Abstract
UR-144, known as a mild synthetic cannabinoid, exhibits selective binding as a full agonist to the peripheral cannabinoid receptor CB2. It falls under the category of synthetic cannabinoids. UR-144 offers the option of oral consumption or vaporization and inhalation. This publication provides a thorough examination of UR-144, encompassing its basic details, physicochemical characteristics, recreational pharmacology, toxicity and mortality risks, dangerous interactions, effects and symptoms, popular street names, estimated prices and dosages, legal standing, synthesis methods for UR-144, conclusion, and references.
General Information About UR-144 [1-5]
Other synonyms names of UR-144 are: KM-X1; UR144; YX-17; TCMP-018; 2,2,3,3-tetramethylcyclopropyl(1-pentyl-1h-indole-3-yl) ketone
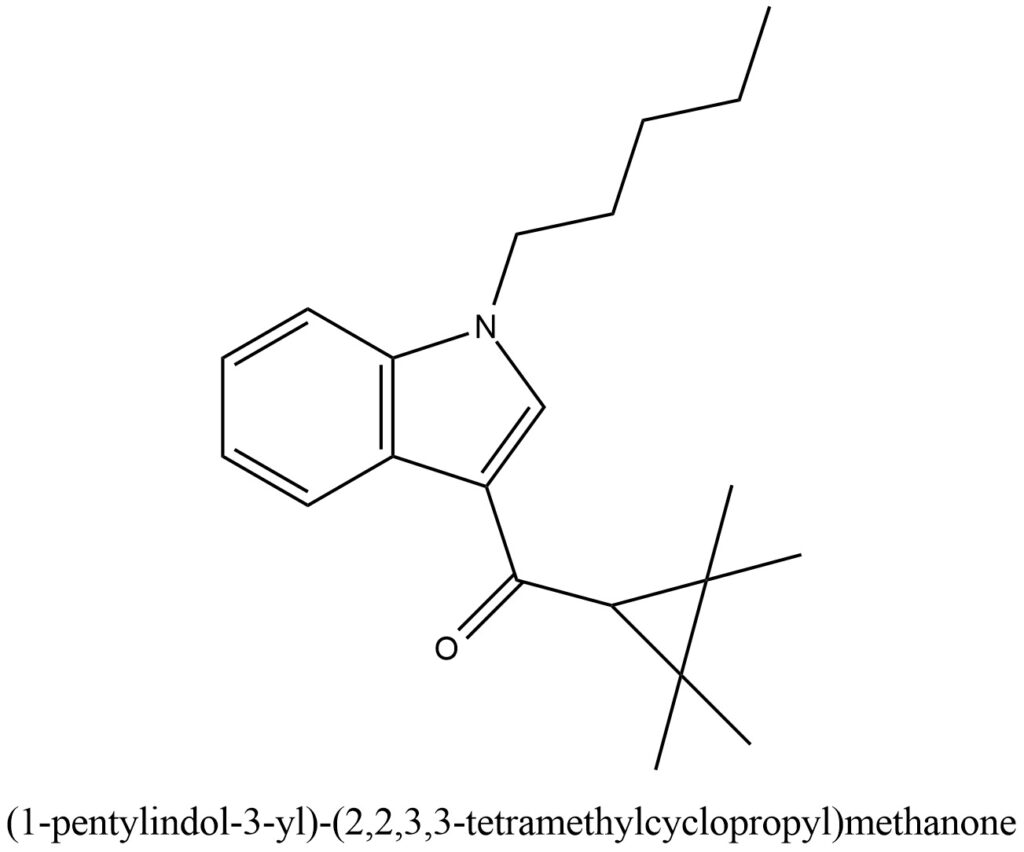
IUPAC Name of UR-144: (1-pentylindol-3-yl)-(2,2,3,3-tetramethylcyclopropyl)methanone
CAS number is 1199943-44-6
Physico-Chemical Properties of UR-144 [1-5]
- Molecular Formula C21H29NO
- Molar Weight 311.5 g/mol
- Boiling point 426.6±18.0 °C at 760 mmHg
- Melting Point 66 – 70 °C
- Color/Form: white to off-white, Solid
Structural formula present on Figure 1.
Powder and crystalline solid possible of the UR-144 can be seen in the pictures provided in Figure 2 and Figure 3.


General Information of UR-144 in Recreational Use and Pharmacology [5-10]
UR-144, a synthetic cannabinoid with moderate potency, acts as a selective full agonist on the peripheral cannabinoid receptor CB2 (EC50 = 72 nM). It also exhibits lower affinity binding to CB1 (EC50 = 421 nM). Recent studies have demonstrated its ability to hinder cell proliferation and trigger apoptotic pathways in vitro, employing distinct mechanisms. Additionally, UR-144 has been found to disrupt mitochondrial membrane potential in placental trophoblasts during in vitro experiments. Salmonella/microsome assays have further indicated the induction of DNA damage in lymphocytes, without causing gene aberrations. In vivo mouse models have shown that a dosage of 10mg/kg leads to decreased locomotor activity, catalepsy, and hypothermia, suggesting a mild cannabinoid-like effect.
Toxicity, Mortality, Danger Interactions

A sporadic consumer of SC (“Black Mamba”) succumbed to sudden death approximately 4-5 hours after their last usage. The individual was discovered in their bedroom following a tragic incident. Prior to the fatality, they had smoked a combination of “dope” and a UR-144-infused cigarette. This resulted in hallucinations, psychosis, and a loss of self-control, ultimately leading them to jump out of a second-floor window. The impact caused multiple fractures to the skull, thoracic and lumbar spine, pubic and ischial bones, as well as injuries to various organs, all stemming from the induced psychosis.
The deceased had a documented history of alcohol and drug abuse, alongside previous suicidal thoughts. They exhibited mental disorders and aggressive behavior, even injuring a witness with an axe. Their demise was attributed to asphyxia due to hanging.
Prior to another incident, the individual displayed a history of mental instability, characterized by psychomotor agitation and aggressive behavior, subsequent to smoking a legal high known as Orange Flame. This episode resulted in acute toxic liver damage, kidney failure, rhabdomyolysis, disseminated intravascular coagulation, gastrointestinal bleeding, and traumatic hematomas. These complications eventually led to cardiac arrest and massive multiorgan failure, stemming from the toxic substances’ impact.
In a separate case, an individual experienced sudden collapse after smoking an herbal blend called “Mary Joy Annihilation.” The cause of death was attributed to stenosing coronary sclerosis, accompanied by pulmonary and cerebral edema, as well as congestion of inner organs.
It is crucial to note the dangerous interactions associated with the deceased individual’s substance use, including ethanol, lidocaine, amphetamine, alprazolam, citalopram, escitalopram, hydrocodone, morphine, and pentedrone.
Effects and symptoms of UR-144 Use
UR-144 is predominantly consumed through smoking, often mixed with substances such as tobacco or herbs, aiming to induce effects similar to marijuana use. These effects include euphoria, restlessness, joyful behavior, hallucinations, increased heart rate, impaired coordination, slurred speech, drowsiness, and bloodshot eyes. During observation, individuals exhibited symptoms such as slowed or mumbled speech, dilated pupils, decreased response of pupils to light, cheerful demeanor, successful finger-to-nose coordination test, unsteady and swaying gait, excessive talkativeness, abnormal pupil reactions, constricted pupils, positive Romberg’s test (loss of balance when standing with eyes closed), hesitant lifting of objects, loss of consciousness, pale or flushed facial skin, temporary memory loss, and restlessness.
In specific instances, individuals experienced seizures, convulsions, heightened euphoria, hallucinations, difficulties in communication, aggressive behavior, aphasia (language impairment), drowsiness, delayed movements, trembling hands, and bloodshot eyes. Four cases reported tachycardia (heart rate of 110-140 beats per minute), while others exhibited heart rates ranging from 64 to 100 beats per minute (with an average of 82 beats per minute and a median of 80 beats per minute) (Figure 4).
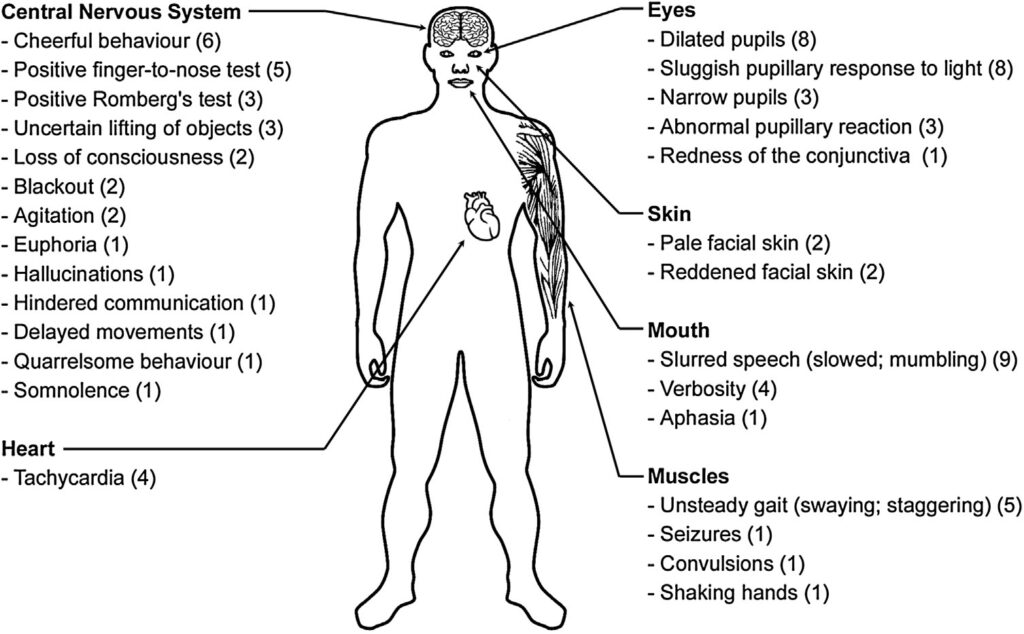
Figure 4. The effects of UR-144 observed in 35 examined individuals. In brackets the number of cases, in which the specific symptom was observed [8]
Street Names, Prices and Approximate Dosage
Other commonly used abbreviations for UR-144 include TMCP-018, KM-X1, MN-001, and YX-17.
In addition to smoking, UR-144 can be taken orally or vaporized and inhaled. According to user reports, initial doses of UR-144 range from 0.5 to 2 mg, while experienced users typically consume doses ranging from 2.5 to 20 mg. The effects of UR-144 are reported to manifest within 0.5 to 2 minutes after smoking, reach their peak after 3 to 5 minutes, and generally subside after approximately 1 to 2 hours. However, high doses may prolong the effects for up to 4 hours. It’s worth noting that tolerance to UR-144 may develop over time, leading users to consume higher doses to achieve the desired effects.
The price for UR-144 in laboratory settings is approximately $1202 for 25 mg. In other contexts, it is available at around $600 per 100 grams.
Legal Status
In October 2012, the UK Advisory Council on the Misuse of Drugs (ACMD) recommended the inclusion of UR-144 in generic prohibition legislation. Accepting this recommendation, the UK Home Office implemented legislation on February 26, 2013, classifying UR-144 as a class B drug under The Misuse of Drugs Act 1971 (Amendment) Order 2013.
In 2012, UR-144 was detected in Korea. This compound bears close resemblance to KM-X1, MN-001, YX-17, and Kr-11.
UR-144, protected by an Abbott patent, was identified as an ingredient in synthetic cannabis smoking blends in New Zealand. As a result, it was temporarily classified as a class drug and banned from sale on April 6, 2012. It has also been discovered in smoking blends and subsequently prohibited in Russia.
As of October 2015, UR-144 is classified as a controlled substance in China. It is also banned in the Czech Republic.
Regarding its regulatory status in various countries:
- Canada: Schedule II
- Germany: Anlage II (Restricted to authorized trade, not available through prescription)
- New Zealand: Temporary Class
- United Kingdom: Class B
- United States: Schedule I
Synthesis of UR-144 [10, 11]
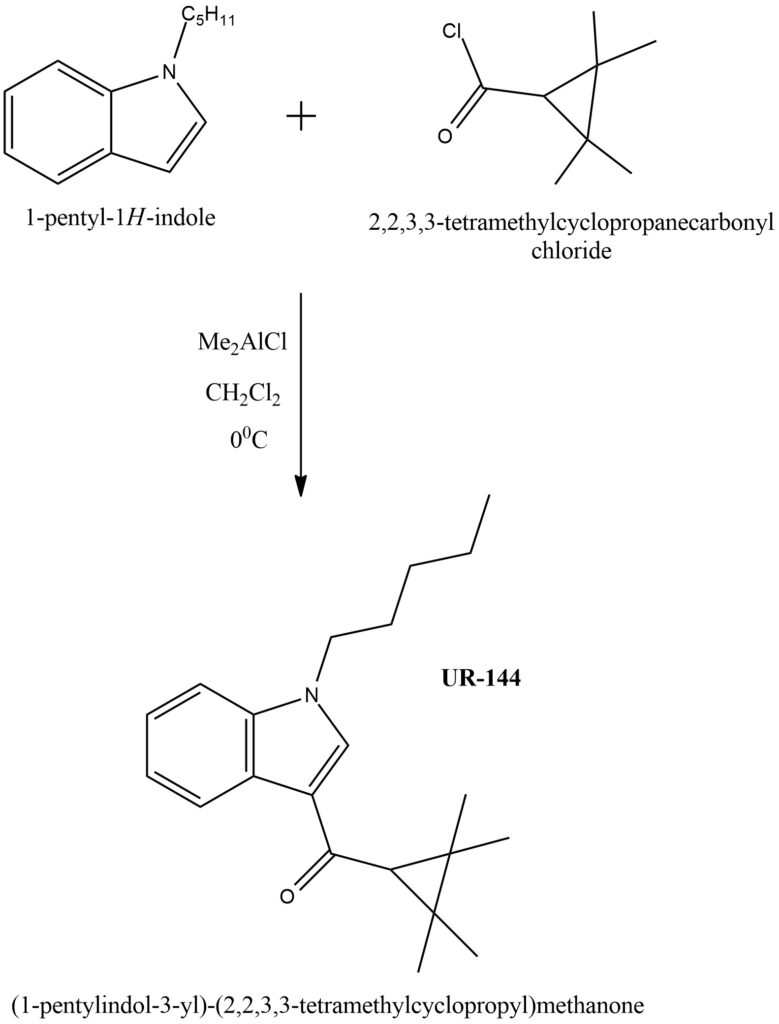
The production of UR-144 and similar cannabimimetic indoles entails utilizing 1-pentylindole and the corresponding chloride in a modified Friedel-Crafts reaction known as the Okauchi method. In this process, the indole substrate is stirred in dichloromethane alongside dimethylaluminum chloride, followed by the introduction of the acyl halide. A visual representation of the synthesis of UR-144 is depicted in Figure 4.
Conclusion
UR-144 is a synthetic cannabinoid with relatively low potency that acts as a selective full agonist on the peripheral cannabinoid receptor CB2. It is commonly consumed through smoking, often mixed with tobacco or herbs, to replicate the effects typically associated with marijuana use. UR-144 is a component of synthetic cannabis blends like Spice, which is also known as Black Mamba. It is important to note that UR-144 is deemed illegal in numerous countries due to its classification as a controlled substance.
Bibliography
- https://en.wikipedia.org/wiki/UR-144
- https://pubchem.ncbi.nlm.nih.gov/compound/44626619
- https://www.chemspider.com/Chemical-Structure.24634882.html
- https://www.lgcstandards.com/GB/en/p/LGCFOR1396.10
- Banister et al Effects of bioisosteric fluorine in synthetic cannabinoid designer drugs JWH-018, AM-2201, UR-144, XLR-11, PB-22, 5F-PB-22, APICA, and STS-135. ACS Chem. Neurosci., 2015, 6, 8, pp. 1445–1458. https://doi.org/10.1021/acschemneuro.5b00107 https://pubs.acs.org/doi/abs/10.1021/acschemneuro.5b00107
- Almada et al Synthetic cannabinoids JWH-018, JWH-122, UR-144 and the phytocannabinoid THC activate apoptosis in placental cells. Toxicology Letters, 2020, 319, pp. 129-137. https://doi.org/10.1016/j.toxlet.2019.11.004 https://www.sciencedirect.com/science/article/pii/S0378427419303558
- Koller et al Genotoxic properties of representatives of alkylindazoles and aminoalkyl-indoles which are consumed as synthetic cannabinoids. Food Chemical Toxicology, 2015, 80, pp. 130-136. https://doi.org/10.1016/j.fct.2015.03.004 https://www.sciencedirect.com/science/article/abs/pii/S0278691515000836
- Piotr Adamowicz, Joanna Gieron The effects of synthetic cannabinoid UR-144 on the human body — A review of 39 cases. Forensic Science International, 2017, Volume 273, pp. e18-e21. https://doi.org/10.1016/j.forsciint.2017.02.031 https://www.sciencedirect.com/science/article/abs/pii/S0379073817300853#:~:text=The%20determined%20concentrations%20of%20UR,%2C%20poor%20coordination%2C%20and%20staggering
- Arianna Giorgetti, Francesco Paolo Busardo Post-Mortem Toxicology: A Systematic Review of Death Cases Involving Synthetic Cannabinoid Receptor Agonists. SYSTEMATIC REVIEW article. Front. Psychiatry, 2020, Sec. Addictive Disorders, Volume 11, 464, 22 p. https://doi.org/10.3389/fpsyt.2020.00464 https://www.frontiersin.org/articles/10.3389/fpsyt.2020.00464/full
- John W. Huffman, Paul V. Szklennik 1-Pentyl-3-phenylacetylindoles, a new class of cannabimimetic indoles. Bioorganic & Medicinal Chemistry Letters, 2005, 15, 18, pp. 4110–4113. https://doi.org/10.1016/j.bmcl.2005.06.008 https://www.sciencedirect.com/science/article/abs/pii/S0960894X05007481
- http://bbzzzsvqcrqtki6umym6itiixfhni37ybtt7mkbjyxn2pgllzxf2qgyd.onion/threads/tmcp-indole-synthesis-ur-144-intermediate-cas-895152-66-6-large-scale.313/



