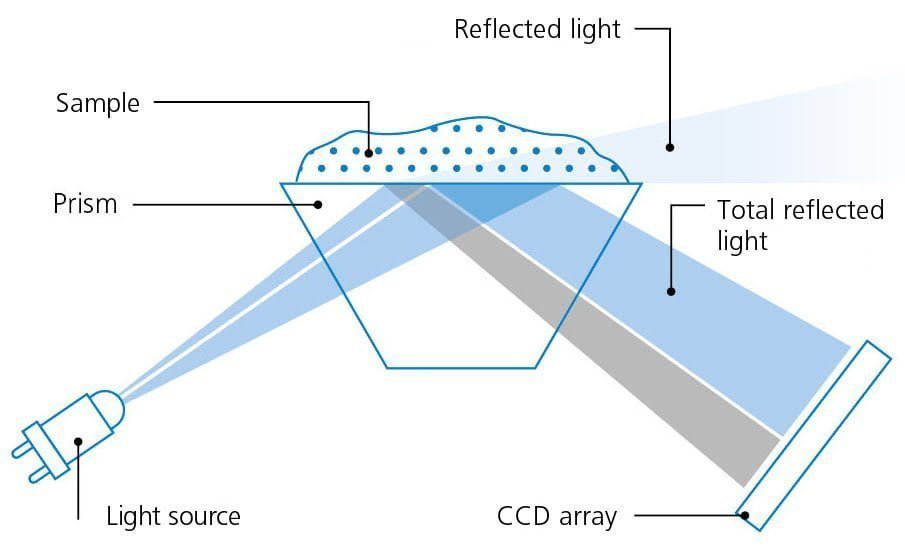
Conventional refractometer gauge the degree of light bending (as a component of the refractive index) in transparent materials, whether in a liquid or solid state. This measurement is subsequently employed to recognize a liquid sample, assess its purity, and ascertain the quantity or proportion of dissolved components within the sample. When light traverses the liquid from the air, it undergoes a reduction in speed, giving rise to an apparent optical deviation known as ‘bending.’ The extent of this bending effect correlates with the concentration of substances dissolved in the liquid. For instance, it is proportional to the quantity of sugar in a glass of water.
Since the late nineteenth century refractometry was one of the main techniques used in chemical analysis. Refractometry was used to determine concentrations of solutions and as an aid in the identification of unknown substances.
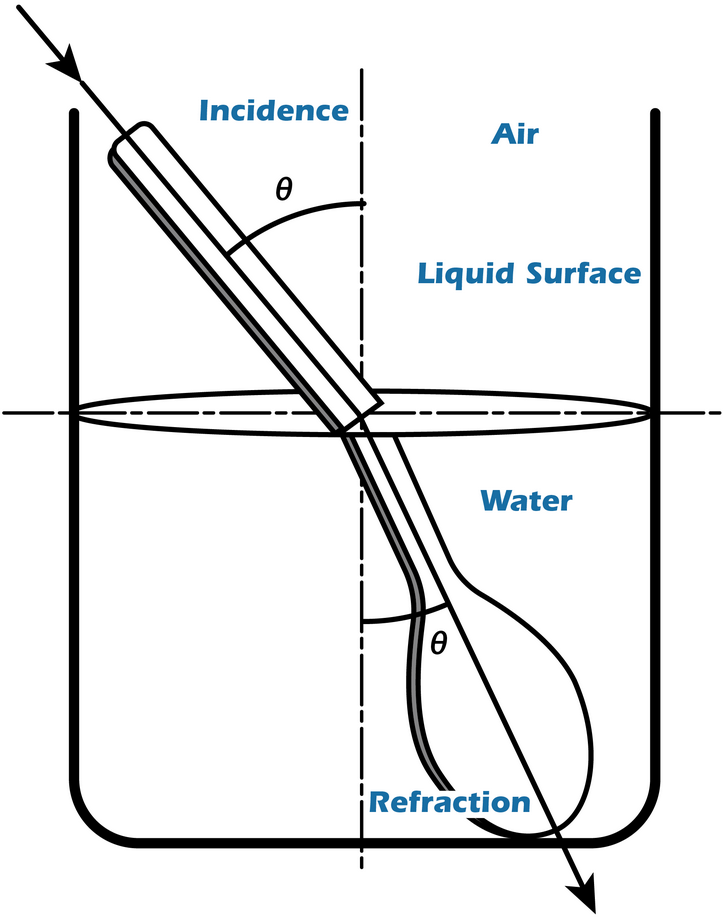
The speed of light in a vacuum is always the same, but when light moves through any other medium it travels more slowly since it is constantly being absorbed and re-emitted by the atoms in the material. The refractive index is a quantity which is a constant for a pure substance under standard conditions of temperature and pressure.
Experiment
In an experiment Atago Pocket refractometer is used to determine P2P (phenylacetone) refraction index. Then, analysis result is compared with literature data.

- The refractometer is calibrated with distilled water.
- The analyzed P2P is placed into the refractometer.
- The P2P refraction index is 1.498, instead of 1.5155-1.5175 for a pure sample.
According to Wikipedia, the pure P2P refraction index is 1.507. It means that this P2P solution isn’t pure enough to correspond literature data for 100% pure standart sample.
Conclusion
Through the presented experiment, we have witnessed the real-world impact of refractometry in identifying even the subtlest impurities. As a tool that has endured for over a century, refractometry continues to be an indispensable asset in modern scientific exploration and quality control, shaping our comprehension of materials and their characteristics.
Bibliography
- Ioffe, Boris Veniaminovich. “Refractometry as a method for the physicochemical analysis of organic systems.” Russian Chemical Reviews 29.2 (1960): 53. https://iopscience.iop.org/article/10.1070/RC1960v029n02ABEH001218/meta
- https://atagousa.corecommerce.com/PAL-RI-p346.html
- https://en.wikipedia.org
