- The process of creating a drug - May 23, 2023
- How do scientists model mental disorders in animals? - May 23, 2023
Introduction
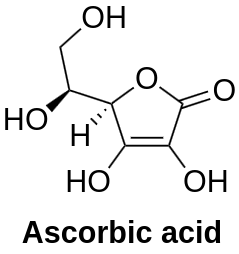
Vitamin C, also known as ascorbic acid, is an essential water-soluble nutrient that holds significant importance in maintaining human health. Its diverse properties make it a crucial component for various physiological processes. In this article, we will delve into the world of Vitamin C, exploring its general information, physico-chemical properties, synthesis, pharmacology, and wide-ranging applications.

General information about Vitamin C
Vitamin C, scientifically known as ascorbic acid, is a water-soluble vitamin that plays a fundamental role in maintaining optimal health. It is an essential nutrient, meaning the human body cannot synthesize it, and therefore, it must be obtained through the diet. Rich dietary sources of Vitamin C include citrus fruits like oranges, lemons, and grapefruits, as well as strawberries, kiwi, guava, bell peppers, broccoli, and leafy greens. The recommended daily intake of Vitamin C varies depending on age, gender, and specific health needs, but generally, it ranges from 65 to 90 milligrams for most adults.
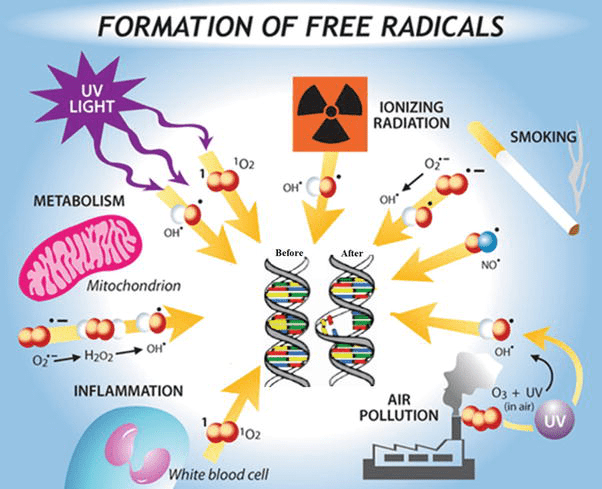
One of the most well-known functions of Vitamin C is its potent antioxidant capacity. As an antioxidant, it helps neutralize harmful free radicals that can damage cells and DNA, protecting the body from oxidative stress and its associated health risks. Moreover, Vitamin C is a vital co-factor in the synthesis of collagen, a protein that forms the structural framework for skin, bones, tendons, and other connective tissues. This makes Vitamin C essential for wound healing, tissue repair, and maintaining healthy skin elasticity.
Another significant role of Vitamin C is its ability to enhance the absorption of non-heme iron, the type of iron found in plant-based foods such as grains, legumes, and vegetables. By facilitating iron uptake in the gut, Vitamin C helps prevent iron deficiency anemia, a common nutritional disorder. Additionally, Vitamin C supports the immune system, assisting in the production and function of white blood cells that defend the body against infections and illnesses.
Vitamin C is water-soluble, which means the body does not store excess amounts. Instead, it is readily excreted through urine, making regular dietary intake essential to maintain adequate levels. Cooking and storage methods can lead to Vitamin C degradation, as it is sensitive to heat, light, and oxygen. Therefore, consuming fresh fruits and vegetables or storing them properly to minimize nutrient loss is crucial to ensure optimal Vitamin C intake.
Overall, Vitamin C’s diverse roles in promoting antioxidant protection, collagen synthesis, immune support, and iron absorption highlight its significance for overall health and well-being. By incorporating Vitamin C-rich foods into the diet or through dietary supplements when necessary, individuals can harness the numerous benefits this essential nutrient offers for maintaining a healthy and vibrant life.
Physico-chemical Properties of Vitamin C
Vitamin C, or ascorbic acid, possesses several notable physico-chemical properties that influence its behavior and functionality. At the molecular level, it is a six-carbon compound with the chemical formula C6H8O6, originally called hexuronic acid. This water-soluble vitamin forms white, but impure samples can appear yellowish, odorless, and acidic crystalline powder lending it the characteristic sour taste found in Vitamin C-rich fruits. Its solubility in water allows for efficient absorption in the gastrointestinal tract, contributing to its bioavailability for various physiological processes.
Ascorbic acid exists as two enantiomers (mirror-image isomers), commonly denoted “l” (for “levo”) and “d” (for “dextro”). The l isomer is the one most often encountered: it occurs naturally in many foods, and is one form (“vitamer”) of vitamin C, an essential nutrient for humans and many animals. Deficiency of vitamin C causes scurvy, formerly a major disease of sailors in long sea voyages. The “d” form can be made via chemical synthesis but has no significant biological role.
Furthermore, Vitamin C’s chemical structure makes it a reducing agent, meaning it has the ability to donate electrons in chemical reactions. The stability of Vitamin C is a crucial consideration in various applications, from food processing to pharmaceutical formulations. The presence of metal ions, such as copper and iron, can catalyze its oxidation and lead to the formation of degradation products. To maintain its potency, Vitamin C is often used in combination with antioxidants or stored in a controlled environment to minimize oxidative reactions.

- Molar mass: 176.124 g/mol
- Melting point: 190-192°C
- Other names: l-ascorbic acid, ascorbic acid, ascorbate, Ascor, Cevalin, Allercorb, Antiscorbic vitamin, Antiscorbutic vitamin, Ascorbajen
- CAS Number: 50-81-7
Acidity
Ascorbic acid is a lactone derived from 2-ketogluconic acid, with a furan-based structure. It possesses an enediol group next to the carbonyl. This particular structural arrangement −C(OH)=C(OH)−C(=O)− is a defining feature of reductones, which enhances the acidity of one of the enol hydroxyl groups. The deprotonated form, known as the ascorbate anion, is stabilized by electron delocalization, brought about by resonance between two distinct forms:

Due to this factor, ascorbic acid exhibits significantly higher acidity compared to what one would anticipate if the compound solely comprised isolated hydroxyl groups.
Salts
The ascorbate anion can create various salts, including sodium ascorbate, calcium ascorbate, and potassium ascorbate.
Esters
Ascorbic acid has the ability to undergo reactions with organic acids in an alcohol-like manner, leading to the formation of esters like ascorbyl palmitate and ascorbyl stearate.
Vitamin C Synthesis
In organisms that can synthesize Vitamin C, the process primarily occurs in the liver. It starts with the conversion of glucose, a simple sugar, into sorbitol by an enzyme called aldose reductase. Sorbitol is then converted to gulonolactone, another sugar derivative, by the enzyme sorbitol dehydrogenase. The final step involves the transformation of gulonolactone into ascorbic acid through the enzyme gulonolactone oxidase.
The loss of the ability to synthesize Vitamin C in some animals and primates is thought to be a consequence of evolutionary changes. While it remains unclear why this occurred, it is believed that during evolution, these species may have acquired other means of protection against oxidative stress, reducing the necessity for Vitamin C synthesis.

Industrial Synthesis
Ascorbic acid is industrially prepared from glucose using a method based on the historical Reichstein process. In the initial step of this five-step process, glucose undergoes catalytic hydrogenation to yield sorbitol, which is further oxidized by the microorganism Acetobacter suboxydans to form sorbose. During this enzymatic reaction, only one of the six hydroxy groups undergoes oxidation. From this point, two routes are available for further synthesis.
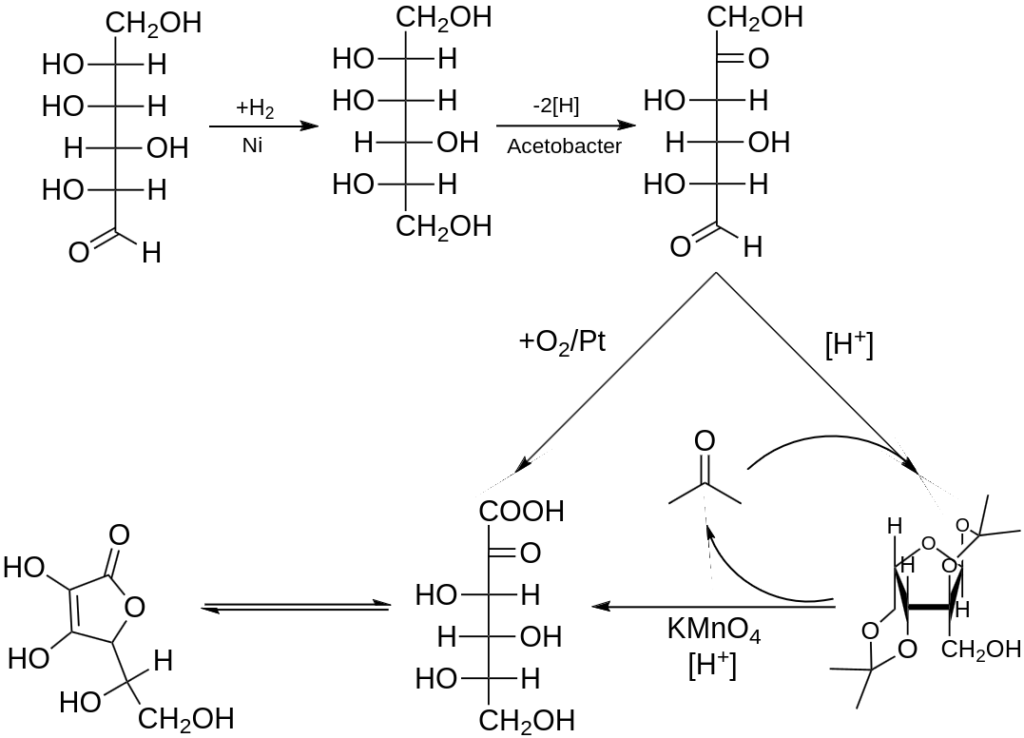
One route involves treating the product with acetone in the presence of an acid catalyst, leading to the conversion of four of the remaining hydroxyl groups into acetals. The unprotected hydroxyl group is then oxidized to the carboxylic acid by reacting with the catalytic oxidant TEMPO (regenerated by sodium hypochlorite, i.e., bleaching solution). In the past, the industrial preparation via the Reichstein process utilized potassium permanganate as the bleaching solution. The next step involves acid-catalyzed hydrolysis of this product, which removes the two acetal groups and induces ring-closing lactonization, ultimately yielding ascorbic acid. Each of these five steps exhibits a yield greater than 90%.
Alternatively, a more biotechnological process was first developed in China during the 1960s and further improved in the 1990s, eliminating the use of acetone-protecting groups. In this approach, a second genetically modified microbe species, such as mutant Erwinia, oxidizes sorbose into 2-ketogluconic acid (2-KGA), which then undergoes ring-closing lactonization via dehydration. This method represents the predominant process employed by the ascorbic acid industry in China, which supplies 80% of the world’s ascorbic acid. American and Chinese researchers are competing to engineer a mutant capable of carrying out a one-pot fermentation directly from glucose to 2-KGA, eliminating the need for a second fermentation and the reduction of glucose to sorbitol.
Furthermore, there exists d-ascorbic acid, which is not found in nature but can be artificially synthesized. Notably, l-ascorbate is known to participate in numerous specific enzyme reactions that require the correct enantiomer (l-ascorbate, not d-ascorbate). l-Ascorbic acid possesses a specific rotation of [α] = +23°.
Pharmacology of Vitamin C
Ascorbate, a variant of Vitamin C, plays a multitude of vital roles within the human body, acting as both an enzyme substrate and an electron donor. Its functions encompass various physiological processes, such as the synthesis of collagen, carnitine, and neurotransmitters, as well as the metabolism of microsomes. Ascorbate functions as a reducing agent during biosynthesis, aiding in the donation of electrons and preventing oxidation to maintain iron and copper atoms in their reduced forms.
The essential functions of Vitamin C as a cofactor are as follows:
- It serves as a cofactor for three groups of enzymes (prolyl-3-hydroxylases, prolyl-4-hydroxylases, and lysyl hydroxylases) involved in the hydroxylation of proline and lysine during collagen synthesis. These enzymatic reactions introduce hydroxyl groups to proline or lysine amino acids in the collagen molecule, facilitated by prolyl hydroxylase and lysyl hydroxylase, both of which require Vitamin C. Vitamin C’s role as a cofactor is to oxidize prolyl hydroxylase and lysyl hydroxylase from Fe2+ to Fe3+ and then reduce them back to Fe2+. This hydroxylation process is crucial for the collagen molecule to adopt its triple helix structure, making Vitamin C indispensable for the development and maintenance of scar tissue, blood vessels, and cartilage.
- Two enzymes (ε-N-trimethyl-L-lysine hydroxylase and γ-butyrobetaine hydroxylase) involved in carnitine synthesis also require Vitamin C as a cofactor. Carnitine plays a vital role in transporting fatty acids into mitochondria for ATP generation.
- Vitamin C acts as a cofactor for Hypoxia-inducible factor-proline dioxygenase enzymes (specifically isoforms EGLN1, EGLN2, and EGLN3).
- It is involved in the biosynthesis of norepinephrine from dopamine, participating as a cofactor for dopamine beta-hydroxylase.
- Peptidylglycine alpha-amidating monooxygenase relies on Vitamin C as a cofactor to amidate peptide hormones, removing the glyoxylate residue from their c-terminal glycine residues. This modification enhances peptide hormone stability and activity.
According to the U.S. National Institutes of Health, in the human body, around 70% to 90% of vitamin C gets assimilated with moderate daily intakes ranging from 30 to 180 mg. Nevertheless, when the dosage exceeds 1,000 mg per day, the absorption rate declines to less than 50%. The transportation of vitamin C through the intestines occurs via mechanisms that can be influenced by the presence of glucose. Both glucose-sensitive and glucose-insensitive mechanisms are involved, and the presence of significant amounts of sugar in the intestine can lead to a slowdown in absorption.
Applications of Vitamin C
Vitamin C plays a crucial role in the treatment of scurvy, a condition arising from its deficiency. However, its potential role in preventing or treating other diseases remains controversial, with conflicting findings reported in various reviews. In a 2012 Cochrane review, no significant impact of vitamin C supplementation on overall mortality was observed. Despite this, Vitamin C is listed on the World Health Organization’s List of Essential Medicines.
Scurvy
Scurvy, a disease caused by insufficient vitamin C, can be prevented and treated by incorporating vitamin C-rich foods or dietary supplements. Symptoms usually emerge after at least a month of vitamin C deficiency. Early signs include lethargy and malaise, progressing to bone pain, bleeding gums, easy bruising, poor wound healing, and eventually fever, convulsions, and death. The damage caused by scurvy can be reversed until the disease reaches an advanced stage. With vitamin C replenishment, healthy collagen replaces the faulty collagen. Treatment can involve oral vitamin supplementation or intramuscular/intravenous injections. The knowledge of scurvy dates back to Hippocrates in classical times. Royal Navy surgeon James Lind demonstrated the preventive properties of citrus fruits in an early controlled trial aboard HMS Salisbury in 1747. From 1796 onwards, lemon juice was issued to all Royal Navy crew members.
Common cold
Research on the effects of vitamin C on the common cold has focused on prevention, duration, and severity. A Cochrane review examining doses of at least 200 mg/day found no significant preventive effect of regular vitamin C intake on the common cold. Similarly, analyzing trials with at least 1000 mg/day showed no preventive benefit. However, regular vitamin C intake did reduce the average duration of colds by 8% in adults and 14% in children, as well as mitigated the severity of symptoms. For certain groups, like marathon runners, skiers, or soldiers in subarctic conditions, supplementation halved the incidence of colds. Therapeutic use, starting vitamin C upon cold symptom onset, did not affect duration or severity. A previous review stated that vitamin C did not prevent colds, reduced duration but not severity. The Cochrane review authors concluded that while routine vitamin C supplementation is not justified to reduce the incidence of colds in the general population, considering its consistent effects on duration and severity in regular supplementation studies, its low cost and safety, it might be worth considering individualized therapeutic vitamin C supplementation for common cold patients.
Immune system regulation
Vitamin C is quickly absorbed into immune cells in substantial amounts and exhibits antimicrobial properties, enhances natural killer cell activities, and supports lymphocyte proliferation. These effects suggest a significant role in regulating the immune system. The European Food Safety Authority has recognized a cause-and-effect relationship between dietary vitamin C intake and the normal functioning of the immune system in adults and children under three years of age.
Numerous studies have identified specific antiviral properties of Vitamin C, wherein it disrupts the RNA or DNA of viruses or interferes with virus assembly.
COVID-19
According to information from ClinicalTrials.gov in early 2021, there were 50 ongoing or completed clinical trials investigating the use of vitamin C as a treatment for COVID-19. In October 2021, a meta-analysis of six published trials was conducted. The treatments involved both oral and intravenous administration, with doses ranging from 50 mg/kg/day to 24 g/day. The reported outcomes included mortality rates, duration of hospitalization, intensive care stay, and the need for ventilation. The conclusion of the meta-analysis stated that the administration of vitamin C did not yield any significant effects on major health outcomes in COVID-infected patients, in comparison to placebo or standard therapy. Further sub-group analysis also demonstrated that regardless of dosage, method of administration, or disease severity, vitamin C did not provide any apparent benefits to these patients. As a result, larger prospective randomized trials are required to thoroughly assess the effects of isolated vitamin C administration, both for individuals with adequate vitamin C levels and those who are deficient.
Cardiovascular disease
A study conducted in 2017, involving 15,445 participants, independently examined the potential impact of vitamin C on reducing the risk of cardiovascular disease. The results of this study did not provide any evidence to support the notion that vitamin C decreases the risk of cardiovascular disease. These findings align with a review from 2013 that also found no evidence indicating that antioxidant vitamin supplementation reduces the risk of myocardial infarction, stroke, cardiovascular mortality, or all-cause mortality (however, this review did not offer a subset analysis for trials specifically using vitamin C).
Contrastingly, another review from 2013 revealed a connection between higher levels of circulating vitamin C or dietary vitamin C intake and a reduced risk of stroke.
Additionally, a review from 2014 highlighted a positive effect of vitamin C on endothelial dysfunction when taken at doses exceeding 500 mg per day. The endothelium refers to a layer of cells that line the interior surface of blood vessels.
Iron deficiency
Reduced iron absorption is one of the contributing factors to iron-deficiency anemia. Vitamin C can enhance iron absorption when consumed alongside iron-containing food or supplements. However, the instability of vitamin C during cooking and storage restricts the number of foods suitable for this purpose. By keeping iron in the reduced ferrous state, vitamin C aids in its solubility and facilitates easier absorption.
Conclusion
Vitamin C stands as a remarkable nutrient with diverse properties that impact our health and well-being. Its chemistry, pharmacology, and applications demonstrate the wide-ranging benefits it offers. From collagen synthesis and immune support to its antioxidant prowess, Vitamin C remains a valuable asset in our quest for better health and longevity. Harnessing the power of this essential nutrient opens doors to innovative applications and furthering our knowledge of human biology. As we continue to explore the depths of Vitamin C’s potential, we are reminded of the profound impact such simple molecules can have on our lives.
Bibliography
- Food and Nutrition Board, Institute of Medicine. “Dietary reference intakes for vitamin C, vitamin E, selenium, and carotenoids.” National Academy Press, Washington, DC (2000). https://nap.nationalacademies.org/catalog/9810/dietary-reference-intakes-for-vitamin-c-vitamin-e-selenium-and-carotenoids
- Higdon, Jane, et al. “Linus Pauling Institute Micronutrient Information Center.” Oregon State University (2003–2018) (2000). https://lpi.oregonstate.edu/mic/vitamins/vitamin-C
- Hemilä, Harri, and Elizabeth Chalker. “Vitamin C for preventing and treating the common cold.” Cochrane database of systematic reviews 1 (2013). https://www.cochranelibrary.com/cdsr/doi/10.1002/14651858.CD000980.pub4/abstract
- Ye Y, Li J, Yuan Z (2013). “Effect of antioxidant vitamin supplementation on cardiovascular outcomes: a meta-analysis of randomized controlled trials” https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0056803
- Michels A, Frei B (2012). “Vitamin C”. In Caudill MA, Rogers M (eds.). Biochemical, Physiological, and Molecular Aspects of Human Nutrition (3 ed.). Philadelphia: Saunders. pp. 627–654. https://www.sciencepublishinggroup.com/journal/paperinfo?journalid=223&doi=10.11648/j.sjac.20200802.16
- https://en.wikipedia.org/wiki/Vitamin_C