- The process of creating a drug - May 23, 2023
- How do scientists model mental disorders in animals? - May 23, 2023
Introduction
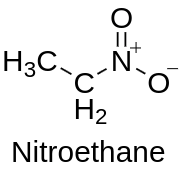
Nitroethane, with the chemical formula C2H5NO2, is an organic compound that belongs to the family of nitro compounds. It is a colorless liquid with a distinctive odor, and it has gained significant attention due to its diverse applications in various industries. In this article, we will explore the chemical composition, structure, physical properties, synthesis methods, applications, safety considerations, and uses of nitroethane.
Nitroethane: Chemical Composition, Structure, and Physical Properties
Nitroethane consists of two carbon atoms, five hydrogen atoms, and one nitro group (-NO2) attached to one of the carbon atoms. It has a linear molecular structure, with the nitro group bonded to the carbon atom through a single bond and the other carbon atom bonded to three hydrogen atoms.
The nitro group (-NO2) is a functional group composed of a nitrogen atom bonded to two oxygen atoms. It imparts unique chemical properties to nitroethane, including its high reactivity and oxidizing ability.
Nitroethane is a colorless liquid with a slightly sweet odor. It has a boiling point of 114 °C (237 °F) and a melting point of -51 °C (-60 °F). It is slightly soluble in water but highly soluble in most organic solvents, making it a versatile compound for various chemical reactions and applications.
It has a molecular weight of 75.07 g/mol and a density of approximately 1.05 g/cm3 at room temperature.
The physical and chemical properties of nitroethane are influenced by the presence of the nitro group, which imparts certain characteristics to the compound. The nitro group is a highly polar and electron-withdrawing group, which results in the overall polarity of nitroethane. This polarity leads to its relatively high boiling point and low vapor pressure compared to non-polar compounds of similar molecular weight.
The chemical structure and properties of nitroethane also make it susceptible to various chemical reactions. For example, the nitro group can undergo reduction reactions, where it can be converted to amino groups (-NH2) through hydrogenation or other reduction methods. Nitroethane can also participate in condensation reactions, such as nitroaldol reactions, where it can react with other compounds to form new chemical bonds and create complex organic molecules.

Synthesis of Nitroethane: Chemical Reactions and Production Methods
From Sodium Ethyl Sulfate and a Metal Nitrite
One common method for synthesizing nitroethane is by reacting sodium ethyl sulfate (NaC2H5SO4) with a metal nitrite, such as sodium nitrite (NaNO2) or potassium nitrite (KNO2). The reaction involves the displacement of the sulfate group in sodium ethyl sulfate by the nitrite group, resulting in the formation of nitroethane.
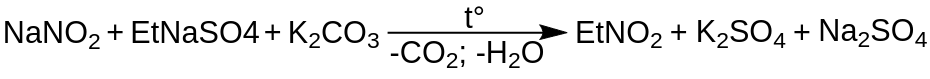
From Ethyl Halide and Silver Nitrite
Another method for synthesizing nitroethane is through the reaction between an ethyl halide, such as ethyl bromide (C2H5Br) or ethyl iodide (C2H5I), and silver nitrite (AgNO2). This reaction proceeds through a substitution reaction, where the halide group in the ethyl halide is replaced by the nitro group from silver nitrite, resulting in the formation of nitroethane. The reaction is typically carried out in diethyl ether, at 0 °C for 24h.
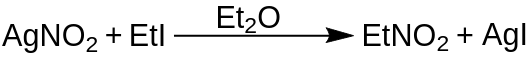
From Ethyl Bromide (Iodide) and Sodium Nitrite
A third method for synthesizing nitroethane involves the reaction between ethyl bromide (C2H5Br) or ethyl iodide (C2H5I) and sodium nitrite (NaNO2). This reaction also proceeds through a substitution reaction, where the halide group in the ethyl bromide or ethyl iodide is replaced by the nitro group from sodium nitrite, resulting in the formation of nitroethane. The reaction is typically carried out in a DMSO solvent.
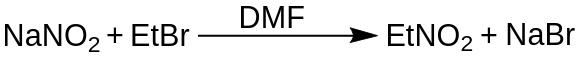
Applications of Nitroethane in Various Industries: From Chemistry to Motorsports
Nitroethane, with its unique chemical properties and reactivity, finds applications in various industries, ranging from chemistry to motorsports. Let’s take a closer look at some of the key applications of nitroethane in different industries.
Chemical Industry
In the chemical industry, nitroethane serves as an important raw material and intermediate for the synthesis of various chemicals. It is commonly used as a solvent, reagent, or reactant in chemical reactions. Nitroethane is often employed as a starting material in the synthesis of pharmaceuticals, agrochemicals, dyes, and other fine chemicals. It can also be used as a reagent for the preparation of other organic compounds, such as alkylated amines, alkylated ketones, and alkylated carboxylic acids. Nitroethane’s ability to undergo various chemical reactions, such as nucleophilic substitution, reduction, and condensation, makes it a versatile compound in the chemical industry.
Explosives Industry
Nitroethane is used in the production of explosives, particularly as a component in the manufacture of blasting agents and propellants. It is known for its high energy content and fast burning rate, which makes it suitable for use in explosive formulations. Nitroethane is often used in combination with other explosives or oxidizers to enhance their performance, such as in detonators, blasting caps, and pyrotechnic compositions. However, due to its high reactivity and potential for explosion, the handling and use of nitroethane in the explosives industry requires strict safety protocols and regulations.
Research and Development
Nitroethane is also used in research and development activities, particularly in the field of organic chemistry. It serves as a valuable reagent for the synthesis of new compounds and the modification of existing molecules. Nitroethane can be used in various reactions, such as condensation, reduction, and substitution, to introduce specific functional groups or modify the structure of organic molecules. Additionally, nitroethane is used in analytical chemistry as a reference material or standard for calibration purposes in gas chromatography and other analytical techniques.
Other Applications
Nitroethane finds applications in other industries as well. It is used as a solvent in some cosmetic and personal care products, as a flavoring agent in the food industry, and as a component in some cleaning agents and coatings. Nitroethane is also utilized in some niche applications, such as in the production of model rocket engines and in the manufacturing of specialty chemicals for specific industrial processes.
It’s worth noting that the handling and use of nitroethane in various industries require strict adherence to safety regulations and guidelines due to its hazardous properties, including its flammability, reactivity, and potential for explosion. Proper precautions, such as adequate ventilation, proper storage, and appropriate personal protective equipment (PPE), should be followed when working with nitroethane to ensure the safety of personnel and the environment.
Nitroethane in Chemical Syntheses
Nitroethane finds several applications in chemical synthesis, particularly in organic chemistry. Some of its prominent uses are as follows.
Nitroethane is used as a precursor for the synthesis of amines. Amines are a class of organic compounds that contain a nitrogen atom bonded to one or more alkyl or aryl groups. The reduction of nitroethane with metal hydrides such as sodium borohydride or lithium aluminum hydride produces primary amines.
Through reactions such as the Henry process, nitroethane can be transformed into numerous commercially important compounds. Combining it with 3,4-dimethoxybenzaldehyde produces the precursor for the antihypertensive drug methyldopa, while combining it with an unsubstituted benzaldehyde results in 1-phenyl-2-nitropropene, a precursor for amphetamine-based drugs. Through a two-step process of condensation and hydrogenation, 2-amino-2-methyl-1,3-propanediol is created, which then condenses with oleic acid to form an oxazoline. This can be protonated to produce a cationic surfactant.
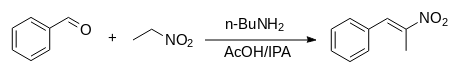
Nitroethane is also utilized as a fuel additive and a precursor for rockets. It is a useful solvent for polymers such as polystyrene and is especially useful for dissolving cyanoacrylate adhesives. It has been employed in the cosmetics industry as an ingredient in nail remover and overhead sealant sprays.

Previously, nitroethane was widely used as a feedstock in the creation of many substances and consumer goods. For instance, Pervitin (methamphetamine) was popular in the 19th and 20th centuries, and during WWII was frequently taken by troops of both sides for mood elevation, appetite and sleep suppression, and increasing focus and alertness. Nitroalkanes were among the ingredients used in the manufacture of various phenethylamines, including drugs such as Pervitin and Benzedrine (amphetamine), which was utilized as an anorectic medication for obesity.
Safety Considerations When Handling Nitroethane: Hazards and Precautions
- Nitroethane is a highly flammable and explosive compound, and proper safety precautions must be followed when handling it. It can form explosive mixtures in air, and ignition sources should be strictly controlled in its presence.
- Nitroethane is toxic when ingested or absorbed through the skin, and it can cause irritation to the eyes, skin, and respiratory tract. Personal protective equipment, such as gloves, goggles, and adequate ventilation, shouldbe used when handling nitroethane to minimize exposure and prevent potential health hazards.
- Nitroethane can react violently with oxidizing agents, strong acids, and bases, and should be stored and handled away from incompatible substances.
- Proper storage and transportation procedures should be followed, including using approved containers, avoiding exposure to heat and flames, and ensuring adequate ventilation in storage areas.
Conclusion
In conclusion, nitroethane is a versatile organic compound with diverse applications in various industries, including chemistry, explosives, motorsports, and food and beverage. It is synthesized through several methods, and its chemical and physical properties make it suitable for different reactions and processes. However, safety precautions must be strictly followed when handling nitroethane due to its flammability, toxicity, and potential for hazardous reactions. Nitroethane plays a crucial role in chemical syntheses, serving as a reagent and intermediate in the production of a wide range of organic compounds. Understanding the properties, synthesis methods, applications, and safety considerations of nitroethane is essential for its safe and effective use in various industries.
Bibliography
- Sheldon B. Markofsky “Nitro Compounds, Aliphatic” in Ullmann’s Encyclopedia of Industrial Chemistry, Wiley-VCH, Weinheim, 2002. https://onlinelibrary.wiley.com/doi/10.1002/14356007.a17_401
- Wang, Qingsheng, Dedy Ng, and M. Sam Mannan. “Study on the reaction mechanism and kinetics of the thermal decomposition of nitroethane.” Industrial & engineering chemistry research 48.18 (2009): 8745-8751. https://pubs.acs.org/doi/abs/10.1021/ie900849n
- Ruano, Jose Luis Garcia, et al. “Preparation of α-amino ketones, β-amino hydroxylamines using asymmetric aza-Henry reactions of Np-tolylsulfinylimines with nitroethane.” Tetrahedron 62.52 (2006): 12197-12203. https://www.sciencedirect.com/science/article/pii/S0040402006016401
- Reich, Hans. “Bordwell pKa table: “Nitroalkanes””. University of Wisconsin Chemistry Department. Retrieved 17 January 2016.
- Matthews, Walter; et al. (1975). “Equilibrium acidities of carbon acids. VI. Establishment of an absolute scale of acidities in dimethyl sulfoxide solution”. Journal of the American Chemical Society. 97 (24): 7006. https://pubs.acs.org/doi/abs/10.1021/ja00857a010
- Fayyazbakhsh, Ahmad, and Vahid Pirouzfar. “Investigating the influence of additives-fuel on diesel engine performance and emissions: Analytical modeling and experimental validation.” Fuel 171 (2016): 167-177. https://www.sciencedirect.com/science/article/pii/S001623611501282X
- https://en.wikipedia.org/wiki/Nitroethane

