Abstract
In the 1970s, Pfizer developed a series of cyclohexylphenols (CPs). One of the CPs, CP 47,497, is classified as a ‘non-classical’ cannabinoid in the scientific literature. CP 47,497 is composed of several different isomers, including (C6)-CP 47,497, (C7)-CP 47,497, and their (+) and (-) isomers, as well as (C8)-CP 47,497 and its four enantiomers, and (C9)-CP 47,497. It is important to note that CP 47,497 is significantly more potent than THC, with studies indicating that it can be up to 28 times more potent.
This article provides a comprehensive overview of CP 47,497, covering its general information, physico-chemical properties, pharmacology in recreational use, potential effects and symptoms, street names, prices and approximate dosage, legal status, synthesis of CP 47,497, conclusion, and bibliography.
From Spice to Delta 8 THC with Cannabinoid Expert Dr. Sam Banister
General Information About CP 47497 [1-19]
- Other synonyms names of CP 47497 are: Phenol, 5-(1,1-dimethylheptyl)-2-[(1S,3R)-3-hydroxycyclohexyl]; 5-(1,1-Dimethyl-heptyl)-2-(3-hydroxy-cyclohexyl)-phenol; 5-(1,1-Dimethylheptyl)-2-[(1R,3S)-3-hydroxycyclohexyl]phenol; `2-((1R,3S)-3-hydroxycyclohexyl)-5-(2-methyloctan-2-yl)phenol; cis-3-(2-Hydroxy-4-(1,1-dimethylheptylphenyl)-cyclohexan-1-ol); cis-5-(1,1-dimethylnonyl)-2-(3-hydroxycyclohexyl)-phenol; (cis)-3-[2-Hydroxy-4-(1,1-dimethyloctyl)phenyl]cyclohexanol; cis-5-(1,1-dimethylhexyl)-2-(3-hydroxycyclohexyl)-phenol
- IUPAC Name of (C6)-CP 47497: 2-[(1S,3R)-3-Hydroxycyclohexyl]-5-(2-methyl-2-heptanyl)phenol
- IUPAC Name of (C7)- (-)-CP 47497: 2-[(1S,3R)-3-Hydroxycyclohexyl]-5-(2-methyl-2-octanyl)phenol
- IUPAC Name of (C7)- (+)-CP 47497: 2-[(1R,3S)-3-Hydroxycyclohexyl]-5-(2-methyl-2-octanyl)phenol
- IUPAC Name of (C8)-CP 47497: 2-[(1R,3S)-3-hydroxycyclohexyl]-5-(2-methylnonan-2-yl)phenol
- CAS numbers for (C6)-CP 47497 are 132296-20-9; 70435-06-2
- CAS numbers for (C7)-(-)-CP 47497 are 70434-82-1; 134308-14-8
- CAS numbers for (C7)-(+)-CP 47497 are 114753-51-4; 70434-82-1
- CAS number for (C8)-CP 47497 is 70434-92-3
- CAS numbers for (C9)-CP 47497 are 70435-08-4; 132296-12-9
- Trade names are CP 47497; (C6)-CP 47,497; (C7)-CP 47,497; (C8)-CP 47,497; (C9)-CP 47,497; FAPQAUKC04; CP 47,497-C6-homolog; CP 47,497-C7-homolog; CP 47,497-C8-homolog; CP 47,497-C9-homolog; CCH; Cannabicyclohexanol; CP 47,497 dimethylhexyl homologue; CP 47,497 dimethyloctyl homologue; CP 47,497 dimethylnonyl homologue; Cis-CP 47497C8
Physico-Chemical Properties of CP 47497 [1-19]
Molecular Formulas:
- (C6)-CP 47497 C20H32O2
- (C7)-CP 47497 C21H34O2
- (C8)-CP 47497 C22H36O2
- (C9)-CP 47497 C23H38O2
Molar Weights
- 304.474 g/mol (C6)-CP 47497
- 318.493 g/mol (C7)-CP 47497
- 332.528 g/mol (C8)-CP 47497
- 346.555 g/mol (C9)-CP 47497
- 407.3±45.0 °C at 760 mmHg (C6)-CP 47497
- 421.2 ºC at 760 mmHg (C7)-CP 47497
- 434.8±45.0 °C at 760 mmHg (C8)-CP 47497
- 448.2±45.0 °C at 760 mmHg (C9)-CP 47497
- Melting Point of (C7)-(-)-CP 47497 161.80 ºC
- Melting Point of (C7)-(+)-CP 47497 161.80 ºC
- Solubility of all CP 47497: DMF: 10 mg/ml; DMSO: 15 mg/ml; Ethanol: 10 mg/ml; Methanol: 12.5 mg/ml; not in water
- Color/Form: A crystalline solid
- Odor: Characteristic
- Bio activity of (C7)-(-)-CP 47497: Potent CB1 agonist
- Bio activity of (C7)-(+)-CP 47497: Potent CB2 agonist
Structural formula of (C6)-CP 47497 is present on Figure 1.
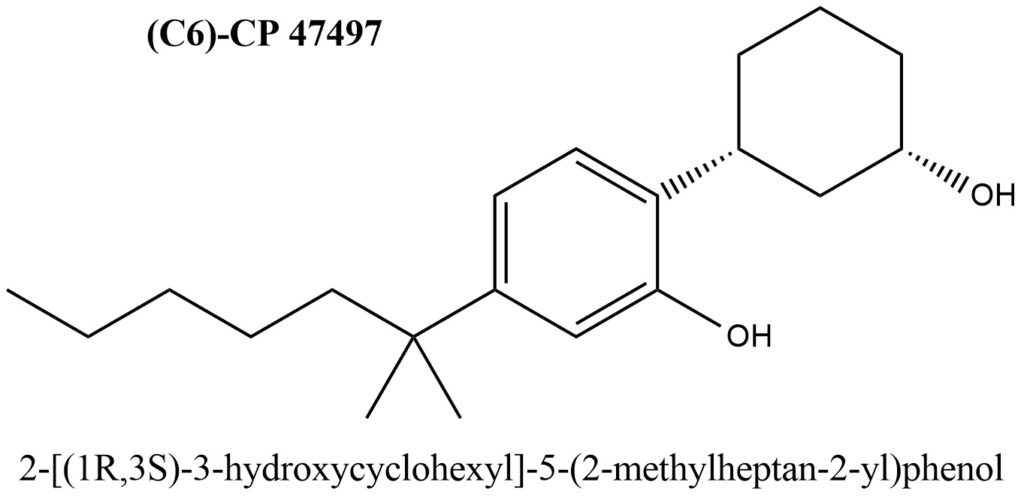
Figure 1. Structure of (C6)-CP 47497
Structural formulas of (C7)-(-)-CP 47497 and (C7)-(+)-CP 47497 are present on Figure 2.
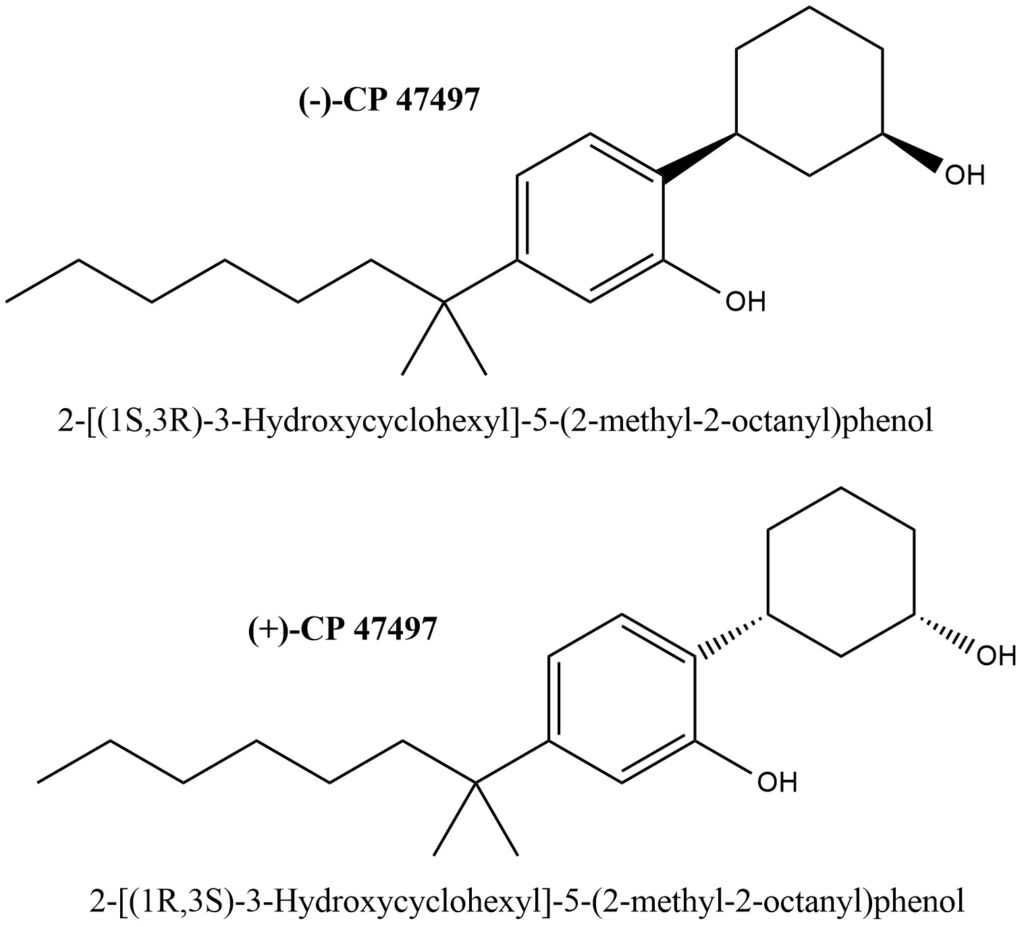
Figure 2. Structures of (C7)- (+)- and (-)-CP 47497
Figure 3 displays the structural formulas for the four enantiomers of cannabicyclohexanol (C8)-CP 47497.
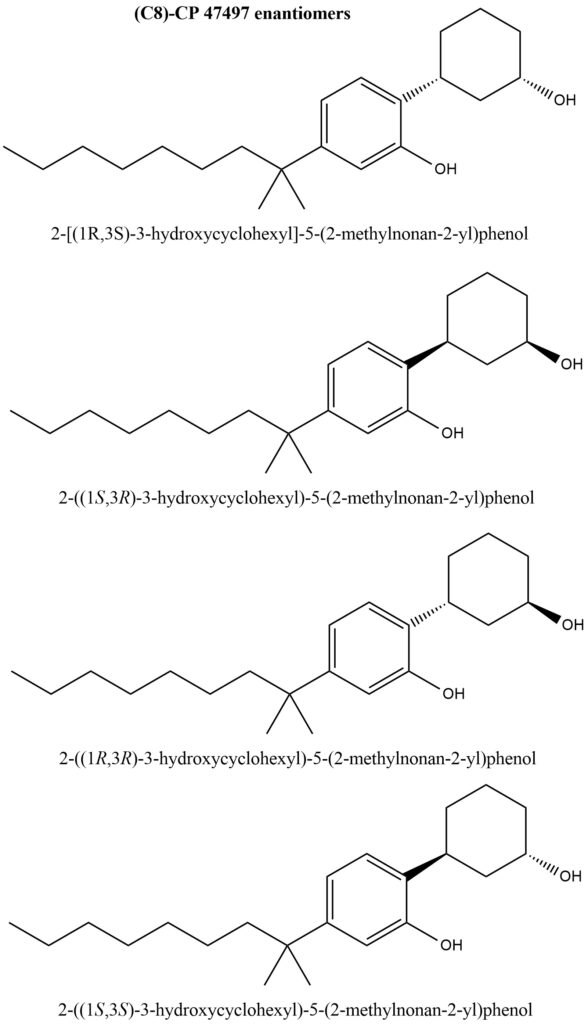
Figure 3. Structures of the four enantiomers of cannabicyclohexanol (C8)-CP 47497
Structural formula of (C9)-CP 47497 is present on Figure 4.
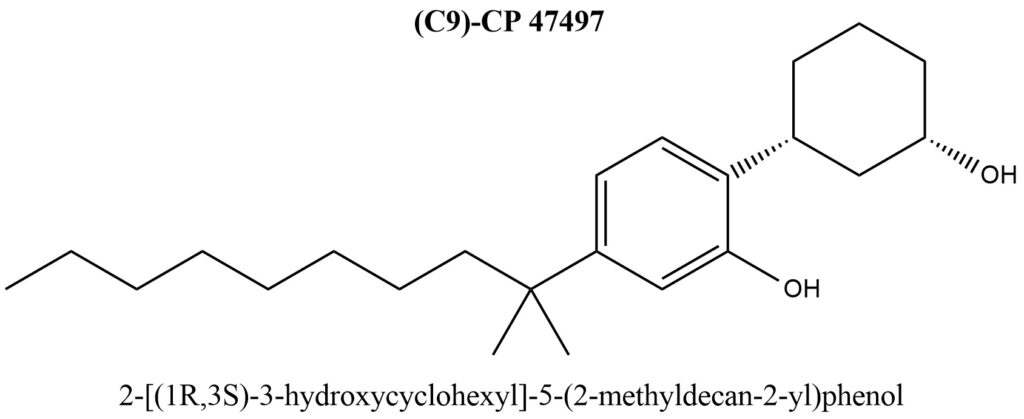
Figure 4. Structure of (C9)-CP 47497
Powder and crystalline solid possible of the CP 47497 can be seen in the picture provided in Figure 5.

Figure 5. Crystalline Powder of CP 47497
General Information of CP 47497 in Recreational Use and Pharmacology [20-25]
CP 47,497 is a synthetic cannabinoid that is 3-28 times more potent than THC. It has a strong affinity for both the central CB1 and peripheral CB2 receptors. The alkyl chain length of C4 to C6 has been found to possess equal or higher affinity to the CB receptors compared to THC. This compound is intended for research and forensic purposes.
A related synthetic cannabinoid, (+)-CP 47947-C7-homolog, binds specifically to the CB2 receptor and has been used as an antidote for CP 47,497 overdoses. It has also been found to have an effect on autoimmune diseases, possibly due to its ability to block the production of proinflammatory cytokines.
(-)-CP-47,497-C7-homolog is a monophenolic synthetic cannabinoid receptor agonist that binds strongly to the CB1 receptor. This binding triggers inhibition of adenylate cyclase and N-type voltage-dependent Ca2+ channels, as well as activation of MAPK. In vitro studies have shown that binding to the CB1 receptor results in activation of the caspase pathway, leading to dose-dependent cytotoxicity in neuroblastoma and glioma cell lines. In vivo studies on animal models have shown depression of locomotory activity, anticonvulsant and hypothermic effects, and strong antinociceptive activity.
Cannabicyclohexanol, also known as (±)-CP 47,497-C8-homolog, is a bicyclic cannabinoid analog that avidly binds to the CB1 receptor and exhibits high antinociceptive activity. It is ten times more potent than THC in its analgesic, motor depressant, anticonvulsant, and hypothermic effects in mice. Cannabicyclohexanol has four enantiomers, which are expected to have varying affinities for cannabinoid receptors and thus show differing levels of potency. This compound shows promise as a potential therapeutic drug, but further research is needed to fully understand its effects and potential applications.
Effects and symptoms of CP 47497 Use
Most psychological disturbances can be managed with simple reassurance, possibly with the addition of lorazepam, diazepam, or midazolam. Sinus tachycardia typically doesn’t require treatment, but if necessary, it can be controlled with beta-blockers. Orthostatic hypotension responds to a head-down position and intravenous fluids. No specific antidote exists for marijuana.
Although gastrointestinal effects, such as nausea, vomiting, and retching, are the most common after consumption of synthetic cannabinoids, cardiovascular effects, such as extremely elevated heart rate and blood pressure, chest pain, and cardiac ischemia, are among the most dangerous consequences. Occasional inappropriate laughter, injected conjunctiva, xerostomia, and nystagmus have also been described. Synthetic cannabinoids also induce metabolic effects, such as hypokalemia, hyperglycemia, and acidosis, and autonomic effects, such as fever and mydriasis.
In general, the desired effects of synthetic cannabinoids include a sense of empathy and well-being. However, there is an increasing number of clinical reports describing patients presenting for emergency medical care after smoking synthetic cannabinoid products, with the most common symptoms being nausea, anxiety, agitation/panic attacks, tachycardia, paranoid ideation, and hallucinations.
Symptoms associated with synthetic marijuana use include an elevated heart rate, elevated blood pressure, palpitations, diaphoresis, tremulousness, anxiety, and agitation. Other physical findings may include mydriasis, although this finding is not consistent with intoxication. Psychosis with auditory and visual hallucinations is mentioned in rare circumstances, most commonly with individuals who have underlying psychiatric disorders. Seizures and arrhythmias have been reported but are rare.
Accidental overdosing with a risk of severe psychiatric complications may be more likely to occur because the type and amount of cannabinoid can vary considerably from batch to batch, even within the same product. In general, there may be a risk of the appearance of a full CB receptor agonist leading to life-threatening conditions if overdosed, unlike THC, which acts only as a partial agonist.
The duration of effects in humans compared to THC seems to be longer for CP 47,497-C8, with effects lasting between 5 and 6 hours.
Street Names, Prices and Approximate Dosage

Various brands of herbal mixtures, such as “Spice”, “Spice Gold”, “Spice Diamond”, “Arctic Spice”, “Silver”, “Aroma”, “K2”, “Genie”, “Scene”, “Dream”, Yucatan Fire, Smoke, Sence, ChillX, Highdi’s, Almdröhner, Earth Impact, Gorillaz, Skunk, Galaxy Gold, Space Truckin, Solar Flare, Moon, Rocks, Blue Lotus, and Scope, are available on the internet. Gold, Diamond, Arctic Synergy, and Tropical Energy are among those that contain CP 47,497 and can be directly ordered online. These herbal mixtures are marketed under the name “Spice” and are mainly sold online and in dedicated stores that offer legal alternatives to banned drugs. They are affordable, typically costing around 9-12€ per gram, and a sachet of 3 g is sufficient for around eight joints and costs 27-36€.
A 20-year-old man who had been smoking “Spice Gold,” which typically contains CP-47,497-C8 and JWH-018, daily for eight months as a means of relief from his internal unrest and nervousness experienced withdrawal phenomena and a dependence syndrome. The man voluntarily entered the hospital and requested detoxification treatment after experiencing a similar syndrome during a phase of abstinence owing to a short supply. Internal unrest and profuse sweating were among the first symptoms observed by doctors, followed by drug craving, nocturnal nightmares, tremor, and headache. Other physical withdrawal symptoms included palpitation, nausea and vomiting, similar to those described during cannabis withdrawal. In addition to a clear withdrawal syndrome, the patient developed drug tolerance, needed to rapidly increase the dose from 1 to 3 g/day, and continued to have a strong desire for the drug, despite adverse consequences, indicating dependency.
Legal Status

These compounds are classified as Schedule I controlled substances in the USA, meaning that they are considered to have a high potential for abuse and no currently accepted medical use in the United States. CP-47,497 and cannabicyclohexanol were initially used in basic scientific research to study structure activity relationships.
According to the regulations set forth by the DEA, Schedule I substances consist of drugs and other substances that are considered to have a high potential for abuse, no accepted medical use in treatment in the United States, and a lack of accepted safety for use under medical supervision. These substances are identified by their official, common, or chemical names, as well as their brand names, and each has been assigned a DEA Controlled Substances Code Number.
Additionally, any material, compound, mixture or preparation that contains any quantity of certain substances, such as 5-(1,1-Dimethyloctyl)-2-[(1R,3S)-3-hydroxycyclohexyl]-phenol, its optical, positional, and geometric isomers, salts and salts of isomers (DEA Code Number: 7298), are also included on this list.
The compounds are classified differently in other countries, such as in the UK where they are categorized as Class B, in Germany where they are authorized for trade only and not prescriptible (Anlage II), and in France, Latvia, Lithuania, Sweden, Romania where they are illegal.
Synthesis of CP 47497 [22, 25, 26]
During the 1970s, Pfizer developed a series of compounds known as cyclohexylphenols (CP). This series included examples such as CP 59,540, CP 47,497, and their n-alkyl homologues. These compounds are often referred to as “non-classical” cannabinoids in the scientific literature.
The phenolic side-chain portion of prototype 1 is prepared from the nitrile 2, 11 which is first alkylated (98% yield), reduced with diisobutylaluminum hydride, and then hydrolyzed to aldehyde 4 (99%). The crude aldehyde 4 is reacted via the Wittig condensation to yield olefin 5 (57%). Catalytic hydrogenation of 5 provides 3-(l,l-dimethylheptyl)phenol, 6 (78%). Bromination of 6 occurs regioselectively and is followed by benzylation to afford the desired Grignard precursor 8 (100%).
Cuprous ion catalyzed conjugate addition of the Grignard reagent generated from 8 with cyclohexenone gives ketone 9 in 79% yield. Sodium borohydride reduction of ketone 9 in methanol gives a 4:l ratio of epimeric alcohols 10 and 11 (63%). Low reduction temperatures favor the formation of cis-alcohol 10, while reduction of ketone 9 with K-Selectride yields predominately the trans-alcohol 11. The required cis-alcohol 10 is hydrogenolyzed over palladium on carbon to yield racemic 1 (77%),and the trans-alcohol 11 similarly yields 12 (71%).
The synthesis process described above starts with the compound shown in Figure 6 and follows the outlined steps.
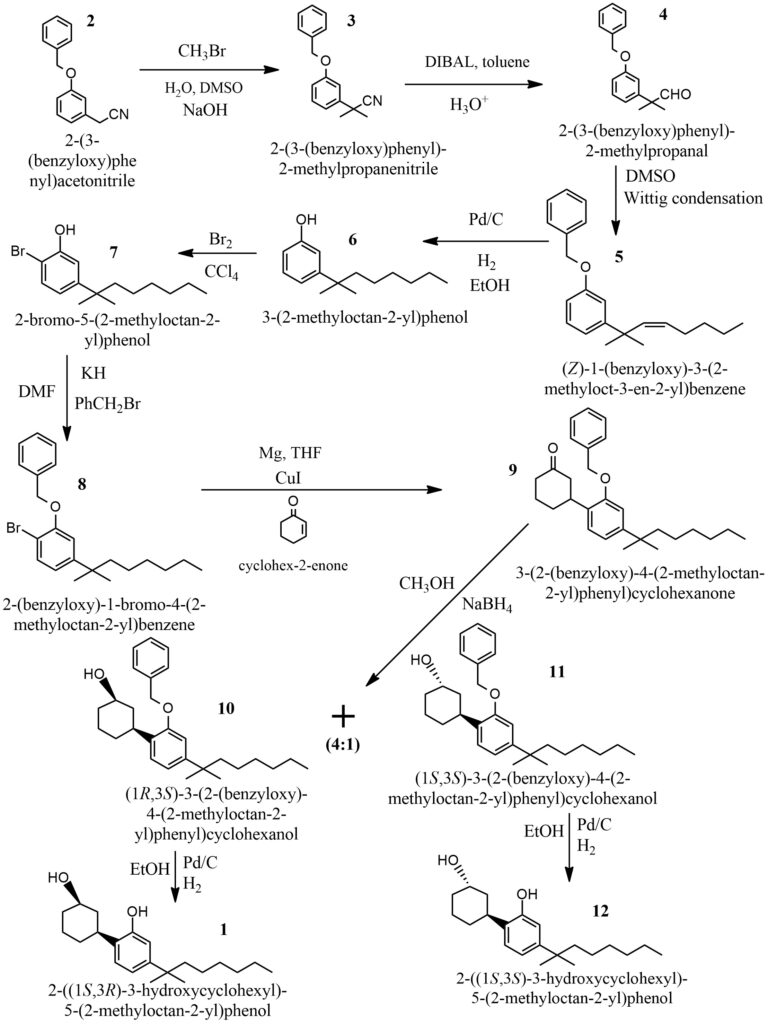
Figure 6. General scheme of CP 47497 synthesis
Conclusion
During the 1970s, Pfizer created a series of compounds known as cyclohexylphenols (CP). CP 47,497 from this series is often referred to as a “non-classical” cannabinoid in scientific literature. Compared to THC, CP 47,497 is reported to be 3-28 times more potent. Additionally, the effects of CP 47,497-C8 in humans appear to last longer than those of THC (around 5-6 hours). Notably, various products with names such as Gold, Diamond, Arctic Synergy, and Tropical Energy containing CP 47,497 were directly ordered from the internet. It is important to note that CP 47,497 is a controlled substance.
Bibliography
- https://en.wikipedia.org/wiki/CP_47,497
- https://en.wikipedia.org/wiki/(C6)-CP_47,497
- https://en.wikipedia.org/wiki/Cannabicyclohexanol
- https://en.wikipedia.org/wiki/(C9)-CP_47,497
- https://pubchem.ncbi.nlm.nih.gov/compound/12788252
- https://pubchem.ncbi.nlm.nih.gov/compound/125835
- https://pubchem.ncbi.nlm.nih.gov/compound/15942731
- https://pubchem.ncbi.nlm.nih.gov/compound/12788231
- https://pubchem.ncbi.nlm.nih.gov/compound/12788255
- https://www.chemspider.com/Chemical-Structure.29342068.html
- https://www.chemspider.com/Chemical-Structure.10205286.html
- https://www.chemspider.com/Chemical-Structure.111910.html
- https://www.chemspider.com/Chemical-Structure.26668381.html
- https://www.chemspider.com/Chemical-Structure.29342069.html
- https://www.caymanchem.com/product/10917
- https://www.caymanchem.com/product/9001448
- https://www.musechem.com/product/cannabicyclohexanol-i005530/
- https://www.musechem.com/product/cp-47497-c6-homolog-r062148/
- https://www.musechem.com/product/5-11-dimethylhexyl-2-1s3r-3-hydroxycyclohexylp-m112018/
- Weissman et al (1982). Cannabimimetic activity from CP-47,497, a derivative of 3-phenylcyclohexanol. Journal of Pharmacology and Experimental Therapeutics 223 (2):51623
- Tomiyama and Funada (2011). Cytotoxicity of synthetic cannabinoids found in Spice products: The role of cannabinoid receptors and the caspase cascade in the NG 108-15 cell line.Toxicology Letters 207(1):12-17
- John W Huffman, Jianzhong Lu etc. A Pyridone Analogue of Traditional Cannabinoids. A New Class of Selective Ligands for the CB2 Receptor. Bioorganic & Medicinal Chemistry, 2001, Volume 9, Issue 11, pp. 2863-2870. https://doi.org/10.1016/S0968-0896(01)00155-9 https://www.sciencedirect.com/science/article/abs/pii/S0968089601001559
- Liana Fattore, Walter Fratta Beyond THC: the new generation of cannabinoid designer drugs. Front Behav Neurosci., 2011, 5, 60. doi: 10.3389/fnbeh.2011.00060 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3187647/
- Understanding the ‘Spice’ phenomenon. The European Monitoring Centre for Drugs and Drug Addiction (EMCDDA). https://www.emcdda.europa.eu/publications/thematic–papers/understanding–spice–phenomenon_en
- Lawrence S. Melvin, M. Ross Johnson A cannabinoid derived prototypical analgesic. J. Med. Chem., 1984, 27, 1, pp. 67–71. https://doi.org/10.1021/jm00367a013 https://pubs.acs.org/doi/pdf/10.1021/jm00367a013
- http://bbzzzsvqcrqtki6umym6itiixfhni37ybtt7mkbjyxn2pgllzxf2qgyd.onion/threads/synthesis-of-cp-47-497.360/

