- The process of creating a drug - May 23, 2023
- How do scientists model mental disorders in animals? - May 23, 2023
Introduction
Methamphetamine, also known as ‘crystal meth’, is a highly addictive stimulant drug that has been used for recreational and medical purposes for many years. It has a high potential for abuse and has been linked to a range of serious health risks. This article will provide an overview of the chemical and physical properties of methamphetamine, discuss the various approaches to its synthesis, and conclude with a summary of the findings. By understanding the properties of methamphetamine and the various synthesis approaches, readers will have a better understanding of this powerful drug.
Methamphetamine General information
Methamphetamine is a highly potent stimulant of the central nervous system, usually used recreationally and occasionally as a substitute remedy for attention deficit hyperactivity disorder and obesity. Its discovery dates back to 1893, and it comes in two enantiomers: levo(-)-methamphetamine and dextro(+)-methamphetamine, first synthesized from ephedrine by the Japanese chemist Nagai Nagayoshi. Crystal methamphetamine was synthesized in 1919 by Japanese chemist Akira Ogata. The compound known as methamphetamine is, specifically, the racemic free base, consisting of equal parts of levomethamphetamine and dextromethamphetamine in their amine forms. Dextromethamphetamine is a more potent CNS stimulant than levomethamphetamine; its more rapid passage through the blood-brain barrier is attributed to its higher lipid solubility. It exerts its effects by augmenting the levels of serotonin, dopamine, and norepinephrine in the brain.
Methamphetamine falls under the chemical classes of substituted phenethylamines and substituted Amphetamines. It has the same chemical formula C10H15N as other dimethylphenethylamines, being one of them in terms of position.
The Controlled Substances Act lists it as a Schedule II stimulant, meaning it could be easily abused and holds a legal medical purpose (in FDA-approved products). A prescription is needed for accessing it and is not refillable. The only existing meth product is Desoxyn®, available in 5, 10, and 15-milligram tablets with immediate and extended release formulations, employed for the treatment of obesity and ADHD.
Methamphetamine Chemical Properties
Methamphetamine is a chiral chemical compound with two distinct forms, dextromethamphetamine and levomethamphetamine. At room temperature, the free base of methamphetamine is a transparent and colorless liquid with a recognizable geranium leaves smell. It is soluble in diethyl ether and ethanol and can be mixed with chloroform. Dextromethamphetamine is a more potent central nervous system (CNS) stimulant compared to levomethamphetamine; nevertheless, both are thought to be dependence-forming and addictive when abused and capable of causing similar toxicity symptoms at high recreational doses.
Conversely, the hydrochloride salt of methamphetamine has no scent with a sour taste. It has a melting point between 170 and 175 °C (338 and 347 °F) and is usually presented as white crystals or a white powder at room temperature. The hydrochloride salt is also easily soluble in ethanol and water. In a 2013 study of bioreactors in wastewater, it was discovered that methamphetamine is generally degraded within 30 days under the influence of light.
IUPAC name: (RS)-N-methyl-1-phenylpropan-2-amine
Other names: Batu, Bikers Coffee, Black Beauties, Chalk, Chicken,Feed, Crank, Crystal, Glass, Go-Fast, Hiropon, Ice, Meth, Methlies Quick, Poor Man’s Cocaine, Shabu, Shards, Speed, Stove Top, Tina, Trash, Tweak, Uppers, Ventana, Vidrio, Yaba, and Yellow Bam, Desoxyn, Methedrine, N-methylamphetamine, N,α-dimethylphenethylamine, desoxyephedrine;
Methamphetamine Physical Properties
- Formula C10H15N;
- Molar mass 149.237 g/mol;
- Density 0.9 g/cm3 at 25 °C;
- Melting point 170 – 175 °C (338 – 347 °F) for hydrochloride salt;
- Boiling point 212 °C (414 °F) at 760 mmHg;
Methamphetamine Synthesis Approaches
The Leuckart or reductive amination methods can be used to make racemic methamphetamine from phenylacetone. The Leuckart reaction involves reacting one part phenylacetone with two parts N-methylformamide, generating the formyl amide of methamphetamine, carbon dioxide and methylamine as byproducts. This process leads to the formation of an iminium cation which is then reduced by the additional N-methylformamide. The ensuing formyl amide is then broken down with acidic water to get methamphetamine.
Instead of phenylacetone (P2P), methamphetamine can be produced by reacting it with methylamine under reducing circumstances. This reduction process can be accomplished using aluminum amalgam (Al/Hg).
Additionally, using NaBH4, P2P can be converted to methamphetamine through a series of reactions.
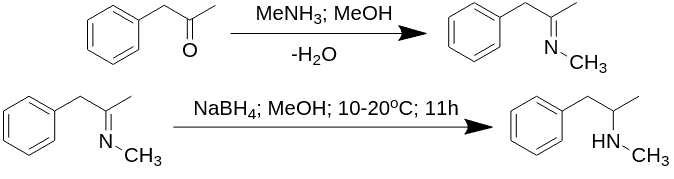
A common way to obtain methamphetamine is through the reduction of ephedrine, which can be extracted from over-the-counter medications.
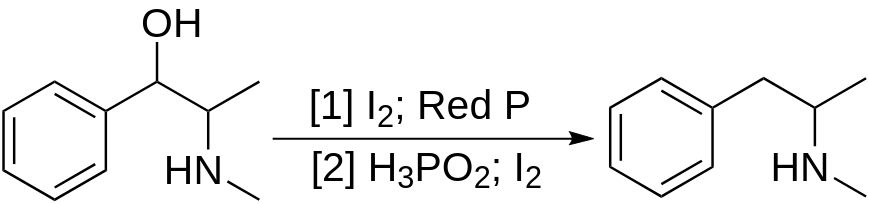
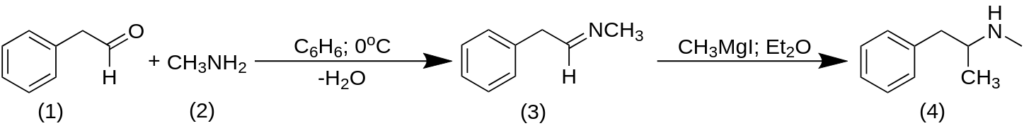
Methamphetamine can be produced using phenylacetaldehyde in two steps, one of which involves the use of a Grignard reagent.
The same process used to separate Amphetamine can be utilized to separate the d– and l-methamphetamine isomers with the aid of tartaric acid.
Conclusion
Methamphetamine is a powerful stimulant drug with a wide range of potential effects. It has a high potential for abuse and can lead to an array of negative health consequences. However, when used safely and responsibly, it can be an effective tool to treat certain medical conditions. By understanding the general information, chemical properties, and physical properties of methamphetamine, as well as the various synthesis approaches available, it is possible to safely and responsibly use this drug. With this knowledge, we can make informed decisions about the use of methamphetamine and ensure its safe and responsible use.
Bibliography
- Allen, Andrew, and Thomas S. Cantrell. “Synthetic reductions in clandestine amphetamine and methamphetamine laboratories: a review.” Forensic science international 42.3 (1989): 183-199. https://www.sciencedirect.com/science/article/abs/pii/0379073889900868
- Skinner, Harry F. “Methamphetamine synthesis via hydriodic acid/red phosphorus reduction of ephedrine.” Forensic science international 48.2 (1990): 123-134. https://www.sciencedirect.com/science/article/abs/pii/0379073890901047
- Puder, Karoline S., Doreen V. Kagan, and John P. Morgan. “Illicit methamphetamine: analysis, synthesis, and availability.” The American journal of drug and alcohol abuse 14.4 (1988): 463-473. https://www.tandfonline.com/doi/abs/10.3109/00952998809001564
- G.Patton “Methamphetamine” BB Wiki http://bbzzzsvqcrqtki6umym6itiixfhni37ybtt7mkbjyxn2pgllzxf2qgyd.onion/wiki/methamphetamine/
- https://en.wikipedia.org/wiki/Methamphetamine

