Phenylacetic Acid Properties, Reactions and Applications
Introduction
Phenylacetic acid also known as α-phenylacetic acid or benzenacetic acid, is one of the most interesting and important compound in organic chemistry. This aromatic carboxylic component has unique properties and chemical characteristics. The purpose of this article is to provide an overview of phenylacetic acid synthesis, properties, reactions and uses of phenylacetic acid.
It was discovered in 1885. Phenylacetic acid is found in tobacco, cocoa and chicory from which it can be extracted in small quantities. Synthetic phenylacetic acid is more produced than natural.
Phenylacetic acid is the starting product for the synthesis of phenylacetone, an intermediate of amphetamine and methamphetamine, which have a strong stimulating effect. As a result, it is under special control in many countries.
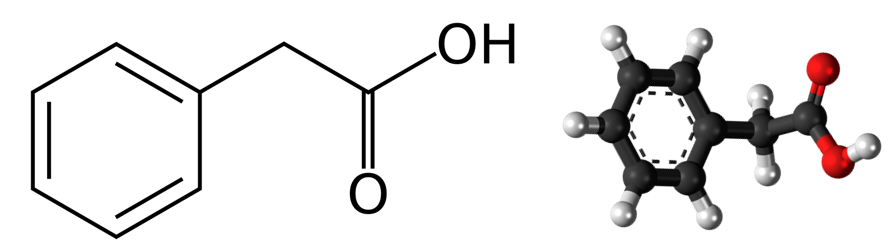
Physical and Chemical Properties of Phenylacetic Acid
The physicochemical properties of phenylacetic acid have a significant impact on its properties in chemical reactions and on possible applications in science and industry. Phenylacetic acid is a weak carboxylic acid with a molecular weight – 136.15 g/mol and a density – 1.08 g/cm³. In pure form, phenylacetic acid is colorless crystals or white powder with a characteristic odor. The melting point of phenylacetic acid is about 77°C and the boiling point is about 265°C. Phenylacetic acid is slightly soluble in water and soluble in various organic solvents such as ethanol, diethyl ether and acetone.
Synthesis of Phenylacetic Acid
Phenylacetic acid can be obtained by hydrolysis of phenylacetonitrile, which is the most common route of synthesis. Sulfuric acid is often used as a catalyst, usually at a concentration of 70%, but concentrated hydrochloric acid can also be used.
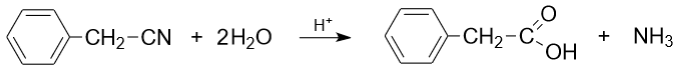
It is also possible to obtain phenylacetic acid by oxidation of phenylacetaldehyde with copper (II) hydroxide.

Another way to obtain phenylacetic acid by hydrolysis of a salt, such as sodium phenylethanoate.

Obtaining phenylacetic acid by acid hydrolysis of phenylacetate ester such as methylphenylacetate.

Phenylacetic acid can also be obtained by biosynthesis via microorganisms (e.g., Bacteroidaceae) by different metabolic pathways.
These synthetic methods provide a variety of routes to obtain phenylacetic acid of varying complexity. The choice of one or another method depends on the available reagents.

Chemical Properties of Phenylacetic Acid
Salt formation: Phenylacetic acid forms salts when reacted with bases such as metal hydroxides. For example, the sodium salt of phenylacetic acid (sodium phenylacetate) is formed upon reaction with sodium hydroxide.
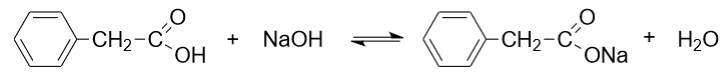
Esterification: Phenylacetic acid can form esters (esters) with various alcohols by reaction with strong acids or acid catalysts. For example, the reaction of the acid with methanol produces methyl ester of phenylacetic acid.

Reaction with halogens: Phenylacetic acid can be halogenated by reaction with chlorides or bromides to form halogen derivatives of the corresponding halogens (e.g. chloro- and bromo-phenylacetic acids).

Ketone decarboxylation: Heating a mixture of phenylacetic acid and acetic anhydride gives phenylacetone. Other acetates such as sodium and potassium acetate can be used, giving different yields. A large excess of acetic anhydride must be used in this reaction, otherwise dibenzyl ketone will be formed.

Reaction with amines: Phenylacetic acid can form amides when reacted with amines in the presence of an acid catalyst.
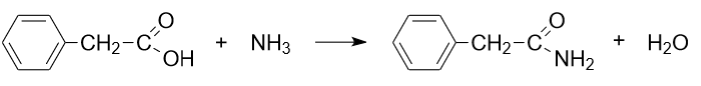
These chemical properties and reactions of phenylacetic acid play an important role in its applications in organic synthesis, drug production and other fields of science and industry.

Applications of Phenylacetic Acid
1. Organic synthesis: Phenylacetic acid is widely used in organic synthesis as an important component for the preparation of various organic compounds.
2. Manufacture of pharmaceuticals: Phenylacetic acid plays an important role in the manufacture of pharmaceuticals. Its derivatives can be used as drug additives and intermediates for the synthesis of active components of many drugs.
3. Agriculture: Phenylacetic acid is used in agriculture to enhance the absorption of micronutrients from the ground.
4. Polymer industry: Phenylacetic acid can be used as a component in the production of various plastics.
5. Industrial Processes: Phenylacetic acid can be used in various chemical processes in industry, including synthesis of various organic compounds, production of food additives, dyes and as a reagent for analytical research.

Health Effects of Phenylacetic Acid
Phenylacetic acid, like many chemical compounds, can have negative health effects. Appropriate precautions should be taken when handling it and using it in various processes. Some aspects of the health effects of phenylacetic acid are listed below:
- Irritant effect: Phenylacetic acid in its pure form may cause irritation to the skin, eyes and mucous membranes.
- Toxicity: Health effects may occur from inhalation of vapors or aerosols of phenylacetic acid. Precautions are especially necessary when working with large volumes or in enclosed spaces.
- Digestive System: If phenylacetic acid is ingested, it can cause irritation to the stomach and intestines. In case of internal contact with phenylacetic acid, seek medical advice.
- Cumulative effects: With long-term exposure to phenylacetic acid in people who produce or use the compound, the acid accumulates in the body over time and causes negative effects.
Safety Precautions
1. Storage: Phenylacetic acid is stored in a well-closed container, avoiding moisture and air. Keep it separate from other chemicals to avoid possible reactions.
2. Ventilation: Handle phenylacetic acid in a well-ventilated area or under an exhaust fume hood. This will minimize body exposure to vapors or aerosols.
3. Use of personal protective equipment: When working with phenylacetic acid, use personal protective equipment such as chemical resistant gloves, goggles and lab coats. If there are respirators, use them for respiratory protection.
4. Avoid contact with skin and eyes: Avoid contact with skin and eyes when working with phenylacetic acid. In case of accidental contact with skin, rinse immediately with plenty of water. In case of contact with eyes, rinse with running water for several minutes and seek medical advice.
Following these precautions will help prevent possible risks and minimize health effects when working with phenylacetic acid.
Conclusion
Phenylacetic acid is an important organic compound with unique properties and chemical reactions. Its ability to form various derivatives makes it a valuable tool in organic synthesis, drug production, polymerization and other functional materials. The applications of phenylacetic acid are varied, from research to industry. Precautions must be taken when working with phenylacetic acid to minimize health risks.
In general, phenylacetic acid is an important component for the chemical industry and scientific research, its unique properties open opportunities for creating new materials, drugs and technologies, which makes it a subject of interest for further research and development in chemistry.
Bibliography
- An Historical Review of Phenylacetic Acid Sam D Cook Plant and Cell Physiology, Volume 60, Issue 2, February 2019, Pages 243–254 Url: https://academic.oup.com/pcp/article/60/2/243/5289543
- Phenylacetic acid D. J. Hodgson and R. O. Asplund, Acta Crystallographica Section C: Structural Chemistry, 1991 C47, 1986-1987 Url: https://doi.org/10.1107/S0108270191002275
- Solubility of Phenylacetic Acid, p-Hydroxyphenylacetic Acid, p-Aminophenylacetic Acid, p-Hydroxybenzoic Acid, and Ibuprofen in Pure Solvents, Sandra Gracin and Åke C. Rasmuson, J. Chem. Eng. Data 2002, 47, 6, 1379–1383 Url: https://doi.org/10.1021/je0255170
- Production of Phenylacetic Acid by Anaerobes, D. Mayrand, G. Bourgeau, ASM Journals
- Journal of Clinical Microbiology, Vol. 16, No. 4, 1 October 1982 Url: https://doi.org/10.1128/jcm.16.4.747-750.1982
- Chemical profiling of seized methamphetamine putatively synthesized from phenylacetic acid derivatives, Kenji Tsujikawa and Kenji Kuwayama, Forensic Science International Volume 227, Issues 1–3, 10 April 2013, Pages 42-44 Url: https://doi.org/10.1016/j.forsciint.2012.08.036

