- The process of creating a drug - May 23, 2023
- How do scientists model mental disorders in animals? - May 23, 2023
Abstract
Leritine is a unique compound that falls under the category of organic compounds known as phenylpiperidines. These compounds are characterized by a phenylpiperidine skeleton, which consists of a piperidine molecule bonded to a phenyl group. Anileridine, on the other hand, is a potent analgesic with effects and applications similar to morphine. Leritine is a synthetic opioid medication that is widely used for its strong pain-relieving properties. It is classified as a narcotic pain reliever and is prescribed to manage moderate to severe pain. This article provides a comprehensive overview of Anileridine, including information on its general properties, pharmacology, effects, pricing, street names, symptoms of use, potential dangerous interactions, legal status, synthesis of ethyl 1-[2-(4-aminophenyl)ethyl]-4-phenylpiperidine-4-carboxylate, conclusion, and bibliography.
General Information About Anileridine [1-4]
Other synonyms names of Anileridine are: Ethyl 1-(p-aminophenethyl)-4-phenylisonipecotate; N-beta-(p-Aminophenyl)ethylnormeperidine; N-(beta-(p-Aminophenyl)ethyl)-4-phenyl-4-carbethoxypiperidine; Ethyl 1-(2-(4-aminophenyl)ethyl)-4-phenyl-4-piperidinecarboxylate; 1-(2-(4-Aminophenyl)ethyl)-4-phenyl-4-piperidinecarboxylic acid ethyl ester
IUPAC Name of Anileridine: ethyl 1-[2-(4-aminophenyl)ethyl]-4-phenylpiperidine-4-carboxylate
CAS number is 144-14-9
Related CAS are 126-12-5 (di-hydrochloride); 53421-22-0 (mono-hydrochloride)
Trade names are Leritine; Apodol (Bristol-Myers Squibb); Leritine (Merck); Alidine; Nipecotan; Adopol; Leritin; Apidol; Anileridinum; Leritines
Physico-Chemical Properties of Leritine [1-4]
- Molecular Formula C22H28N2O2
- Molar Weight 352.5 g/mol
- Boiling point 491.5±45.0 °C at 760 mmHg
- Melting Point 83 °C and 275-277 °C (decomposition)
- Solubility: Water Solubility 0.0124 mg/mL; in 1 in 2 parts of alcohol and 1 in 1 of chloroform
- Color/Form: white to yellowish white crystalline powder; Leritine Inj 25mg/ml Intramuscular, Intravenous, Subcutaneous Liquid; Leritine Oral Tablets 25mg/tab
- Odor: odorless to practically odorless
- Taste: slightly bitter taste
Structural formula present on Figure 1.
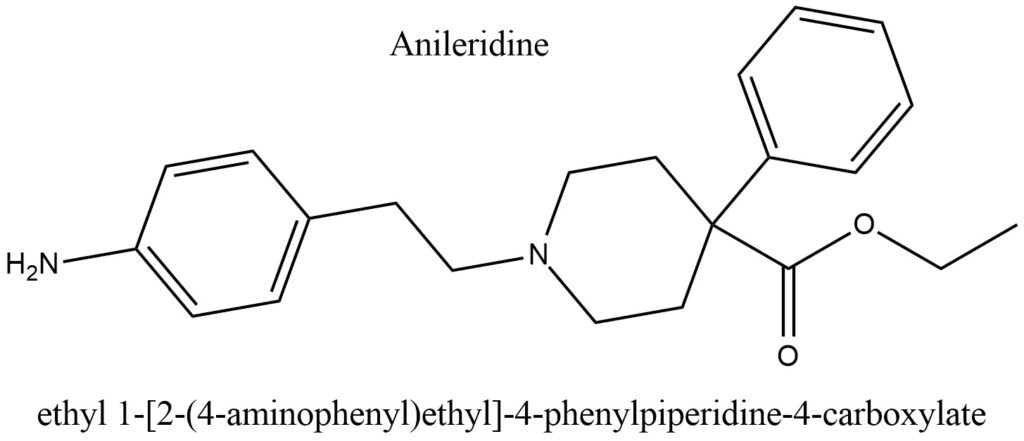
Figure 1. Structure of Anileridine

Figure 2. Apodol

Figure 3. Leritine
General Information, Pharmacology, Effects, Symptoms of Leritine Use [5-10]
General Information, Pharmacology
Anileridine is a powerful synthetic opioid medication known for its potent analgesic properties. It is commonly used as a narcotic pain reliever to treat moderate to severe pain. The main mechanism of action of anileridine is in the central nervous system (CNS), where it acts on opiate receptors coupled with G-protein receptors. These receptors function as both positive and negative regulators of synaptic transmission, and binding of anileridine stimulates the exchange of GTP for GDP on the G-protein complex. This results in a decrease in intracellular cAMP by inhibiting adenylate cyclase, leading to the inhibition of the release of nociceptive neurotransmitters such as substance P, GABA, dopamine, acetylcholine, and noradrenaline. Additionally, anileridine inhibits the release of vasopressin, somatostatin, insulin, and glucagon. Anileridine also has mild antihistaminic, spasmolytic, and antitussive effects. Furthermore, anileridine has been shown to close N-type voltage-operated calcium channels (OP2-receptor agonist) and open calcium-dependent inwardly rectifying potassium channels (OP3 and OP1 receptor agonist), resulting in hyperpolarization and reduced neuronal excitability. Anileridine may also be used as an adjunct in general anesthesia to reduce the amount of anesthetic needed, facilitate relaxation, and reduce laryngospasm, similar to meperidine. Respiratory depression, if it occurs, is of shorter duration compared to morphine or meperidine when equipotent analgesic doses are used.
Effects, Symptoms, Dosage, Price, Street Name of Leritine Use [9, 10]
Common side effects of anileridine may include stomach upset, nausea, vomiting, blurred vision, drowsiness, dizziness, dry mouth, or constipation. If these side effects persist or become bothersome, it is important to inform your doctor. Additionally, you should notify your doctor if you experience rapid heart rate, fainting, mental confusion, nervousness, irritability, or seizures. In rare cases, an allergic reaction to anileridine may occur, which can present as a rash, itching, swelling, dizziness, or trouble breathing. If you suspect an allergic reaction, seek immediate medical attention.
It’s important to note that anileridine is known on the street as “Leritines.” The pharmacy cost of anileridine is typically around $0.35 per tablet, while the street price may range from $3 to $5 per tablet. The estimated minimum lethal dose of anileridine is 0.5 g.
Anileridine typically takes effect within 15 minutes of oral or intravenous administration and lasts for about 2-3 hours. The studied dose for anileridine is typically up to 450 mg multiple times orally per day as the maximum total daily dose.
PHARMACOLOGY; NARCOTIC ANALGESICS by Professor Fink
Dangerous interactions

Figure 4. Drugs
Overexposure to this medication may result in symptoms such as dizziness, perspiration, warmth, dry mouth, visual difficulty, itching, euphoria, restlessness, nervousness, and excitement. It is important to inform your doctor if you have a history of lung diseases, liver problems, heart disease, thyroid disease, Addison’s disease, a seizure disorder, drug/alcohol abuse, or drug allergies.
Caution should be exercised when engaging in activities that require alertness, such as driving, as this medication may cause drowsiness. Limiting alcohol intake is also advised as it may intensify the dizziness and drowsiness effects of the medication. Elderly patients may be more sensitive to the effects of the drug and should use it with caution.
If you are pregnant, this medication should only be used when clearly needed, and you should discuss the risks and benefits with your doctor. It is not known if this medication is excreted into breast milk, so consulting your doctor before breastfeeding is recommended.
When taking Leritine, caution should be taken when coadministering with other opioids, sedatives, phenothiazines, or anesthetics, as these agents may increase respiratory and circulatory depression.
Symptoms of an overdose may include cold and clammy skin, low body temperature, slowed breathing, slowed heartbeat, drowsiness, dizziness, lightheadedness, deep sleep, and loss of consciousness.
The cause of death in cases of combined intoxication from anileridine, pethidine, and ethyl alcohol is known as “intoxieatio eombinata” anileridini, petidini, alcoholi aethylici.
Legal Status
Anileridine is not currently produced in the United States or Canada. It is classified as a Schedule II controlled substance under the Controlled Substances Act of 1970 in the United States, with an aggregate manufacturing quota of zero as of 2014 (ACSCN 9020). It is also internationally controlled under United Nations treaties.
In Australia, Anileridine is classified as a Schedule 8 controlled drug.
In Canada, it is classified as a Schedule I controlled substance.
In Germany, Anileridine is listed as Anlage I, which means it is authorized for scientific use only.
Synthesis of ethyl 1-[2-(4-aminophenyl)ethyl]-4-phenylpiperidine-4-carboxylate [11]
This compound falls within the phenylpiperidines class of organic compounds. Phenylpiperidines are characterized by a phenyl group attached to a piperidine skeleton.
To synthesize this compound, a mixture of 6-(p-aminophenyl)ethyl chloride hydrochloride, 4-phenyl-4-carboethoxy-piperidine carbonate, sodium bicarbonate, and anhydrous ethanol is combined, stirred, and heated under reflux for approximately 40 hours. The resulting mixture is then concentrated under vacuum until dry. The residual material is then treated with water, decanted, washed with additional water, and dried under vacuum, yielding N-(B-(p-aminophenyl)ethyl]-4-phenyl-4-carboethoxypiperidine.
The synthesis is carried out according to the scheme shown in Figure 5, using starting materials as described.
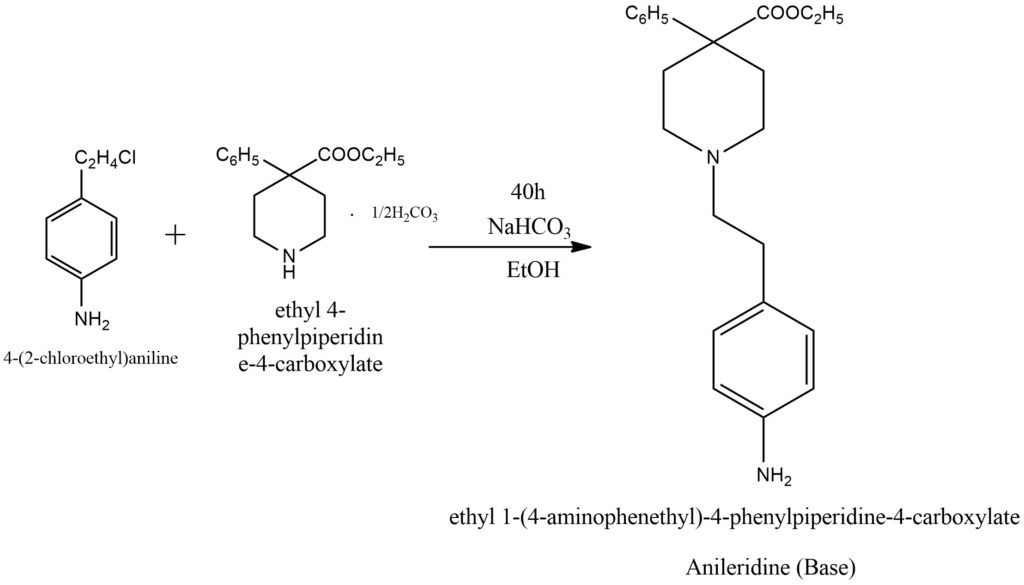
Figure 5. General scheme of Anileridine base synthesis
Conclusion
Leritine, also known as Anileridine, is a synthetic opioid and potent analgesic medication used for the relief of moderate to severe pain. Similar to morphine, it carries risks of adverse effects, dangerous interactions, and legal consequences if misused.
Anileridine acts on the central nervous system (CNS) to relieve pain, but it also has potential side effects resulting from its CNS actions. It is important to be aware of the potential risks associated with its use. Anileridine is sometimes referred to by the street name “Leritines” and has a pharmacy cost of approximately $0.35 per tablet, but its street price can range from $3 to $5 per tablet.
It is important to note that the estimated minimum lethal dose of anileridine is 0.5 g, and combining anileridine with other substances such as pethidine and ethyl alcohol can result in combined intoxication, leading to serious health consequences or even death.
Bibliography
- https://en.wikipedia.org/wiki/Anileridine
- https://pubchem.ncbi.nlm.nih.gov/compound/8944
- https://www.chemspider.com/Chemical-Structure.8600.html
- https://go.drugbank.com/drugs/DB00913
- https://web.archive.org/web/20120328085403/http://www.medicinenet.com/anileridine-oral_tablet/article.htm
- http://smpdb.ca/view/SMP0000674?highlight[compounds][]=DB00913&highlight[proteins][]=DB00913
- http://www.medicinenet.com/anileridine-oral_tablet/article.htm
- Drugs of Abuse A DEA RESOURCE GUIDE. 2017 https://www.dea.gov/sites/default/files/drug_of_abuse.pdf
- A Sajan The street value of prescription drugs. CMAJ, 1998, 159, pp. 139-42. https://www.cmaj.ca/content/cmaj/159/2/139.full.pdf
- Alha, A., Karlsson, M. & Korte, T. Fatal combined anileridine-pethidine poisoning. A gas chromatography, thin layer chromatography and mass spectrometry investigation. Z Rechtsmed 75, 293–298 (1975). https://doi.org/10.1007/BF00201184 https://link.springer.com/article/10.1007/BF00201184
- Weijlard, J.and Pfister, K., Ill; US. Patent 2,966,490; December 27, 1960; assigned to Merck & Co., Inc. https://patents.google.com/patent/US2966490A/en

