- The process of creating a drug - May 23, 2023
- How do scientists model mental disorders in animals? - May 23, 2023
Introduction
Acetone is a colorless, flammable liquid that is commonly used as an industrial solvent, a household cleaner, and in the manufacture of various products. Despite its widespread use, acetone can be hazardous if not handled properly. In this article, we will explore the different aspects of acetone, including its uses, safety precautions, synthesis, chemical and physical properties.
What is Acetone? Acetone Chemical and Physical Properties.
Acetone is a widely used colorless and volatile liquid belonging to the ketone family. Its chemical formula is C3H6O, with a molecular weight of 58.08 g/mol. The compound has a sweet and pungent odor, which is commonly associated with nail polish remover and other household products containing acetone. Acetone is miscible in water and most organic solvents, and it has a boiling point of 56.05 °C and a melting point of -94.7 °C.
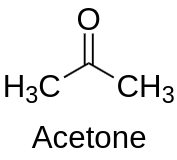
Acetone is highly reactive due to the presence of its carbonyl functional group, which makes it an excellent solvent for a wide range of organic and inorganic compounds. It has a high polarity, low viscosity, and low surface tension, which contribute to its excellent solvent properties. Acetone’s solvency capabilities make it a valuable ingredient in many industrial and commercial applications, including the manufacture of plastics, fibers, and pharmaceuticals.
Keto/enol tautomerism
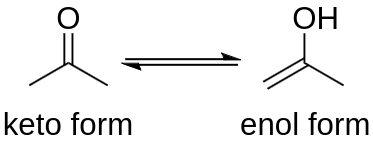
As is typical of ketones, acetone displays the keto–enol tautomeric transition in which the standard keto construction (CH3)2C=O of acetone itself is in balance with the enol isomer (CH3)C(OH)=(CH2) (prop-1-en-2-ol). In acetone vapor at ordinary temperature, only 2.4×10−7% of the particles are in the enol structure.
Aldol condensation
In the nearness of suitable catalysts, two acetone molecules can also combine to create the compound diacetone liquor (CH3)C=O(CH2)C(OH)(CH3)2, which on dehydration forms mesityl oxide (CH3)C=O(CH)=C(CH3)2. This substance can further join with another acetone particle, with the loss of another molecule of water, delivering phorone and other substances.
Polymerisation
It is reasonable to anticipate that acetone would also form polymers and (possibly cyclic) oligomers of two sorts. In one sort, units could be acetone molecules connected by ether bridges −O− obtained by from the opening of the double bond, to give a polyketal-like (PKA) chain [−O−C(CH3)2−]n. The other type could be acquired through successive aldol combination, with one molecule of water eliminated at each progression, delivering a poly(methylacetylene) (PMA) chain [−CH=C(CH3)−]n.
Synthesis of Acetone: Production and Manufacturing Methods
Acetone is produced directly or indirectly from propene. Approximately 83% of acetone is produced via the cumene process; as a result, acetone production is tied to phenol production. In the cumene process, benzene is alkylated with propylene to produce cumene, which is oxidized by air to produce phenol and acetone:
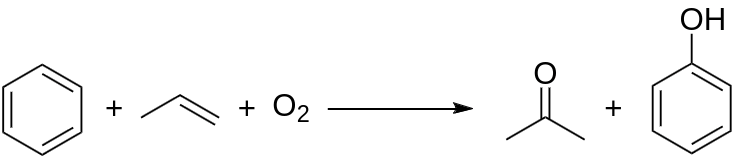
Other processes involve the direct oxidation of propylene (Wacker-Hoechst process), or the hydration of propylene to give 2-propanol, which is oxidized (dehydrogenated) to acetone.
Earlier, acetone was produced by the dry decomposition of acetates, such as calcium acetate in ketonic decarboxylation.
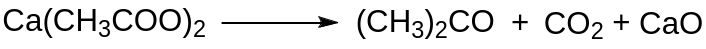
During WWI, acetone was manufactured with acetone-butanol-ethanol fermentation utilizing Clostridium acetobutylicum bacteria, developed by Chaim Weizmann (later the first president of Israel) to help the British in the production of Cordite. This acetone-butanol-ethanol fermentation was eventually stopped when newer techniques with higher yields were discovered.
Uses of Acetone: From Industrial Solvent to Household Cleaner
Acetone has a wide range of uses, from industrial solvents to household cleaners. Its excellent solvency properties make it a valuable ingredient in many industries, including the manufacture of plastics, fibers, and pharmaceuticals. Acetone is also used as a solvent for the production of various chemicals such as methyl methacrylate, bisphenol A, and methyl isobutyl ketone.

Moreover, acetone’s solvency properties also make it a common ingredient in household cleaning products, such as nail polish remover, paint thinner, and degreasers. It is a powerful solvent that can dissolve a variety of organic materials, including grease, oil, and wax. Acetone can also be used to remove adhesives, glue, and other sticky residues from surfaces.
Acetone is also commonly used in the beauty industry. It is a key ingredient in many nail polish removers, as it effectively removes nail polish from nails. It is also used as a thinner for nail polish, making it easier to apply and resulting in a smoother finish.
The Pros and Cons of Acetone: Benefits and Limitations
Acetone has several benefits and limitations that should be considered when using it in various applications. One of the significant benefits of acetone is its high solvency power. It is an excellent solvent for a broad range of organic and inorganic compounds, making it useful in various industries. It is also an effective cleaner for household and industrial applications, as it can dissolve grease, oils, and other organic residues.
However, acetone’s high volatility and flammability are notable limitations that should not be overlooked. Acetone has a low flashpoint, which makes it highly flammable and hazardous to use in environments with potential ignition sources. It can also pose health risks if inhaled, as it can irritate the eyes, nose, and throat, leading to headaches, dizziness, and nausea.
Another limitation of acetone is its potential for environmental harm. Acetone is highly volatile, and if not properly stored and handled, it can contribute to air pollution. Additionally, improper disposal of acetone can lead to contamination of water sources and soil.
Despite its limitations, acetone’s benefits make it a widely used solvent. It is cost-effective, easy to obtain, and useful in various industries and applications. To minimize its potential hazards, it is important to follow proper safety precautions when handling and using acetone, such as using adequate ventilation and appropriate personal protective equipment. Proper storage and disposal methods should also be followed to prevent environmental contamination.
Safety Precautions with Acetone: Handling, Storage, and Disposal
Proper safety precautions should be followed when handling, storing, and disposing of acetone due to its flammable and hazardous nature. When handling acetone, it is essential to wear appropriate personal protective equipment, including gloves, goggles, and a respirator, to avoid skin and eye irritation and respiratory issues. Adequate ventilation should also be provided to prevent the buildup of acetone vapors, which can ignite and cause an explosion in the presence of an ignition source.

Acetone should be stored in a cool, dry, and well-ventilated area away from ignition sources. It should be kept in a tightly sealed container made of compatible material, such as glass or metal. The storage area should be clearly labeled and secure, away from children and unauthorized personnel.
Disposal of acetone should be done in accordance with local regulations and guidelines. It should not be disposed of in the regular garbage or poured down the drain as it can contaminate the environment. Acetone should be properly packaged in a labeled, sealed, and compatible container and disposed of through an approved hazardous waste disposal service.
It is crucial to be aware of the hazards of acetone and to follow proper safety precautions to avoid accidents and environmental contamination. In case of accidental spills or exposure, it is important to seek medical attention and follow the appropriate emergency response procedures. By following these safety guidelines, the risks associated with handling, storing, and disposing of acetone can be minimized.
Acetone Alternatives: Sustainable and Safer Options
Acetone is a commonly used solvent, but due to its flammable and hazardous nature, many people are searching for sustainable and safer alternatives. There are several options available that can replace acetone in various applications.
One alternative is isopropyl alcohol, which has a lower toxicity level and is less flammable than acetone. It is commonly used as a disinfectant and cleaner and is safe for use on most surfaces.
Another alternative is soy-based solvents, which are derived from renewable sources and are biodegradable. They have low toxicity and do not release harmful vapors, making them a safe and environmentally friendly option.

D-limonene is another alternative solvent that is extracted from citrus peels. It is biodegradable and non-toxic and can be used as a cleaner and degreaser.

In addition to these alternatives, there are also water-based cleaners and solvents that can replace acetone in certain applications. These options are safer for the environment and do not pose health risks to users.
It is important to consider the specific application when choosing an acetone alternative and to ensure that it is compatible with the materials being cleaned or dissolved. By choosing a sustainable and safer option, the risks associated with handling and using acetone can be minimized, while also contributing to a healthier environment.
Conclusion
Acetone is a versatile and widely used solvent with numerous benefits and applications. However, it is essential to handle acetone with care and take appropriate safety precautions. As the demand for sustainable and safer alternatives grows, the use of acetone may decrease, and more environmentally friendly options may become more prevalent.
Bibliography
- Zakoshansky, V. M. “The cumene process for phenol-acetone production.” Petroleum Chemistry 47.4 (2007): 273-284. https://link.springer.com/article/10.1134/S096554410704007X
- Dietz, D. D., et al. “Toxicity Sudies of Acetone Administered in the Drinking Water of Rodents.” Toxicological Sciences 17.2 (1991): 347-360. https://academic.oup.com/toxsci/article-abstract/17/2/347/1716397
- Remler, R. F. “The Solvent Properties of Acetone.” Industrial & Engineering Chemistry 15.7 (1923): 717-720. https://pubs.acs.org/doi/pdf/10.1021/ie50163a023
- Brodka, A., and T. W. Zerda. “Properties of liquid acetone in silica pores: Molecular dynamics simulation.” The Journal of chemical physics 104.16 (1996): 6319-6326. https://aip.scitation.org/doi/abs/10.1063/1.471292
- Morgott, David A. “Acetone.” Patty’s toxicology (2001). https://onlinelibrary.wiley.com/doi/abs/10.1002/0471435139.tox074
- https://en.wikipedia.org/wiki/Acetone

