- The process of creating a drug - May 23, 2023
- How do scientists model mental disorders in animals? - May 23, 2023
Introduction to Lysergic Acid
Lysergic acid CAS 82-58-6 is a naturally occurring compound that belongs to the ergoline family of alkaloids. It serves as a fundamental building block and precursor for the synthesis of various lysergamides, including the well-known and highly potent psychedelic substance known as LSD (lysergic acid diethylamide). Discovered and isolated by Swiss chemist Albert Hofmann in 1938, lysergic acid has since garnered significant attention and intrigue in the fields of chemistry, pharmacology, and psychopharmacology.
Lysergic acid derives its name from the fungus Claviceps purpurea, commonly known as ergot, which grows on various grains, particularly rye. This fungus naturally produces a group of alkaloids called ergot alkaloids, and lysergic acid is one of them. Lysergic acid acts as a central component in the biosynthesis of these ergot alkaloids, which possess a range of biological activities.
While lysergic acid itself does not exhibit significant psychoactive effects, its derivative LSD has gained notoriety for its powerful hallucinogenic properties.
Chemical and Physical Properties of Lysergic Acid
Lysergic acid CAS 82-58-6, with its unique chemical structure, possesses distinct chemical and physical properties that contribute to its intriguing characteristics. It is a crystalline solid that appears as colorless leaf shaped crystals. The molecular formula of lysergic acid is C16H16N2O2, with a molecular weight 268.32 g/mole.
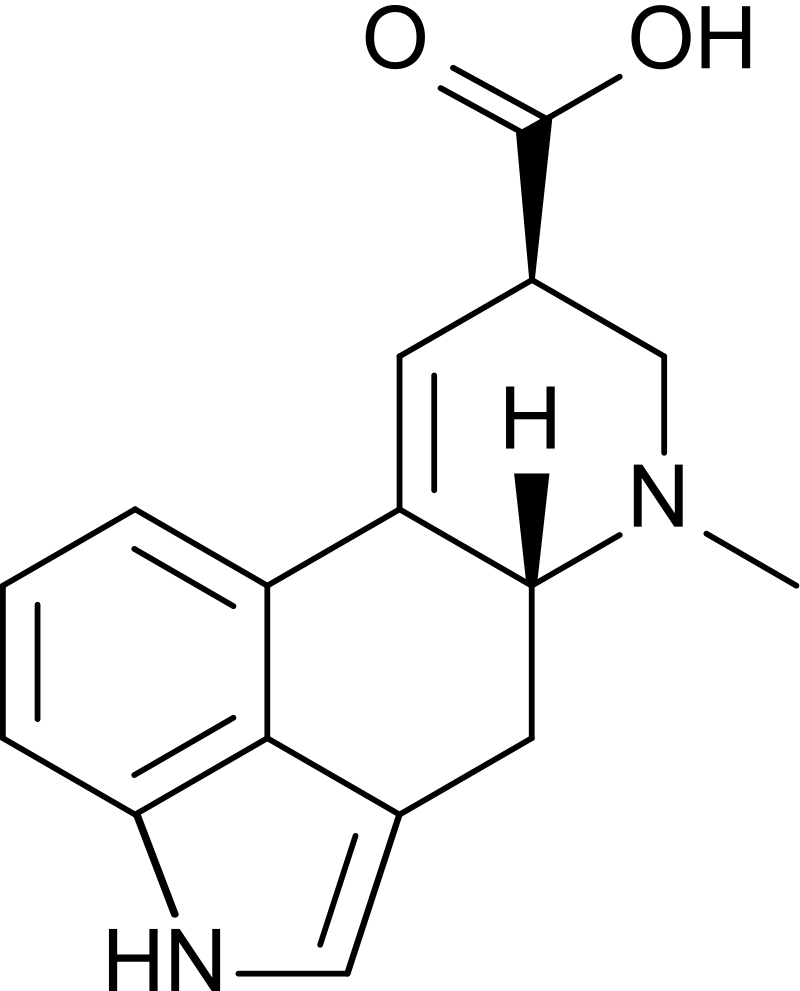
Lysergic acid is sparingly soluble in water, meaning it dissolves only to a limited extent; slightly soluble in benzene, ether. However, it exhibits good solubility in organic solvents such as ethanol, methanol, chloroform, pyridine and dimethyl sulfoxide (DMSO). These solvents are commonly used for the extraction and purification of lysergic acid.
The compound has a melting point ranging from 238 to 240 °C (460 to 464 °F), indicating its solid state at room temperature. However, it is crucial to note that lysergic acid is sensitive to light, heat, and air. Easily oxidized during storage. Exposure to these factors can lead to degradation. Therefore, proper storage and handling in a cool, dark, and airtight environment are necessary to maintain its stability and integrity.
Uses of Lysergic Acid
Lysergic acid, despite lacking significant psychoactive effects itself, plays a crucial role in various applications and research endeavors. Its primary use lies in serving as a precursor for the synthesis of LSD (lysergic acid diethylamide), a powerful psychedelic substance. However, lysergic acid also finds application in other areas, both within and beyond the realm of recreational drug use.
Synthesis of LSD
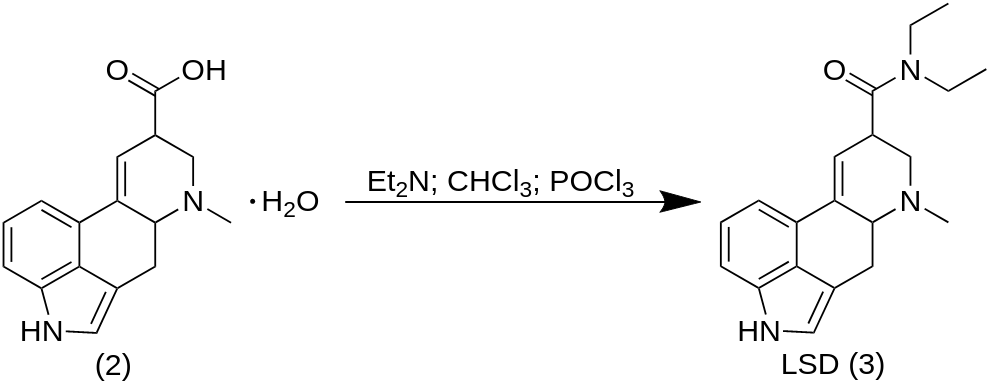
Lysergic acid serves as the key starting material in the synthesis of LSD. Through a chemical processes, lysergic acid is turned into LSD. LSD, known for its profound hallucinogenic effects, has been the subject of extensive research and exploration, particularly in the realms of neuroscience, psychology, and consciousness studies.
Research and Psychopharmacology
Lysergic acid and its derivatives, including LSD, have garnered significant attention in the scientific community. Researchers have conducted studies to better understand the pharmacological effects, mechanisms of action, and potential therapeutic applications of these compounds. Studies involving lysergic acid and LSD have explored areas such as consciousness alteration, psychotherapy, creativity enhancement, and the treatment of psychiatric disorders, including depression, anxiety, and addiction.
Drug Discovery
Lysergic acid and its analogs have been investigated as potential starting points for the development of new medications. Researchers have explored their interactions with various receptors in the brain, aiming to harness their therapeutic potential. These efforts have yielded insights into the serotonin system and have contributed to advancements in drug discovery and the understanding of receptor binding.
Analytical Reference Standard
Lysergic acid is also utilized as an analytical reference standard in laboratory settings. It serves as a benchmark compound for identifying and quantifying lysergic acid derivatives, including LSD, in analytical techniques such as gas chromatography-mass spectrometry (GC-MS) or liquid chromatography-mass spectrometry (LC-MS). The availability of lysergic acid as a reference standard facilitates accurate and reliable analysis in forensic and research applications.
While the recreational use of lysergic acid and LSD remains controversial and subject to legal restrictions in many jurisdictions, scientific interest in their potential benefits continues to grow. Ongoing research seeks to unravel the mechanisms of action, therapeutic potential, and possible risks associated with lysergic acid and its derivatives.
Manufacturing of Lysergic Acid
The manufacturing process of lysergic acid involves several steps, starting from the natural source of the compound, the ergot fungus (Claviceps purpurea). This fungus commonly grows on various grains, particularly rye, and contains ergot alkaloids, including lysergic acid, as natural components.
Cultivation and Harvesting: The first step in manufacturing lysergic acid involves the cultivation and harvesting of ergot fungus. Cultivation typically takes place in controlled environments, where the fungus is grown on a substrate such as sterilized rye grain. This process ensures the optimal growth and development of ergot, allowing for a higher yield of lysergic acid.

Extraction: Once the ergot fungus has matured, it is harvested and subjected to extraction processes to isolate the ergot alkaloids, including lysergic acid. Extraction techniques often involve the use of solvents such as ethanol, methanol, or chloroform. These solvents help dissolve and extract the desired alkaloids from the fungal material, forming a crude extract.
Purification and Isolation: Following extraction, the crude extract undergoes purification steps to isolate lysergic acid. The purification process typically involves various separation techniques such as filtration, chromatography, and crystallization. These methods help separate lysergic acid from other impurities present in the crude extract, resulting in a purified form of the compound.
Chemical Modification: Once lysergic acid has been isolated, it can undergo chemical modifications for specific applications. The most notable application is the synthesis of LSD (lysergic acid diethylamide). Through a chemical process called an amidation reaction, lysergic acid reacts with diethylamine, resulting in the formation of LSD. This reaction typically takes place under controlled laboratory conditions and requires expertise in organic chemistry.
Formulation and Packaging: Depending on the intended use, lysergic acid can be further processed and formulated into various forms such as tablets, capsules, or liquid solutions. These formulations allow for convenient administration and dosing in research or medical settings.
Chemical syntheis
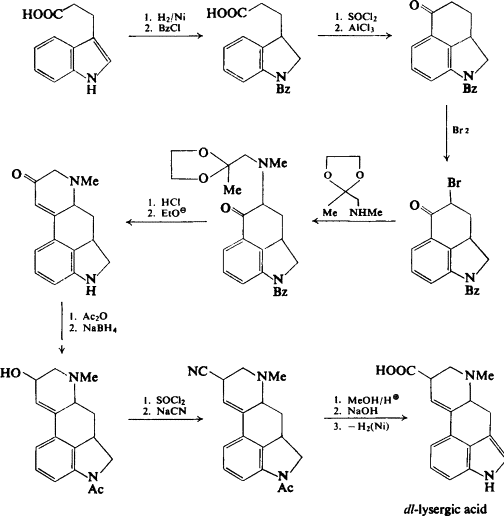
Lysergic acid can be obtained by the hydrolysis of organic lysergamides, although it can be synthetically produced in a lab using a detailed total synthesis, for instance, by using a method developed by Robert Burns Woodward’s group in 1956. In 2011, Fujii and Ohno proposed an enantioselective total synthesis dependent on a domino cyclization reaction catalyzed by palladium. When recrystallized from water, lysergic acid monohydrate crystallizes as very thin hexagonal plates. When dried at 140 °C and a pressure of 2 mmHg (or 270 Pa), lysergic acid is transformed into anhydrous lysergic acid. The manufacturing procedure begins by alkylating the amino acid tryptophan with dimethylallyl diphosphate (which is derived from 3R-mevalonic acid) to form 4-dimethylallyl-L-tryptophan, which is subsequently N-methylated employing S-adenosyl-L-methionine. After oxidative ring closure, decarboxylation, reduction, cyclization, oxidation, and allylic isomerization, D-(+)-lysergic acid is produced.
Lysergic Acid Legal Status
Lysergic acid and its derivatives, including LSD, have a complex and highly regulated legal status in many countries worldwide. Due to the psychoactive properties and potential for misuse, lysergic acid and LSD are classified as controlled substances in numerous jurisdictions. The production, possession, distribution, and use of these substances outside of authorized research or medical contexts are generally illegal. The legal status of lysergic acid varies from country to country, with some nations strictly prohibiting all forms of its use, while others may allow limited research or therapeutic applications under strict regulations. It is essential to be aware of and comply with the legal requirements and restrictions regarding lysergic acid in each respective jurisdiction to avoid legal consequences. Additionally, regulations surrounding lysergic acid are subject to change, so it is crucial to stay updated on current legislation and regulations related to these substances.
Conclusion
In conclusion, the manufacturing process of lysergic acid involves several key steps, starting from the cultivation and harvesting of the ergot fungus to the extraction, purification, and potential chemical modifications for specific applications. The process requires expertise, adherence to safety protocols, and compliance with legal regulations due to the controlled nature of lysergic acid and its derivatives. Insight the intricacies of lysergic acid manufacturing provides valuable data of the complexity and precision required to produce this compound for research, medicinal, or analytical purposes. Continued advancements in manufacturing techniques and ongoing research contribute to a deeper understanding of lysergic acid and its potential applications in various fields.
Bibliography
- Edmund C. Kornfeld, E.J. Fornefeld, G. Bruce Kline, Marjorie J. Mann, Dwight E. Morrison, Reuben G. Jones and R.B. Woodward; Journal of the American Chemical Society Volume 78, pages 3087-3114 (1956) https://pubs.acs.org/doi/abs/10.1021/ja01594a039
- Schiff Jr, Paul L. “Ergot and its alkaloids.” American journal of pharmaceutical education 70.5 (2006). https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1637017/
- Martınková, Ludmila, et al. “Hydrolysis of lysergamide to lysergic acid by Rhodococcus equi A4.” Journal of biotechnology 84.1 (2000): 63-66. https://www.sciencedirect.com/science/article/pii/S0168165600003321
- https://commonchemistry.cas.org/detail?cas_rn=82-58-6
- https://bbgate.com/threads/lsd-synthesis-from-ergotamine.1147/
