- The process of creating a drug - May 23, 2023
- How do scientists model mental disorders in animals? - May 23, 2023
Abstract
AM-2201 is a synthetic cannabinoid that acts as a full agonist for cannabinoid receptors. Although marijuana has been used for centuries for its medicinal properties, its psychoactive effects and abuse potential have hindered its widespread use as a medicine, leading to its continued prohibition in many countries. Synthetic cannabinoids such as AM-2201 have emerged as an alternative to marijuana, but have been associated with serious health risks, including death.
This article provides a comprehensive overview of AM-2201, including its general information, physical and chemical properties, pharmacology in recreational use, effects and symptoms, toxicity, street names, prices and approximate dosages, legal status, synthesis methods, conclusions, and bibliography. The article highlights the potential dangers of synthetic cannabinoids and emphasizes the importance of caution and harm reduction measures when using these substances.

General Information About AM 2201 [1-6]
Other synonyms names of AM 2201 are: 1-(5-fluoropentyl)-3-(1-naphthoyl)indole; [1-(5-Fluoropentyl)-1H-indol-3-yl]-1-naphthalenylmethanone;
IUPAC Name of AM 2201: [1-(5-fluoropentyl)indol-3-yl]-naphthalen-1-ylmethanone
CAS number is 335161-24-5
Trade names are AM 2201; JWH-2201

Physico-Chemical Properties of AM 2201 [1-6]
- Molecular Formula C24H22FNO
- Molar Weight 359.4 g/mol
- Boiling point 551.1±30.0 °C at 760 mmHg
- Melting Point 86 – 88 °C
- Flash Point: 287.1±24.6 °C
- Solubility: slightly soluble, Chloroform, Ethyl acetate; Soluble in DMSO, not in water
- Color/Form: Solid; light yellow
- Target: Cannabinoid Receptor Agonist
Structural formula present on Figure 1.
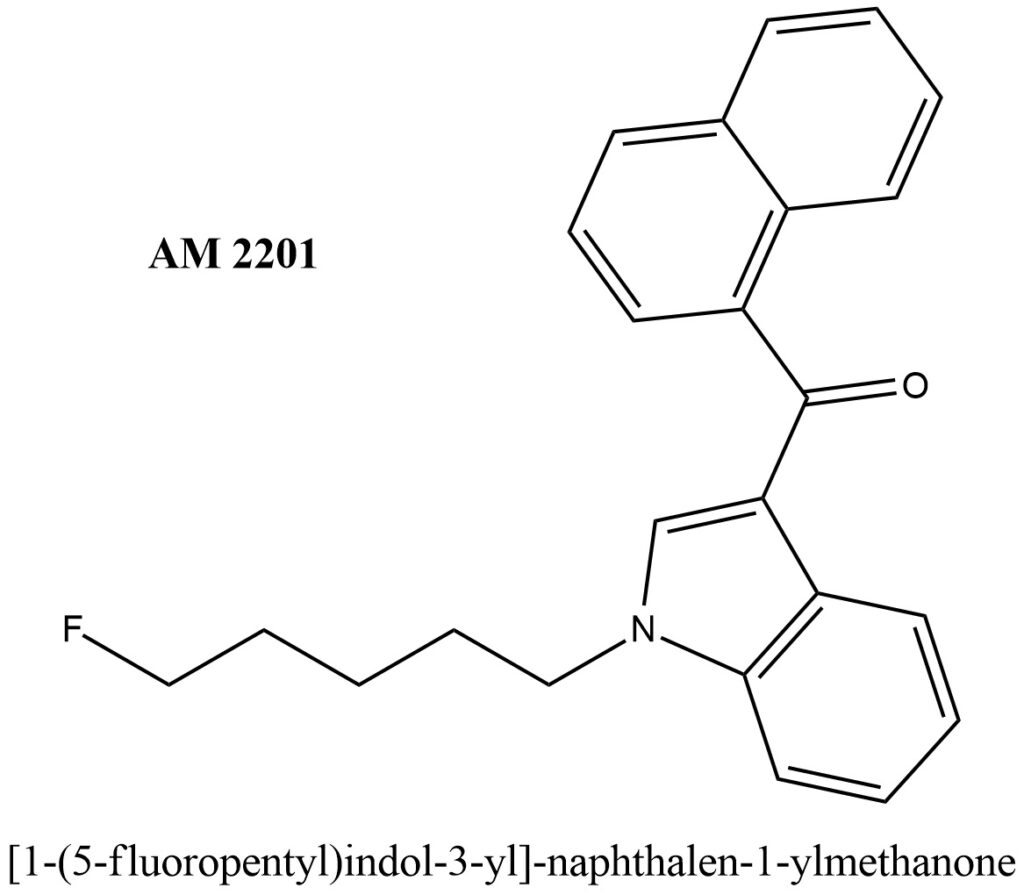
Figure 1. Structure of AM 2201
Powder and crystalline solid possible of the AM 2201 can be seen in the pictures provided in Figure 2 and Figure 3.

Figure 2. Powder of AM 2201

Figure 3. Crystalline solid of AM 2201
General Information of AM 2201 in Recreational Use and Pharmacology [7-12]

Marijuana has been used for centuries for its medicinal properties, its psychoactive effects and potential for abuse have limited its acceptance as a medicine, with many countries continuing to prohibit its use. Efforts to develop new cannabinoids with therapeutic potential and no psychoactivity have led to the discovery of compounds such as AM-2201.
AM-2201 is a recreational designer drug that acts as a potent but nonselective full agonist for the cannabinoid receptor. In recent years, there has been an increase in the use of fluorinated synthetic cannabinoids, including AM-2201, which has been linked to a patent awarded to Makriyannis and colleagues. This patent demonstrated that terminally fluorinated AM-2201 has greater affinity for CB1 than the des-fluoro analogue, JWH-018, and could potentially be used as a radioactive probe in positron emission tomography (PET) imaging.
Recent seizures have identified several compounds, including AM-2201 and AM-694, as well as fluorinated analogues of previously known synthetic cannabinoids. In fact, South Korea reported that 90% of synthetic cannabinoids seized in 2013 were fluorinated, compared to 60% in 2012 and just 15% in 2011, with none reported before 2011.
Despite their popularity, synthetic cannabinoids such as AM-2201 pose significant health risks to users, including death. It is important for individuals to be aware of the dangers associated with these substances and to exercise caution when using them. Medical professionals must also remain vigilant in monitoring and addressing the health consequences of synthetic cannabinoid use.
Toxicity
While not all synthetic cannabinoids are inherently harmful or addictive, the lack of proper testing makes it difficult to determine which products may lead to serious side effects, addiction, or even death. Furthermore, many synthetic products contain multiple compounds that are not publicly disclosed, increasing the risk of adverse effects. Additionally, the addition of other substances like benzoates or opiates further increases the danger of using these products.
Comparatively, synthetic cannabinoids appear to be more toxic than natural cannabis, partly due to their higher potency and the difficulty in properly dosing them. Acute toxic effects can be severe and even fatal in certain circumstances. Studies on rats have shown that AM-2201 produces bradycardia and hypothermia at doses similar in potency to JWH-018, indicating potent cannabinoid-like activity. There have also been reports of deaths associated with the use of synthetic cannabis.
Some synthetic cannabinoids are up to 100 times more potent than naturally occurring ones, which can lead people to consume too much and suffer from overdose. Most overdose deaths resulting from the use of synthetic cannabinoids have been linked to dangerous foreign substances like opiates or benzodiazepines. There is also evidence to suggest that some batches of synthetic cannabinoids are more hazardous than others. For instance, in May 2018, almost 100 individuals died and hundreds more experienced severe side effects after using Spice. Similarly, in later that year, 56 people overdosed in Brooklyn, New York, and another 40 in Dallas, Texas.
Effects and symptoms of AM 2201 Use

Research conducted on experimental mice models has revealed that bradycardia, hypothermia, and catalepsy can occur when synthetic cannabinoids (SCs) are consumed. Unlike cannabis, which is generally considered safe, earlier generations of SCs were relatively benign compared to the newer, more potent examples. Initial side effects of early SCs included anxiety, paranoia, nausea, agitation, tachycardia, and acute psychosis. However, recent SCs have been linked to more severe consequences, including cardiotoxicity, respiratory depression, ischemia, seizures, and even death.
The increase in severe acute toxicity following SC use may be attributed to the overstimulation of CB1 receptors by potent analogues, particularly fluorinated SCs like AM-2201. Additionally, the acute clinical presentation observed in cases of SC toxicity may be due to serotonin syndrome, indicating unintended off-target activity. The new wave of fluorinated SCs also raises health concerns due to their route of administration (smoking), which generates toxic hydrofluoric acid, leading to acute risks like hypocalcaemia, cardiac arrhythmia, and pulmonary oedema. Fluoride poisoning can also lead to delayed, symptom-free onset with chronic risks of osteosclerosis, bronchial and pulmonary necrosis, and nephrotoxicity.
Several metabolic studies have shown that hydrolytic defluorination of the 5-fluoropentyl chain is a common metabolic route in many fluorinated SCs, including 5F-PB-22, AM-2201, XLR-11, and AM-694. Although many SC users are aware of the dangers associated with SC consumption, they continue to use these products for their novelty value or to avoid detection during workplace drug testing.
Side effects observed in people who have used synthetic cannabinoids include:
- High blood pressure
- Rapid heart rate
- Altered perception
- Suicidal thoughts
- Vomiting
- Violent behaviour
- Psychosis or delirium
- Cardiac arrhythmias
- Kidney damage
This Is Why Spice Is ILLEGAL
Street Names, Prices and Approximate Dosage
In a similar fashion to Spice, the trademark name Black Mamba was later used inappropriately to refer to various substances that were infused with potent cannabinoids or a combination thereof. However, initially, the product was marketed in a shiny green and black package and contained only AM-2201 as its active ingredient.

The trend of bioisosteric fluorine substitution in other structurally related SC designer drugs likely stemmed from anecdotal reports that AM-2201 could produce psychoactive effects in humans at submilligram doses. This led to the emergence of several dozen terminally fluorinated SCs reported by forensic laboratories worldwide, with the rate of emergence seemingly increasing over time.
However, synthetic cannabinoids are now illegal in most parts of the world, including the USA, Canada, the UK and Australia. It wasn’t always this way though, as they were previously sold as “smoke” or legal highs in countries where marijuana products were illegal. K2 and Spice were the most popular names for these compounds, and they were available both in brick-and-mortar and online stores. With their colourful packaging and catchy names, such as Joker, Black Mamba, Kush or Kronic, they were quite popular.
Interestingly, despite its high potency, an oral dose of 5 mg of AM-2201 did not produce any physical or psychotropic effects. This is likely due to a strong first-pass effect and slow absorption compared to inhalation, where high serum concentrations are quickly reached and facilitate central nervous system penetration. For those using AM-2201, the recommended dosage is light (250ug+), moderate (500ug+), or heavy (1mg+).
Figure 4 illustrates the chemical configurations found in the smoke produced from thermal decomposition of AM-2201.
Figure 4. Decoding the molecular compositions of AM-2201 artifacts produced under thermal duress. [12]
The cost of obtaining AM-2201 for laboratory purposes is $85 for 2mg and $757 for 50mg. However, on the streets, the prices are significantly lower, with 100g selling for $600, 500g for $1300, and 1000g for $2100.
Legal Status

DEA Schedule I substances are classified as having no currently accepted medical use in the United States, and a high potential for abuse, making them unsafe for medical supervision. Various countries, including Australia, have enacted laws to prohibit SC structural analogues and dissimilar functional agonists of CB1. However, the lack of international law consistency and variation in state laws creates legal loopholes for manufacturers to release increasingly innovative and potentially dangerous SCs, putting the end-users’ health at risk. As a profit-driven grey industry, the SC market cares little for end-user health. Fluorine is just another tool for clandestine chemists to evade law enforcement and exploit legal loopholes. Although fluorine incorporation into pharmaceuticals has produced many benefits and is rigorously assessed for safety, the same cannot be said for the illicit SCs generated through fluorine application, which are likely detrimental to user health. Preliminary metabolic studies support the release of various forms of fluorine into the body, which may be linked to the adverse effects exhibited by these SCs, including the increasing number of SC-related deaths. Prohibition is an insufficient solution due to limited resources available to forensic chemists and law enforcement agencies and the dynamic nature of the SC marketplace. Additionally, biological evaluation is necessary to inform the public of the dangers of experimental drugs and provide medical practitioners with the necessary information to treat users. The classification of AM-2201 in various countries includes Schedule II in Canada, Anlage II in Germany (authorized trade only, not prescriptible), Temporary Class in New Zealand, Class B in the UK, and Schedule I in the US.
Synthesis of AM 2201 [13]

AM-2201 is synthesized using (1H-indol-3-yl)(naphthalen-1-yl)methanone as the starting material, following the scheme shown in Figure 5. The synthesis involves a two-step, one-pot procedure where the indole is deprotonated using KOH in EtOH, followed by treatment with 1-bromo-5-fluoropentane and Na2SO4 in acetone. The final product, AM-2201, is obtained with an 86% yield after column chromatography.
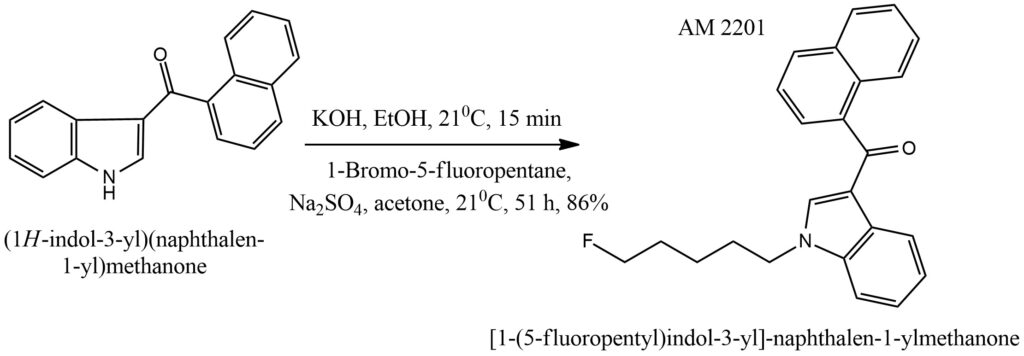
Figure 5. General scheme of AM 2201 synthesis
AM-2201 Horror Stories
Conclusion
Now it is clear that the use of AM-2201 and other synthetic cannabinoids poses significant risks to the health of users. While the dream of finding a safe and legal alternative to marijuana may have driven the initial development of these substances, the reality has been far from ideal. The dangerous health consequences associated with synthetic cannabinoids, including AM-2201, have resulted in legal prohibition of these substances in most parts of the world. Increased public awareness and education about the dangers of these substances are crucial in combating their use. Medical professionals must remain vigilant in monitoring and addressing the health consequences of synthetic cannabinoid use, and individuals seeking to use psychoactive substances must do so in a responsible and informed manner. It is hoped that the continued study and research of these substances will lead to safer and more effective alternatives in the future.
Bibliography
- https://en.wikipedia.org/wiki/AM-2201
- https://pubchem.ncbi.nlm.nih.gov/compound/53393997
- https://www.chemspider.com/Chemical-Structure.24751884.html
- https://www.biosynth.com/p/FA17340/335161-24-5-am-2201
- https://www.lgcstandards.com/GB/en/p/LGCFOR1396.02
- https://www.musechem.com/product/am-2201-i001893/
- Samuel D. Banister, Mark Connor The Chemistry and Pharmacology of Synthetic Cannabinoid Receptor Agonist New Psychoactive Substances: Evolution. New Psychoactive Substances, 2018, pp 191–226. DOI: 10.1007/164_2018_144 https://link.springer.com/chapter/10.1007/164_2018_144
- Shane M. Wilkinson, Samuel D. Banister Bioisosteric Fluorine in the Clandestine Design of Synthetic Cannabinoids. Australian Journal of Chemistry, 2014, 68, 1, pp. 4-8. https://doi.org/10.1071/CH14198 https://www.publish.csiro.au/ch/CH14198
- Robert Kronstrand, Markus Roman Toxicological Findings of Synthetic Cannabinoids in Recreational Users. Journal of Analytical Toxicology, 2013, Volume 37, Issue 8, pp. 534–541, https://doi.org/10.1093/jat/bkt068 https://academic.oup.com/jat/article/37/8/534/777811
- Samuel D. Banister, Jordyn Stuart etc. Effects of Bioisosteric Fluorine in Synthetic Cannabinoid Designer Drugs JWH-018, AM-2201, UR-144, XLR-11, PB-22, 5F-PB-22, APICA, and STS-135. ACS Chem. Neurosci., 2015, 6, 8, pp. 1445–1458. https://doi.org/10.1021/acschemneuro.5b00107 https://pubs.acs.org/doi/10.1021/acschemneuro.5b00107
- Tai, S., Fantegrossi, W.E. (2016). Pharmacological and Toxicological Effects of Synthetic Cannabinoids and Their Metabolites. In: Baumann, M., Glennon, R., Wiley, J. (eds) Neuropharmacology of New Psychoactive Substances (NPS). Current Topics in Behavioral Neurosciences, vol 32. Springer, Cham. https://doi.org/10.1007/7854_2016_60 https://link.springer.com/chapter/10.1007/7854_2016_60
- Melanie Hutter, Bjoern Moosmann Characteristics of the designer drug and synthetic cannabinoid receptor agonist AM-2201 regarding its chemistry and metabolism. J. Mass Spectrom., 2013, 48, pp. 885–894. https://doi.org/10.1002/jms.3229 https://analyticalsciencejournals.onlinelibrary.wiley.com/doi/full/10.1002/jms.3229
- Niki K. Burns, Trent D. Ashton Extraction, identification and detection of synthetic cannabinoids found pre-ban in herbal products in Victoria, Australia. Forensic Chemistry, 2018, 7, pp. 19–25. https://doi.org/10.1016/j.forc.2017.12.003 https://www.sciencedirect.com/science/article/abs/pii/S2468170917300164


